question_answer 1) A radioactive element \[{}_{90}{{X}^{238}}\] decays into \[{}_{83}{{Y}^{222}}.\] The number of\[\beta \text{-}\]particles emitted are
A)
1
done
clear
B)
2
done
clear
C)
4
done
clear
D)
6
done
clear
View Answer play_arrow
question_answer 2) Minimum excitation potential of Bohr's firs orbit in hydrogen atom is
A)
3.6V
done
clear
B)
10.2V
done
clear
C)
13.6 V
done
clear
D)
3.4 V
done
clear
View Answer play_arrow
question_answer 3) A gas expands 0.25 \[{{m}^{3}}\] at constant pressure W\[{{10}^{3}}N\text{/}{{m}^{2}},\]the work done is
A)
250 N
done
clear
B)
250 W
done
clear
C)
250 J
done
clear
D)
2.5 erg
done
clear
View Answer play_arrow
question_answer 4) The work done in increasing the size of a soap film for\[10\,cm\times 6\,m\]to \[10\,cm\times 11\,cm\]is\[3\times {{10}^{-4}}J.\] The surface tension of the film is
A)
\[1.0\times {{10}^{-2}}\text{ }N\text{/}m\]
done
clear
B)
\[6.0\times {{10}^{-2}}\text{ }N\text{/}m\]
done
clear
C)
\[3.0\times {{10}^{-2}}\,N\text{/}m\]
done
clear
D)
\[1.5\times {{10}^{-2}}\text{ }N\text{/}m\]
done
clear
View Answer play_arrow
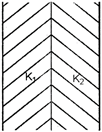
question_answer 5)
A parallel plate condenser is filled with two dielectrics as shown in figure. Area of each pate is \[A\,{{m}^{2}}\] and the separation is d metre. The dielectric constants are\[{{K}_{1}}\]and\[{{K}_{2}}\]respectively. Its capacitance in farad will be
A)
\[\frac{2{{\varepsilon }_{0}}A}{d}\left( \frac{{{K}_{1}}+{{K}_{2}}}{{{K}_{1}}{{K}_{2}}} \right)\]
done
clear
B)
\[\frac{2{{\varepsilon }_{0}}A}{d}\left( \frac{{{K}_{1}}{{K}_{2}}}{{{K}_{1}}+{{K}_{2}}} \right)\]
done
clear
C)
\[\frac{{{\varepsilon }_{0}}A}{d}\left( \frac{{{K}_{1}}+{{K}_{2}}}{2{{K}_{1}}{{K}_{2}}} \right)\]
done
clear
D)
\[\frac{{{\varepsilon }_{0}}A{{K}_{1}}{{K}_{2}}}{2\,({{d}_{2}}{{K}_{1}}+{{d}_{1}}{{K}_{2}})}\]
done
clear
View Answer play_arrow
question_answer 6) A transverse wave is represented by the equation \[y={{y}_{0}}\sin \frac{2\pi }{\lambda }(vt-k)\] For what value of \[\lambda \] is the particle velocity equal to two times the wave velocity.
A)
\[\lambda =\pi {{y}_{0}}\]
done
clear
B)
\[\lambda =\frac{\pi {{y}_{0}}}{2}\]
done
clear
C)
\[\lambda =\frac{\pi {{y}_{0}}}{3}\]
done
clear
D)
\[\lambda =\frac{2\pi }{{{y}_{0}}}\]
done
clear
View Answer play_arrow
question_answer 7) Ionisation potential of hydrogen atom is 13.6 eV. Hydrogen atom on the ground state rarely excited by monochromatic radiation of photon 12.1 eV. The special line emitted by a hydrogen atom according to Bohr's theory will be
A)
one
done
clear
B)
two
done
clear
C)
three
done
clear
D)
four
done
clear
View Answer play_arrow
question_answer 8) The internal resistance of primary cell is 4\[\Omega \]. It generates a current of 0.2 A in an external resistance of 21\[\Omega \]. The rate at which chemical energy to consumed in providing current is
A)
1 J/s
done
clear
B)
5 J/s
done
clear
C)
0.42 J/s
done
clear
D)
0.8 J/s
done
clear
View Answer play_arrow
question_answer 9) The binding energy per nucleon is maximum in the case
A)
\[{}_{92}^{235}U\]
done
clear
B)
\[{}_{56}^{141}Ba\]
done
clear
C)
\[{}_{26}^{56}Fe\]
done
clear
D)
\[{}_{4}^{2}He\]
done
clear
View Answer play_arrow
question_answer 10) Two rigid bodies A and B rotate with rotational kinetic energies \[{{E}_{A}}\] and \[{{E}_{B}}\] respectively. The moments of inertia of A and B about the axis of rotation are \[{{I}_{A}}\] and \[{{I}_{B}}\] respectively. If \[{{I}_{A}}=\frac{{{I}_{B}}}{4}\] and \[{{E}_{A}}=100={{E}_{B}},\] the ratio of angular momentum \[({{L}_{A}})\] of A to the angular momentum \[({{L}_{B}})\] of B is
A)
25
done
clear
B)
5/4
done
clear
C)
5
done
clear
D)
1/4
done
clear
View Answer play_arrow
question_answer 11) A car of mass 1000 kg moves on a circular track of radius 40 m. If the coefficient of friction is 1.28. The maximum velocity with which the car can be moved, is
A)
\[22.4\text{ }m\text{/}s\]
done
clear
B)
\[112\text{ }m\text{/}s\]
done
clear
C)
\[\frac{0.64\times 40}{1000\times 100}\,m\text{/}s\]
done
clear
D)
\[1000\,\,m\text{/}s\]
done
clear
View Answer play_arrow
question_answer 12) The escape velocity for the earth is 11.2 km/s, The mass of another planet 100 times mass of earth and its radius is 4 times radius of the earth. The escape velocity for the planet is
A)
280 km/s
done
clear
B)
56.0 km/s
done
clear
C)
112 km/s
done
clear
D)
56 km/s
done
clear
View Answer play_arrow
question_answer 13) Light travels faster in air than that in glass. This is accordance with
A)
wave theory of light
done
clear
B)
corpuscular theory of light
done
clear
C)
neither (a) nor (b)
done
clear
D)
Both (a) and (b)
done
clear
View Answer play_arrow
question_answer 14) The speed of air flow on the upper and lower surfaces of a wing of an aeroplane are \[{{v}_{1}}\] and \[{{v}_{2}}\] respectively. If A is the cross-section area of the wing and \[\rho \] is the density of air, then the upward life is
A)
\[\frac{1}{2}\rho A({{v}_{2}}-{{v}_{1}})\]
done
clear
B)
\[\frac{1}{2}\rho A({{v}_{1}}+{{v}_{2}})\]
done
clear
C)
\[\frac{1}{2}\rho A(v_{1}^{2}-v_{2}^{2})\]
done
clear
D)
\[\frac{1}{2}\rho A(v_{1}^{2}+v_{2}^{2})\]
done
clear
View Answer play_arrow
question_answer 15) A body is thrown with a velocity of 9.8 m/s making an angle of \[30{}^\circ \]with the horizontal. It will hit the ground after a time
A)
1.5 s
done
clear
B)
1 s
done
clear
C)
3s
done
clear
D)
2 s
done
clear
View Answer play_arrow
question_answer 16) At room temperature, copper has free electron density of\[8.4\times {{10}^{28}}{{m}^{-3}}\]. The electron drift velocity in a copper conductor of cross-sectional area of \[{{10}^{-6}}{{m}^{2}}\] and carrying a current of 5.4 A, will be
A)
\[4\,m{{s}^{-1}}\]
done
clear
B)
\[0.4\,m{{s}^{-1}}\]
done
clear
C)
\[4\,cm\,{{s}^{-1}}\]
done
clear
D)
\[0.4\,\,mm\,{{s}^{-1}}\]
done
clear
View Answer play_arrow
question_answer 17) A uniform wire of resistance R and length L is cut into four equal parts, each of length L/4 which are then connected in parallel combination. The effective resistance of the combination will be
A)
\[R\]
done
clear
B)
\[4R\]
done
clear
C)
\[\frac{R}{4}\]
done
clear
D)
\[\frac{R}{16}\]
done
clear
View Answer play_arrow
question_answer 18) The half-life of radio isotope is 4 h. If initial mass of the isotope was 200 g, then mass remaining after 24 h will be
A)
1.042 g
done
clear
B)
2.084 g
done
clear
C)
3.125 g
done
clear
D)
4.167 g
done
clear
View Answer play_arrow
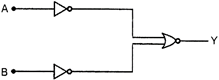
question_answer 19)
Which logic gate is represented by the following combination of logic gates?
