A) methanol
B) phenyl methanol
C) 2,2-dimethyl butanal
D) phenyl ethanol
Correct Answer: D
Solution :
Only those aldehydes in which a-hydrogen atoms are not present, give Cannizaro reaction. The structure of the given compounds is \[\underset{\begin{smallmatrix} methanal \\ (no\,\alpha -H) \end{smallmatrix}}{\mathop{HCHO}}\,\]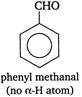 \[\underset{(no\text{ }\alpha -H\text{ }atom)}{\mathop{C{{H}_{3}}C{{H}_{2}}\underset{\begin{smallmatrix} | \\ C{{H}_{3}} \end{smallmatrix}}{\overset{\begin{smallmatrix} C{{H}_{3}} \\ | \end{smallmatrix}}{\mathop{C\overset{\alpha }{\mathop{{}}}\,}}}\,CHO}}\,\]
\[\underset{(no\text{ }\alpha -H\text{ }atom)}{\mathop{C{{H}_{3}}C{{H}_{2}}\underset{\begin{smallmatrix} | \\ C{{H}_{3}} \end{smallmatrix}}{\overset{\begin{smallmatrix} C{{H}_{3}} \\ | \end{smallmatrix}}{\mathop{C\overset{\alpha }{\mathop{{}}}\,}}}\,CHO}}\,\] 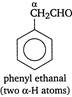 Thus, phenyl ethanal because of the presence of\[\alpha -H\] atoms, does not give Cannizaro reaction.
Thus, phenyl ethanal because of the presence of\[\alpha -H\] atoms, does not give Cannizaro reaction.
You need to login to perform this action.
You will be redirected in
3 sec
