A) \[\text{2}\text{.35}\times \text{1}{{\text{0}}^{2}}\,\,N/{{m}^{2}}\]
B) \[4.70\times {{10}^{2}}\,\,N/{{m}^{2}}\]
C) \[2.35\times {{10}^{3}}\,\,N/{{m}^{2}}\]
D) \[4.70\times {{10}^{3}}\,\,N/{{m}^{2}}\]
Correct Answer: C
Solution :
| [c] |
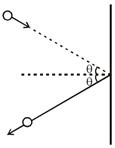 |
| Force = rate of change of momentum |
| (Perpendicular to area) |
| \[=n(2mu\,\,\cos \theta )\] |
| Pressure\[=\frac{Force}{Area}=\frac{n(2mu\,\,\cos \,\,\theta )}{A}\] |
| \[=\frac{3.32}{\sqrt{2}}\times \frac{{{10}^{-1}}}{{{10}^{-4}}}=\frac{3.32}{1.41}\times {{10}^{3}}=2.35\times {{10}^{3}}N/{{m}^{2}}\] |
You need to login to perform this action.
You will be redirected in
3 sec