A)
OR
done
clear
B)
NOR
done
clear
C)
AND
done
clear
D)
NAND
done
clear
View Answer play_arrow
question_answer 20) The work function for metals A, B and C are respectively 1.92 eV, 2.0 eV and 5 eV. According to Einstein's equation the metals which will emit photo, electrons for a radiation of wavelength \[4100\text{ }\overset{\text{o}}{\mathop{\text{A}}}\,\] is/are
A)
none
done
clear
B)
A only
done
clear
C)
A and B only
done
clear
D)
All the three metals
done
clear
View Answer play_arrow
question_answer 21) A luminous object is placed at a distance of 30 cm from the convex lens of focal length 20 cm. On the other side of the lens, at what distance from the lens a convex mirror of radius of curvature 10 cm be placed in order to have an upright image of the object coincident with it.
A)
30 cm
done
clear
B)
60 cm
done
clear
C)
50 cm
done
clear
D)
12 cm
done
clear
View Answer play_arrow
question_answer 22) A battery of emf 10 V and internal resistance of 0.5 ohm is connected across a variable resistance R. The maximum value of R is given by
A)
0.5\[\Omega \]
done
clear
B)
1.00\[\Omega \]
done
clear
C)
2.0\[\Omega \]
done
clear
D)
0.25\[\Omega \]
done
clear
View Answer play_arrow
question_answer 23) For a gas \[\frac{R}{{{C}_{v}}}\]= 0.67. This gas is made up of molecules which are
A)
mono atomic
done
clear
B)
poly atomic
done
clear
C)
mixture of diatomic and poly atomic molecules
done
clear
D)
diatomic
done
clear
View Answer play_arrow
question_answer 24) A point source of light is placed 4 m below the surface of water of refractive index 5/3. The minimum diameter of a disc which should be placed over the source on the surface of water to cut-off all light coming out of water is
A)
6m
done
clear
B)
3 m
done
clear
C)
4m
done
clear
D)
2m
done
clear
View Answer play_arrow
question_answer 25) A moving body of mass m and velocity 3 km/h collides with a rest body of mass 2 m and stick to it. Now the combined mass starts to move. What will be the combined velocity?
A)
4 km/h
done
clear
B)
1 km/h
done
clear
C)
2 km/h
done
clear
D)
3 km/h
done
clear
View Answer play_arrow
question_answer 26) Two boys are standing at the ends A and B of a ground, where AB = a. The boy at B starts running in a direction perpendicular to AB with velocity\[{{v}_{1}}\]? The boy at A starts running simultaneously with velocity\[v\]and catches the other boy in a time t, where t is
A)
\[\frac{a}{\sqrt{{{v}^{2}}+v_{1}^{2}}}\]
done
clear
B)
\[\sqrt{\frac{{{a}^{2}}}{{{v}^{2}}-v_{1}^{2}}}\]
done
clear
C)
\[\frac{a}{(v-{{v}_{1}})}\]
done
clear
D)
\[\frac{a}{(v+{{v}_{1}})}\]
done
clear
View Answer play_arrow
question_answer 27) A 5 amp fuse wire can withstand a maximum power of 1 W in circuit. The resistance of the fuse wire is
A)
0.2\[\Omega \]
done
clear
B)
\[5\,\,\Omega \]
done
clear
C)
0.4\[\Omega \]
done
clear
D)
0.04 \[\Omega \]
done
clear
View Answer play_arrow
question_answer 28) A force F is given \[F=at+b{{t}^{2}},\] where t is time. What are the dimensions of a and b?
A)
\[[ML{{T}^{-1}}]\]and \[[ML{{T}^{0}}]\]
done
clear
B)
\[[ML{{T}^{-3}}]\]and \[[M{{L}^{2}}{{T}^{4}}]\]
done
clear
C)
\[[ML{{T}^{-4}}]\] and \[[ML{{T}^{1}}]\]
done
clear
D)
\[[ML{{T}^{-3}}]\] and \[[ML{{T}^{-4}}]\]
done
clear
View Answer play_arrow
question_answer 29) Two equal negative charges - q are fixed at the point (0, a) and (0, - a) on the y-axis. A positive charge Q is released from rest at the point (2a, 0) on the x-axis. The charge will
A)
execute SHM about the origin
done
clear
B)
move to the origin and remain at rest
done
clear
C)
move to infinity
done
clear
D)
execute oscillatory but not SHM
done
clear
View Answer play_arrow
question_answer 30) An ice-cube of density 900 \[kg\text{/}{{m}^{3}}.\] is floating in water of density 1000\[kg\text{/}{{m}^{3}}.\] The percentage volume of ice-cube outside the water is
A)
20%
done
clear
B)
35%
done
clear
C)
10%
done
clear
D)
25%
done
clear
View Answer play_arrow
question_answer 31) In 0.2 s, the current in a coil increases from 2.0 A to 3.0 A. If inductance of coil is 60 mH then induced current in external resistance of \[3\,\Omega \] will be
A)
1 A
done
clear
B)
0.5 A
done
clear
C)
0.2 A
done
clear
D)
0.1 A
done
clear
View Answer play_arrow
question_answer 32) Two coherent light beams of intensities \[I\] and\[4I\]are superposed. The maximum and minimum possible intensities in the resulting beam are
A)
\[5I\] and \[I\]
done
clear
B)
\[5I\] and \[3I\]
done
clear
C)
\[9I\] and \[I\]
done
clear
D)
\[9I\] and \[3I\]
done
clear
View Answer play_arrow
question_answer 33) A galvanometer acting as a voltmeter should have
A)
low resistance in series with its coil
done
clear
B)
low resistance in parallel with its coil
done
clear
C)
high resistance in series with its coil
done
clear
D)
high resistance in parallel with its coil
done
clear
View Answer play_arrow
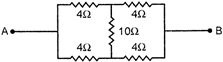
question_answer 34)
The equivalent resistance across A and B is
A)
2\[\Omega \]
done
clear
B)
3\[\Omega \]
done
clear
C)
4\[\Omega \]
done
clear
D)
5\[\Omega \]
done
clear
View Answer play_arrow
question_answer 35) A black body has a wavelength of \[\lambda \] temperature 2000 K. Its corresponding wavelength at temperature 3000 K will be
