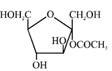
A)
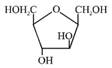
B)
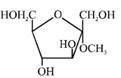
C)
D)
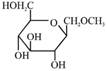
Correct Answer: A
Solution :
[a] Ester in presence of Aqueous KOH solution give SNAE reaction so following reaction takes place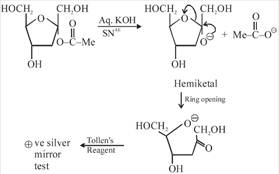 [b] In above compound in presence of Aq. KOH \[\text{(S}{{\text{N}}^{\text{AE}}}\text{)}\] reaction takes place \[\propto -\]Hydroxy carbonyl compound is formed which give \[\oplus \text{ve}\]Tollen's test So this compound behave as reducing sugar in an aqueous KOH solution.
[b] In above compound in presence of Aq. KOH \[\text{(S}{{\text{N}}^{\text{AE}}}\text{)}\] reaction takes place \[\propto -\]Hydroxy carbonyl compound is formed which give \[\oplus \text{ve}\]Tollen's test So this compound behave as reducing sugar in an aqueous KOH solution.
You need to login to perform this action.
You will be redirected in
3 sec
