A) square planar
B) pyramidal
C) tetrahedral
D) trigonal planar
Correct Answer: B
Solution :
Hydrides of VA elements (\[N{{H}_{3}},\,P{{H}_{3}}\]etc.) have pyramidal structures. The central atoms in these undergo \[s{{p}^{3}}\text{-}\]hybridisation and these are surrounded by \[3\sigma bps\] and \[1-lps\].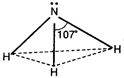
You need to login to perform this action.
You will be redirected in
3 sec