A)
\[\frac{2\lambda }{3}\]
done
clear
B)
\[\frac{3\lambda }{2}\]
done
clear
C)
\[\frac{4\lambda }{9}\]
done
clear
D)
\[\frac{5\lambda }{9}\]
done
clear
View Answer play_arrow
question_answer 36) The working principle of a ball point pen is
A)
Bernoulli's theorem
done
clear
B)
surface tension
done
clear
C)
gravity
done
clear
D)
viscosity
done
clear
View Answer play_arrow
question_answer 37) Progressive waves are represented by the equation \[{{y}_{1}}=a\sin \,(\omega \,t-x)\] and \[{{y}_{2}}=b\,\,\cos \,(\omega \,t-x)\] The phase difference between waves is
A)
\[0{}^\circ \]
done
clear
B)
\[45{}^\circ \]
done
clear
C)
\[90{}^\circ \]
done
clear
D)
\[180{}^\circ \]
done
clear
View Answer play_arrow
question_answer 38) Two simple pendulums of length 0.5 m and 3 m respectively are given small linear displacement in one direction at the same time. They will again be in the phase when the pendulum of shorter length has completed x oscillations, where k is
A)
1
done
clear
B)
3
done
clear
C)
2
done
clear
D)
5
done
clear
View Answer play_arrow
question_answer 39) A balloon contains 500\[{{m}^{3}}\] of helium at \[27{}^\circ C\] and 1 atmosphere pressure. The volume of the helium at \[-3{}^\circ C\] temperature and 0.5 atmosphere pressure will be
A)
1000\[{{m}^{3}}\]
done
clear
B)
900\[{{m}^{3}}\]
done
clear
C)
700\[{{m}^{3}}\]
done
clear
D)
500\[{{m}^{3}}\]
done
clear
View Answer play_arrow
question_answer 40) 220 V, 50 Hz, AC source is connected to an inductance of 0.2 H and a resistance of 20\[\Omega \] in series. What is the current in the circuit?
A)
3.33 A
done
clear
B)
33.3 A
done
clear
C)
5 A
done
clear
D)
10 A
done
clear
View Answer play_arrow
question_answer 41) The osmotic pressure of blood is 8.21 atm at\[37{}^\circ C\]. How much glucose would be used for a injection that is at the same osmotic pressure a blood?
A)
22.17\[g{{L}^{-1}}\]
done
clear
B)
58.14\[g{{L}^{-1}}\]
done
clear
C)
61.26\[g{{L}^{-1}}\]
done
clear
D)
75.43\[g{{L}^{-1}}\]
done
clear
View Answer play_arrow
question_answer 42) At equilibrium, the rate of dissolution of a solid solute in a volatile liquid solvent is
A)
less than the rate of crystallisation
done
clear
B)
greater than the rate of crystallisation
done
clear
C)
equal to the rate of crystallisation
done
clear
D)
zero
done
clear
View Answer play_arrow
question_answer 43) A chelating agent has two or more than two donor atoms to bind a single metal ion. Which of the following is not a chelating agent?
A)
Thiosulphato
done
clear
B)
Glycinato
done
clear
C)
Oxalato
done
clear
D)
Ethane-1, 2-diamine
done
clear
View Answer play_arrow
question_answer 44) On addition of small amount of\[KMn{{O}_{4}}\]to cone. \[{{H}_{2}}S{{O}_{4}},\] a green oily compound is obtained which is highly explosive in nature. Identify the compound from the following.
A)
\[M{{n}_{2}}{{O}_{7}}\]
done
clear
B)
\[Mn{{O}_{2}}\]
done
clear
C)
\[MnS{{O}_{4}}\]
done
clear
D)
\[M{{n}_{2}}{{O}_{3}}\]
done
clear
View Answer play_arrow
question_answer 45) The magnetic nature of elements depends on the presence of unpaired electrons. Identify the configuration of transition element, which shows highest magmetic moment.
A)
\[3{{d}^{7}}\]
done
clear
B)
\[3{{d}^{5}}\]
done
clear
C)
\[3{{d}^{8}}\]
done
clear
D)
\[3{{d}^{2}}\]
done
clear
View Answer play_arrow
question_answer 46) Among \[LiCl,\] \[BeC{{l}_{2}},\] \[BC{{l}_{3}}\] and \[CC{{l}_{4}},\] the covalent bond character follows the order
A)
\[LiCl<BeC{{l}_{2}}<BC{{l}_{3}}<CC{{l}_{4}}\]
done
clear
B)
\[BC{{l}_{3}}<CC{{l}_{4}}<BeC{{l}_{2}}<LiCl\]
done
clear
C)
\[BeC{{l}_{2}}<LiCl<CC{{l}_{4}}<BC{{l}_{3}}\]
done
clear
D)
\[CC{{l}_{4}}<BC{{l}_{3}}<BeC{{l}_{2}}<LiCl\]
done
clear
View Answer play_arrow
question_answer 47) Maximum number of electrons in a subshell of an atom determined by the following?
A)
\[4l+2\]
done
clear
B)
\[2{{n}^{2}}\]
done
clear
C)
\[4l-2\]
done
clear
D)
\[2l+1\]
done
clear
View Answer play_arrow
question_answer 48) The average kinetic energy or an ideal gas per molecule is SI units at \[25{}^\circ C\] will be
A)
\[6.17\times {{10}^{-21}}J{{K}^{-1}}\]
done
clear
B)
\[6.17\times {{10}^{-21}}\,kJ{{K}^{-1}}\]
done
clear
C)
\[6.17\times {{10}^{20}}\,J{{K}^{-1}}\]
done
clear
D)
\[7.16\times {{10}^{-20}}\,J{{K}^{-1}}\]
done
clear
View Answer play_arrow
question_answer 49) \[p{{K}_{a}}\] of acetic acid and \[p{{K}_{b}}\] of ammonium hydroxide are 4.76 and 4.75 respectively. Calculate the pH of ammonium acetate solution.
A)
6.02
done
clear
B)
7.005
done
clear
C)
8
done
clear
D)
5.602
done
clear
View Answer play_arrow
question_answer 50) The value of \[{{K}_{c}}\]for the reaction, \[2A\rightleftharpoons B+C\]is\[2\times {{10}^{-3}}\]. At a given time, if the composition of reaction mixture is [A] = [B] = [C] = \[3\times {{10}^{-3}}M\]. Which is true?
A)
The reaction will proceed in forward direction
done
clear
B)
The reaction will proceed in backward direction
done
clear
C)
The reaction will proceed in any direction
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 51) Which of the following elements can be involved in \[p\pi -d\pi \] bonding?
