A) mixture of secondary and tertiary alcohols
B) mixture of primary and secondary alcohols
C) secondary or tertiary alcohol
D) primary alcohol
Correct Answer: C
Solution :
\[C{{H}_{2}}=C{{H}_{2}}\xrightarrow{{{H}_{2}}O/{{H}^{+}}}\underset{{{1}^{o}}\text{alcohol}}{\mathop{C{{H}_{3}}C{{H}_{2}}OH}}\,\] \[C{{H}_{3}}-CH=C{{H}_{2}}\xrightarrow{{{H}_{2}}O/{{H}^{+}}}C{{H}_{3}}\underset{OH}{\mathop{\underset{|}{\mathop{C}}\,}}\,HC{{H}_{3}}\] \[{{2}^{o}}\]alcohol through \[{{2}^{o}}\] carbocation \[(C{{H}_{3}}\overset{\oplus }{\mathop{C}}\,HC{{H}_{3}})\] \[C{{H}_{3}}-\underset{C{{H}_{3}}}{\mathop{\underset{|}{\mathop{C}}\,}}\,=C{{H}_{2}}\xrightarrow{{{H}_{2}}O/{{H}^{+}}}{{(C{{H}_{3}})}_{3}}COH\] \[{{3}^{o}}\]alcohol through \[{{3}^{o}}\] carbocation\[[{{(C{{H}_{3}})}_{3}}\overset{\oplus }{\mathop{C}}\,]\] \[C{{H}_{3}}-\overset{C{{H}_{3}}}{\mathop{\overset{|}{\mathop{C}}\,}}\,H-CH=C{{H}_{2}}\xrightarrow{{{H}_{2}}O/{{H}^{+}}}\]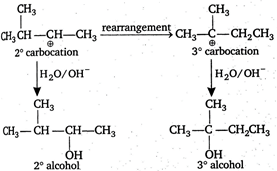 Thus, best alternate is
Thus, best alternate is
You need to login to perform this action.
You will be redirected in
3 sec