A)
Carbon
done
clear
B)
Nitrogen
done
clear
C)
Phosphorus
done
clear
D)
Boron
done
clear
View Answer play_arrow
question_answer 52) On addition of cone. \[{{H}_{2}}S{{O}_{4}}\] to a chloride salt, colourless fumes are evolved but in case of iodide salt, violet fumes come out. This is because
A)
\[{{H}_{2}}S{{O}_{4}}\] reduces\[HI\]to\[{{I}_{2}}\]
done
clear
B)
\[HI\] is of violet colour
done
clear
C)
\[HI\] gets oxidised to \[{{I}_{2}}\]
done
clear
D)
\[HI\] changes to\[HI{{O}_{3}}\]
done
clear
View Answer play_arrow
question_answer 53) Affinity for hydrogen decreases in the group from fluorine to iodine. Which of the halogen acids should have highest bond dissociation enthalpy?
A)
\[HF\]
done
clear
B)
\[HCl\]
done
clear
C)
\[HBr\]
done
clear
D)
\[HI\]
done
clear
View Answer play_arrow
question_answer 54) Which of the following statement is not correct about an inert electrode in a cell?
A)
It does not participate in the cell reaction.
done
clear
B)
It provides surface either for oxidation or for reduction reaction.
done
clear
C)
It provides surface for condition of electrons.
done
clear
D)
It provides surface for redox reaction.
done
clear
View Answer play_arrow
question_answer 55) Which of the following statement is correct?
A)
\[{{E}_{cell}}\] and \[{{\Delta }_{r}}G\] of cell reaction both are extensive properties.
done
clear
B)
\[{{E}_{cell}}\] and \[{{\Delta }_{r}}G\] of cell reaction both are intensive properties.
done
clear
C)
\[{{E}_{cell}}\] in the intensive property while \[{{\Delta }_{r}}G\] of cell reaction is an extensive property.
done
clear
D)
\[{{E}_{cell}}\] is an extensive property while \[{{\Delta }_{r}}G\] of cell reaction is an intensive property.
done
clear
View Answer play_arrow
question_answer 56) \[\underset{n-hexane}{\mathop{C{{H}_{3}}{{(C{{H}_{2}})}_{4}}C{{H}_{3}}}}\,\xrightarrow{Anhy.AlC{{l}_{3}}/HCl}\] \[\underset{2\text{-methyl}\,\,\text{pentane}}{\mathop{C{{H}_{3}}-\underset{\,C{{H}_{3}}}{\mathop{\underset{|}{\mathop{CH}}\,}}\,-{{(C{{H}_{2}})}_{2}}}}\,-C{{H}_{3}}\] \[+\underset{3\text{-}\,\text{methyl}\,\,\text{pentane}}{\mathop{C{{H}_{3}}C{{H}_{2}}-\underset{C{{H}_{3}}}{\mathop{\underset{\mathbf{|}}{\mathop{CH}}\,}}\,-C{{H}_{2}}-C{{H}_{3}}}}\,\] The above reaction is known as
A)
aromatisation
done
clear
B)
pyrolysis
done
clear
C)
isomerization
done
clear
D)
oxidation
done
clear
View Answer play_arrow
question_answer 57) Number of hydrogen-bonded water molecules associated in\[CuS{{O}_{4}}\cdot 5{{H}_{2}}O\]is
A)
one
done
clear
B)
two
done
clear
C)
three
done
clear
D)
All the five
done
clear
View Answer play_arrow
question_answer 58) Which of the following species do not show disproportionation on reaction? \[Cl{{O}^{-}},ClO_{2}^{-},ClO_{3}^{-}\] and \[ClO_{4}^{-}\]
A)
\[ClO_{4}^{-}\]
done
clear
B)
\[ClO_{3}^{-}\]
done
clear
C)
\[Cl{{O}^{-}}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 59) Which one of the following acts as a nucleophile?
A)
\[{{(C{{H}_{3}})}_{3}}N\]
done
clear
B)
\[B{{F}_{3}}\]
done
clear
C)
\[\overset{+}{\mathop{N{{O}_{2}}}}\,\]
done
clear
D)
\[C{{H}_{3}}-\overset{+}{\mathop{C}}\,=O\]
done
clear
View Answer play_arrow
question_answer 60) During estimation of nitrogen in the organic compound by Kjeldahl?s method, the ammonia evolved from 0.5 g of the compound in Kjeldahl?s estimation of nitrogen, neutralized 10 mL of 1M\[{{H}_{2}}S{{O}_{4}}.\] Find out the percentage of nitrogen in the compound.
A)
14%
done
clear
B)
28%
done
clear
C)
56%
done
clear
D)
68%
done
clear
View Answer play_arrow
question_answer 61) Excess fluoride (over 10 ppm) in drinking water can cause
A)
harmful effect of bones and teeth
done
clear
B)
methemoglobinemia
done
clear
C)
kidney damage
done
clear
D)
laxative effect
done
clear
View Answer play_arrow
question_answer 62) For the process to occur under adiabatic conditions, the correct conditions is
A)
\[\Delta T=0\]
done
clear
B)
\[\Delta p=0\]
done
clear
C)
\[q=0\]
done
clear
D)
\[W=0\]
done
clear
View Answer play_arrow
question_answer 63) \[\frac{3}{2}{{O}_{2}}(g)\to {{O}_{3}}(g);\]\[{{K}_{p}}\] for this reaction is \[2.47\times {{10}^{-29}}.\] At 298 K, \[{{\Delta }_{r}}G{}^\circ \]for conversation of oxygen to ozone will be
A)
\[100\text{ }kJ\,mo{{l}^{-1}}\]
done
clear
B)
\[150\text{ }kJ\,mo{{l}^{-1}}\]
done
clear
C)
\[163\text{ }kJ\,mo{{l}^{-1}}\]
done
clear
D)
\[2303\text{ }kJ\,mo{{l}^{-1}}\]
done
clear
View Answer play_arrow
question_answer 64) Which one of the following statements about \[{{C}_{2}}\] molecule is wrong?
A)
The bond order of \[{{C}_{2}}\] is 2.
done
clear
B)
In vapour phase, \[{{C}_{2}}\]molecule is diamagnetic.
done
clear
C)
Double bond in \[{{C}_{2}}\] molecule consists of both \[\pi \text{-}\]bonds because of the presence of \[4{{e}^{-}}_{s}\] in two \[\pi \text{-}\]molecular orbitals.
done
clear
D)
double bond in \[{{C}_{2}}\] molecule consists of one,\[\sigma \text{-}\]bond and on\[\pi \text{-}\]bond.
done
clear
View Answer play_arrow
question_answer 65) The type of hybridisation in\[S{{F}_{6}}\]. molecule is
A)
\[s{{p}^{3}}d\]
done
clear
B)
\[ds{{p}^{3}}\]
done
clear
C)
\[s{{p}^{3}}{{d}^{2}}\]
done
clear
D)
\[{{d}^{2}}s{{p}^{3}}\]
done
clear
View Answer play_arrow
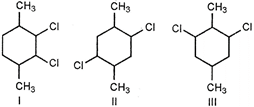
question_answer 66)
Which of the following compounds have highest melting point?
A)
Only I
done
clear
B)
Only II
done
clear
C)
I and II
done
clear
D)
II and III
done
clear
View Answer play_arrow
question_answer 67) Identify the major product 'X' obtained in the following reaction. 2, 3-dimethyl butan-2-\[\text{ol}\xrightarrow{+2Pb}X\]
A)
\[C{{H}_{3}}-\underset{C{{H}_{3}}}{\mathop{\underset{\mathbf{|}}{\mathop{C}}\,}}\,\underset{C{{H}_{3}}}{\mathop{\underset{\mathbf{|}}{\mathop{C}}\,}}\,-C{{H}_{3}}=\]
done
clear
B)
\[C{{H}_{3}}-\overset{C{{H}_{3}}}{\mathop{\overset{\mathbf{|}}{\mathop{CH}}\,}}\,-\overset{C{{H}_{3}}}{\mathop{\overset{\mathbf{|}}{\mathop{C}}\,}}\,=C{{H}_{2}}\]
done
clear
C)
\[C{{H}_{2}}=\underset{C{{H}_{3}}}{\mathop{\underset{\mathbf{|}}{\mathop{C}}\,}}\,-\underset{C{{H}_{3}}}{\mathop{\underset{\mathbf{|}}{\mathop{C}}\,}}\,=C{{H}_{2}}\]
done
clear
D)
\[C{{H}_{3}}-C{{H}_{2}}-\overset{C{{H}_{3}}}{\mathop{\overset{\mathbf{|}}{\mathop{C}}\,}}\,=C{{H}_{2}}\]
done
clear
View Answer play_arrow
question_answer 68) Addition of water to alkynes occurs in acidic medium and in the presence of\[H{{g}^{2+}}\]ions as a catalyst. Which of the following products will be formed on addition of water to but-1-yne under these conditions?
A)
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}CHO\]
done
clear
B)
\[C{{H}_{3}}C{{H}_{2}}COC{{H}_{3}}\]
done
clear
C)
\[C{{H}_{3}}C{{H}_{2}}COOH+C{{O}_{2}}\]
done
clear
D)
\[C{{H}_{3}}COOH+HCHO\]
done
clear
View Answer play_arrow
question_answer 69) The correct order of increasing acidic strength is
A)
phenol < ethanol < chloroacetic acid < acetic acid
done
clear
B)
ethanol < phenol < chloroacetic acid < acetic acid
done
clear
C)
ethanol < phenol < acetic acid < chloroacetic acid
done
clear
D)
chloroacetic acid < acetic acid < phenol < ethanol
done
clear
View Answer play_arrow
question_answer 70) KF has ccp structure. How many\[{{F}^{-}}\]ions and octahedral voids are there in this unit cell respectively?
A)
4 and 4
done
clear
B)
4 and 8
done
clear
C)
8 and 4
done
clear
D)
6 and 6
done
clear
View Answer play_arrow
question_answer 71) In the presence of a catalyst, the heat evolved or absorbed during the reaction
A)
increases
done
clear
B)
decreases
done
clear
C)
remains unchanged
done
clear
D)
may increase or decrease
done
clear
View Answer play_arrow
question_answer 72) The rate of a gaseous reaction is given by the expression k[A] [B]. If the volume of the reaction vessel is suddenly reduced to \[\frac{1}{4}\]th of the initial volume, the reaction rate relating to original rate will be
A)
\[\frac{1}{10}\]
done
clear
B)
\[\frac{1}{8}\]
done
clear
C)
8
done
clear
D)
16
done
clear
View Answer play_arrow
question_answer 73) Which of the following is \[3{}^\circ \] amine?
A)
1-methylcyclohexylamine
done
clear
B)
Triethyl amine
done
clear
C)
Tert-butylamine
done
clear
D)
N-methyl aniline
done
clear
View Answer play_arrow
question_answer 74) Which of the following enhances lathering property of soap?
A)
Sodium carbonate
done
clear
B)
Sodium rosinate
done
clear
C)
Sodium stearate
done
clear
D)
Trisodium phosphate
done
clear
View Answer play_arrow
question_answer 75) The deficiency of vitamin C causes
A)
scurvy
done
clear
B)
rickets
done
clear
C)
pyrrohea
done
clear
D)
pernicious anaemia
done
clear
View Answer play_arrow
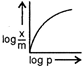
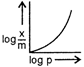
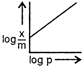
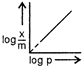
question_answer 76) Which of the following curves is in accordance with Freundlich adsorption isotherm?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
question_answer 77) A number of elements available in earth's crust but most abundant elements are
A)
Al and Fe
done
clear
B)
Al and Cu
done
clear
C)
Fe and Cu
done
clear
D)
Cu and Ag
done
clear
View Answer play_arrow
question_answer 78) The element which forms oxides in all oxidation states + 1 to + 5 is
A)
nitrogen
done
clear
B)
phosphorus
done
clear
C)
arsenic
done
clear
D)
antimony
done
clear
View Answer play_arrow
question_answer 79) Which of the following is the increasing order of enthalpy of vaporization?
A)
\[N{{H}_{3}},P{{H}_{3}},As{{H}_{3}}\]
done
clear
B)
\[As{{H}_{3}},P{{H}_{3}},N{{H}_{3}}\]
done
clear
C)
\[N{{H}_{3}},As{{H}_{3}},P{{H}_{3}}\]
done
clear
D)
\[P{{H}_{3}},As{{H}_{3}},N{{H}_{3}}\]
done
clear
View Answer play_arrow
question_answer 80) When \[B{{r}_{2}}\] is treated with aqueous solutions of \[NaF,\]\[NaCl,\]\[NaI\]separately
A)
\[{{F}_{2}},\]\[C{{l}_{2}}\] and \[{{I}_{2}}\] are liberated
done
clear
B)
only \[{{F}_{2}}\] and \[C{{l}_{2}}\] are liberated
done
clear
C)
only \[{{I}_{2}}\] is liberated
done
clear
D)
only \[C{{l}_{2}}\] is liberated
done
clear
View Answer play_arrow
question_answer 81) Pebrine is a disease of
A)
fish
done
clear
B)
honey bee
done
clear
C)
silk worm
done
clear
D)
lac insect
done
clear
View Answer play_arrow
question_answer 82) Factor govering the earth surface is
A)
topographic
done
clear
B)
edaphic
done
clear
C)
temperature
done
clear
D)
biotic
done
clear
View Answer play_arrow
question_answer 83) The direction of energy flow is
A)
Producers \[\to \] Herbivores \[\to \]Decomposers\[\to \]Omnivores
done
clear
B)
Producers\[\to \]Carnivore Herbivores\[\to \]Decomposers
done
clear
C)
Decomposers\[\to \] Carnivores \[\to \] Herbivores \[\to \] Producers
done
clear
D)
Producers \[\to \] Herbivores \[\to \] Carnivores \[\to \] Decomposers
done
clear
View Answer play_arrow
question_answer 84) If the Bengal tiger become extinct
A)
hyenas and wolves will become scarce
done
clear
B)
its gene pool will be lost forever
done
clear
C)
the wild areas will be safe far man and domestic
done
clear
D)
the population of beautiful animals like deers will get stabilized
done
clear
View Answer play_arrow
question_answer 85) Biological treatment of water pollution is done with the help of
A)
fungi
done
clear
B)
lichen
done
clear
C)
phytoplanktons
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 86) In photosynthesis carbon dioxide is converted to carbohydrates. It is a .......... process.
A)
reductive
done
clear
B)
oxidative
done
clear
C)
catabolic and exergonic
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 87) Which of the following is not an auxin?
A)
IAA
done
clear
B)
IBA
done
clear
C)
Zeatin
done
clear
D)
NAA
done
clear
View Answer play_arrow
question_answer 88) Which of the following properties is shown by cytokinins?
A)
Delay leaf senescence
done
clear
B)
Cause leaf abscission
done
clear
C)
Promote seed dormancy
done
clear
D)
Promote stomatal closing
done
clear
View Answer play_arrow
question_answer 89) Which of the following characteristics is are exhibited by \[{{C}_{4}}\]-plants? I. Kranz anatomy II. The first product of photosynthesis is oxaloacetic acid. III. Both PEP carboxylates and ribulose biphosphate carboxylate act as carbo-xylating enzymes. The correct option is
A)
I and III, but not II
done
clear
B)
I and II, but not III
done
clear
C)
II and III, but not I
done
clear
D)
II and III
done
clear
View Answer play_arrow
question_answer 90) Which of the following plant keeps its stomata open during night and closed during the day?
A)
Orchid
done
clear
B)
Cactus
done
clear
C)
Tea
done
clear
D)
Wheat
done
clear
View Answer play_arrow
question_answer 91) The structures present in the roots to absorb water and minerals is
A)
epidermal extensions
done
clear
B)
hypodermis
done
clear
C)
endodermis
done
clear
D)
epidermal appendages
done
clear
View Answer play_arrow
question_answer 92) Lady finger belongs to family
A)
Malvaceae
done
clear
B)
Cucurbitaceae
done
clear
C)
Brassicaceae
done
clear
D)
Liliaceae
done
clear
View Answer play_arrow
question_answer 93) The interxylary phloem is found in the stem of
A)
Cucurbita
done
clear
B)
Salvia
done
clear
C)
Calotropis
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 94) Wound healing is due to
A)
ventral meristem
done
clear
B)
secondary meristem
done
clear
C)
primary meristem
done
clear
D)
All of these
done
clear
View Answer play_arrow
question_answer 95) Angular collenchyma occurs in
A)
Salvia
done
clear
B)
Helianthus
done
clear
C)
Althaea
done
clear
D)
Cucurbita
done
clear
View Answer play_arrow
question_answer 96) Genetic dwarfism can be overcome by
A)
gibberellin
done
clear
B)
ethylene
done
clear
C)
auxin
done
clear
D)
ABA
done
clear
View Answer play_arrow
question_answer 97) Hormone inducing fruit ripening is
A)
cytokinin
done
clear
B)
ethylene
done
clear
C)
abscissic acid
done
clear
D)
gibberellic acid
done
clear
View Answer play_arrow
question_answer 98) The year 1900 AD is highly significant for geneticists due to
A)
discovery of genes
done
clear
B)
principles of linkage
done
clear
C)
chromosome theory of heredity
done
clear
D)
rediscovery of Mendelism
done
clear
View Answer play_arrow
question_answer 99) \[{{F}_{1}}\text{-}\]generation means
A)
first filial generation
done
clear
B)
first seed generation
done
clear
C)
first flowering generation
done
clear
D)
first fertile generation
done
clear
View Answer play_arrow
question_answer 100) Skin colour is controlled by
A)
single gene
done
clear
B)
3 pairs of genes
done
clear
C)
2 pairs of genes
done
clear
D)
2 pairs of genes with an intragene
done
clear
View Answer play_arrow
question_answer 101) Lactose is composed of
A)
glucose + glucose
done
clear
B)
glucose + galactose
done
clear
C)
glucose + fructose
done
clear
D)
fructose + galactose
done
clear
View Answer play_arrow
question_answer 102) Meiosis is best observed in dividing
A)
cell of lateral meristem
done
clear
B)
cells of apical meristem
done
clear
C)
microsporocytes
done
clear
D)
microspores and anther wall
done
clear
View Answer play_arrow
question_answer 103)
Study the following statements and select the correct option. I. Tapetum nourishes the developing pollen grains. II. Hilum represents the junction between ovule and funicle. III. In aquatic plants such as water hyacinth and water lily, pollination is by water IV. The primary endosperm nucleus is triploid
A)
I, II and IV are correct, but III is incorrect'
done
clear
B)
I and II are correct, but III and IV are incorrect
done
clear
C)
I and IV are correct, but II and III are incorrect
done
clear
D)
I, III and IV are correct, but I is incorrect
done
clear
View Answer play_arrow
question_answer 104) Masses of pollen grains, i.e., pollinia is found in
A)
Gramineae
done
clear
B)
Solanaceae
done
clear
C)
Orchidaceae
done
clear
D)
Malvaceae
done
clear
View Answer play_arrow
question_answer 105) Morphine, which is used as an analgesic is obtained from
A)
Taxus brevifolia
done
clear
B)
Berberis nilgluriensis
done
clear
C)
Cinchona offlcinalis
done
clear
D)
Papaver somnlferum
done
clear
View Answer play_arrow
question_answer 106) Bacterial endotoxin is
A)
a toxic protein that stays inside the bacterial cell
done
clear
B)
a toxic protein that is excreted into the medium
done
clear
C)
lipopolysaccharide located on the surface of the bacteria
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 107) Endosperm of gymnosperms is
A)
haploid
done
clear
B)
tetraploid
done
clear
C)
diploid
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 108) First vascular plant is
A)
thallophyta
done
clear
B)
pteridophyta
done
clear
C)
bryophyta
done
clear
D)
spermatophyte
done
clear
View Answer play_arrow
question_answer 109) Diatomaceous earth is obtained from
A)
Bacillarophyceae
done
clear
B)
Xanthophyceae
done
clear
C)
Rhodophyceae
done
clear
D)
Chrysophyceae
done
clear
View Answer play_arrow
question_answer 110) Which of the following is an epidermal cell containing chloroplast?
A)
Stomata
done
clear
B)
Hydathode
done
clear
C)
Guard cell
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 111) Which of the following cross will produce terminal flower in garden pea?
A)
\[AA\times Aa\]
done
clear
B)
\[AA\times aa\]
done
clear
C)
\[Aa\times Aa\]
done
clear
D)
\[Aa\times AA\]
done
clear
View Answer play_arrow
question_answer 112) Which one of the following pairs of plants are not seed producers?
A)
Funaria and Ficus
done
clear
B)
Fern and Funaria
done
clear
C)
Funaria and Pinus
done
clear
D)
Ficus and Chlamydomonas
done
clear
View Answer play_arrow
question_answer 113) Which one of the following is heterosporous?
A)
Equisetum
done
clear
B)
Dryopteris
done
clear
C)
Salvinia
done
clear
D)
Adiantum
done
clear
View Answer play_arrow
question_answer 114) Cycas revoluta is popularly known as
A)
sago palm
done
clear
B)
royal palm
done
clear
C)
date palm
done
clear
D)
sea palm
done
clear
View Answer play_arrow
question_answer 115)
Match the following with correct combination Column-I Column-II A. Cuscuta 1. Saprophyte B. Eichhomia 2. Pneumatophare C. Monotropa 3. Insectivorous plant D. Rhwphora 4. Parasite E. Utricularia 5. Root pocket
A)
Codes: A-3 B-1 C-5 D-4 E-2
done
clear
B)
A-2 B-3 C-1 D-5 E-4
done
clear
C)
A-4 B-3 C-1 D-5 E-2
done
clear
D)
A-4 B-5 C-1 D-2 E-3
done
clear
View Answer play_arrow
question_answer 116) In pteridophytes, phloem is without
A)
bast fibers
done
clear
B)
sieve tubes
done
clear
C)
companion cells
done
clear
D)
sieve cells
done
clear
View Answer play_arrow
question_answer 117)
Match the following entities of column-I with their respective orders of column-II and choose the correct combination form the option. Column-I Column-II A. Wheat 1. Primate B. Mango 2. Diptera C. Housefly 3. Sapindales D. Man 4. Poales
Codes:
A)
A-4 B-3 C-2 D-1
done
clear
B)
A-1 B-2 C-4 D-3
done
clear
C)
A-3 B-4 C-2 D-1
done
clear
D)
A-2 B-4 C-1 D-3
done
clear
View Answer play_arrow
question_answer 118) Agar-agar is produced by
A)
fungi
done
clear
B)
algae
done
clear
C)
bacteria
done
clear
D)
blue-green algae
done
clear
View Answer play_arrow
question_answer 119) In DNA, when AGCT occurs, their association is as per which of the following pair
A)
A-G, C-T
done
clear
B)
A-T, G-C
done
clear
C)
A-C, G-T, A-C, E-T
done
clear
D)
All of these
done
clear
View Answer play_arrow
question_answer 120) A segment of DNA has 120 adenine and cytosine bases. The total number of nucleotides present in the segment is
A)
60
done
clear
B)
240
done
clear
C)
120
done
clear
D)
480
done
clear
View Answer play_arrow
question_answer 121)
Which of the given option is correct regarding the statements? Statement I: Cephalochordata bears notochord all along the body throughout life. Statement II: Urochordate bears vertebral column only in tail region throughout the life.
A)
I wrong, II correct
done
clear
B)
I correct, II wrong
done
clear
C)
Both I and II are wrong
done
clear
D)
Both are correct
done
clear
View Answer play_arrow
question_answer 122) In which of the following haemocyanin pigment is found?
A)
Lower invertebrates
done
clear
B)
Echinodermata
done
clear
C)
Insecta
done
clear
D)
Annelida
done
clear
View Answer play_arrow
question_answer 123) Which of the following cells in earthworm play a role similar to liver in vertebrates?
A)
Amoebocytes
done
clear
B)
Mucocytes
done
clear
C)
Chloragogen cells
done
clear
D)
Epidermal cells
done
clear
View Answer play_arrow
question_answer 124)
Match the following and select the con option. List-I List-II A. Cyclostomes 1. Hemichordata B. Aves 2. Urochordata C. Tunicates 3. Agantha D. Balanoglossus 4. Pisces E. Osteichthyes 5. Tetrapod
Codes:
A)
A-3 B-5 C-2 D-1 E-4
done
clear
B)
A-3 B-1 C-5 D-2 E-4
done
clear
C)
A-1 B-2 C-3 D-4 E- 5
done
clear
D)
A-2 B-3 C-4 D-1 E-5
done
clear
View Answer play_arrow
question_answer 125) Chondrichthyes is characterised by
A)
placoid scale
done
clear
B)
placoid scale and ventral mouth
done
clear
C)
ventral mouth
done
clear
D)
ctenoid scale and ventral mouth
done
clear
View Answer play_arrow
question_answer 126) Soil salinity is measured by
A)
Porometer
done
clear
B)
Calorimeter
done
clear
C)
Conductivity meter
done
clear
D)
Potometer
done
clear
View Answer play_arrow
question_answer 127) Predation and parasitism are which type of interactions?
A)
(+, +)
done
clear
B)
(+, Q)
done
clear
C)
(-, -)
done
clear
D)
(+, -)
done
clear
View Answer play_arrow
question_answer 128) The ultimate source of energy for living beins is
A)
sunlight
done
clear
B)
ATP
done
clear
C)
fats
done
clear
D)
carbohydrates
done
clear
View Answer play_arrow
question_answer 129) Which of the following species are restricted to an area?
A)
Sympatric species
done
clear
B)
Sibling species
done
clear
C)
Allopatric species
done
clear
D)
Endemic species
done
clear
View Answer play_arrow
question_answer 130) Select the incorrect statement.
A)
Stellar's sea cow and passenger pigeon got extinct due to over exploitation by men
done
clear
B)
The mitotic convention on biological diversity was held in 1992
done
clear
C)
Species diversity increase as we move away from the equator towards the poles
done
clear
D)
Lantana and Eichhomia are invasive weed species in India
done
clear
View Answer play_arrow
question_answer 131) Drugs that cause malformation in developing embryo during pregancy are called
A)
teratogens
done
clear
B)
nicotine
done
clear
C)
tranquillizers
done
clear
D)
alcoholic beverages
done
clear
View Answer play_arrow
question_answer 132) Which set is similar?
A)
Corpus luteum - Graafian follicles
done
clear
B)
Sebum - Sweat
done
clear
C)
Vitamin-\[{{B}_{7}}\] - Niacin
done
clear
D)
Bundle of His - Pacemaker
done
clear
View Answer play_arrow
question_answer 133) Which one out of (a) to (d) given below correctly represents the structural formula of the basic ammo acid?
A)
\[H-\underset{\underset{O}{\mathop{\mathbf{/}}}\,\underset{\,OH}{\mathop{\mathbf{\backslash }}}\,}{\overset{N{{H}_{2}}}{\mathop{\underset{C}{\overset{\mathbf{|}}{\mathop{\underset{\mathbf{|}}{\mathop{\underset{C{{H}_{2}}}{\mathop{\underset{\mathbf{|}}{\mathop{\underset{C{{H}_{2}}}{\mathop{\underset{\mathbf{|}}{\mathop{C}}\,}}\,}}\,}}\,}}\,}}}\,}}}\,-COOH\]
done
clear
B)
\[\underset{N{{H}_{2}}}{\mathop{\underset{|}{\mathop{\underset{C{{H}_{2}}}{\mathop{\underset{|}{\mathop{\underset{C{{H}_{2}}}{\mathop{\underset{|}{\mathop{C{{H}_{2}}OH}}\,}}\,}}\,}}\,}}\,}}\,\]
done
clear
C)
\[H-\underset{OH}{\overset{N{{H}_{2}}}{\mathop{\underset{\mathbf{|}}{\overset{\mathbf{|}}{\mathop{\underset{C{{H}_{2}}}{\mathop{\underset{\mathbf{|}}{\mathop{C}}\,}}\,}}}\,}}}\,-COOH\]
done
clear
D)
\[H-\underset{N{{H}_{2}}}{\overset{N{{H}_{2}}}{\mathop{\underset{\mathbf{|}}{\overset{\mathbf{|}}{\mathop{\underset{C{{H}_{2}}}{\mathop{\underset{\mathbf{|}}{\mathop{\underset{C{{H}_{2}}}{\mathop{\underset{\mathbf{|}}{\mathop{\underset{C{{H}_{2}}}{\mathop{\underset{\mathbf{|}}{\mathop{\underset{C{{H}_{2}}}{\mathop{\underset{\mathbf{|}}{\mathop{C}}\,}}\,}}\,}}\,}}\,}}\,}}\,}}\,}}}\,}}}\,-COOH\]
done
clear
View Answer play_arrow
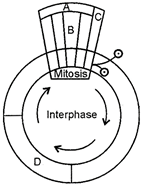
question_answer 134)
Given below is a schematic break-up of the phases/stages of cell cycle. Which one of the following is the correct indication of the stage/phase in the cell cycle?
A)
C-karyokinesis
done
clear
B)
S-synthetic phase
done
clear
C)
A-cytokinesis
done
clear
D)
B-metaphase
done
clear
View Answer play_arrow
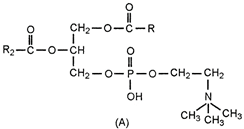
question_answer 135)
Which one of the following structural formula of two organic compounds is correctly identified along with its related function?
A)
B-uracil - a component of DNA
done
clear
B)
A-triglyceride - major source of energy
done
clear
C)
A-lecithin - a component of cell membrane
done
clear
D)
B-adenine - a nucleotide that makes up nucleic
done
clear
View Answer play_arrow
question_answer 136) The effect of cigarette smoking and radon in combination on lungs is
A)
fatal
done
clear
B)
synergistic
done
clear
C)
mutualistic
done
clear
D)
antagonistic
done
clear
View Answer play_arrow
question_answer 137) The thermostable enzymes, Taq and Pfu, isolated from thermophilic bacteria are
A)
RNA polymerases
done
clear
B)
DNA ligases
done
clear
C)
DNA polymerases
done
clear
D)
restriction endonucleases
done
clear
View Answer play_arrow
question_answer 138) Biolistic technique is used in
A)
gene transfer process
done
clear
B)
tissue culture process
done
clear
C)
hybridisation process
done
clear
D)
germplasm conservation process
done
clear
View Answer play_arrow
question_answer 139) The largest gene in man is
A)
insulin gene
done
clear
B)
tumour suppressor gene
done
clear
C)
beta globin gene of haemoglobin
done
clear
D)
dystrophin
done
clear
View Answer play_arrow
question_answer 140) Herbicide resistant gene in plant is
A)
Mt
done
clear
B)
Gt
done
clear
C)
Ct
done
clear
D)
Bt
done
clear
View Answer play_arrow
question_answer 141) Pellagra is caused by deficiency of vitamins
A)
\[{{B}_{5}}\]
done
clear
B)
\[{{B}_{2}}\]
done
clear
C)
\[{{B}_{6}}\]
done
clear
D)
\[{{B}_{1}}\]
done
clear
View Answer play_arrow
question_answer 142) Notochord originates from
A)
mesoderm
done
clear
B)
ectoderm
done
clear
C)
endoderm
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 143) Parthenogenesis is a term of
A)
budding
done
clear
B)
asexual reproduction
done
clear
C)
sexual reproduction
done
clear
D)
regeneration
done
clear
View Answer play_arrow
question_answer 144) Bartholin's gland is found in
A)
penis
done
clear
B)
stomach
done
clear
C)
liver
done
clear
D)
vagina
done
clear
View Answer play_arrow
question_answer 145) Which one of the following statements best characterize the testis?
A)
The seminiferous epithelium contains only proliferative cells
done
clear
B)
Functional compartmentalization of the seminiferous epithelium depends on light junctions
done
clear
C)
The interstitial tissue contains few capillaries
done
clear
D)
The seminiferous epithelium contains numerous capillaries
done
clear
View Answer play_arrow
question_answer 146) Which is substitution of mitochondria in E.coli?
A)
Golgi body
done
clear
B)
Mesosome
done
clear
C)
Ribosome
done
clear
D)
Glyoxysomes
done
clear
View Answer play_arrow
question_answer 147) Animal cell differ from plant cell in possess!)
A)
vacuoles
done
clear
B)
centrosomes
done
clear
C)
pastids
done
clear
D)
mitochondria
done
clear
View Answer play_arrow
question_answer 148) Which of the following organelles does not contain RNA?
A)
Plasmalemma
done
clear
B)
Ribosome
done
clear
C)
Chromosome
done
clear
D)
Nucleolus
done
clear
View Answer play_arrow
question_answer 149) Dutrochet has given the concept about cell in
A)
1834
done
clear
B)
1814
done
clear
C)
1822
done
clear
D)
1824
done
clear
View Answer play_arrow
question_answer 150) The scientific name of gharial is
A)
Naja bungarus
done
clear
B)
Gavialis gangeticus
done
clear
C)
Hemidactylus flavivridis
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 151) Ichthyology is study of
A)
aves
done
clear
B)
amphibians
done
clear
C)
reptiles
done
clear
D)
fishes
done
clear
View Answer play_arrow
question_answer 152) What will happen if ligaments are torn?
A)
Bone will become unfixed
done
clear
B)
Bone will become fixed
done
clear
C)
Bone less movable at joint and pain
done
clear
D)
Bone will move freely at joint and no pain
done
clear
View Answer play_arrow
question_answer 153) Achondroplasia is a disease related with the defect in the formation of
A)
membrane
done
clear
B)
mucosa
done
clear
C)
bone
done
clear
D)
cartilage
done
clear
View Answer play_arrow
question_answer 154) Yellow bone marrow is found specially in the medullary cavity
A)
long bones
done
clear
B)
spongy bones
done
clear
C)
short bones
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 155)
Match the items of column-I with column-II and choose the correct option from the codes given below. Column-I Column-II A. Neuron 1. Ossein B. Bone-matrix 2. Nissl?s bodies C. RBCs of man 3. Antibodies D. Lymphocytes 4. Non-nucleated
Codes:
A)
A-4 B-1 C-2 D-3
done
clear
B)
A-2 B-1 C-4 D-3
done
clear
C)
A-3 B-4 C-1 D-2
done
clear
D)
A-2 B-3 C-4 D-1
done
clear
View Answer play_arrow
question_answer 156) Space in the jaw bone unoccupied by teeth is called
A)
dentine
done
clear
B)
diastema
done
clear
C)
enamel
done
clear
D)
crown
done
clear
View Answer play_arrow
question_answer 157) Identify the correct set, which shows the name of the enzyme from where it is secreted and substrate upon which it acts.
A)
Ptyalin - Intestine - Maltose
done
clear
B)
Ptyalin - Pancreas - Lipid
done
clear
C)
Pepsin - Stomach wall - Caesin
done
clear
D)
Chymotrypsin - Salivary gland - Lactose
done
clear
View Answer play_arrow
question_answer 158) Endemic goitre is a state of
A)
normal thyroid function
done
clear
B)
moderate thyroid function
done
clear
C)
increased thyroid function
done
clear
D)
decreased thyroid function
done
clear
View Answer play_arrow
question_answer 159) Hormone responsible for the secretion of milk after parturition is
A)
ACTH
done
clear
B)
LH
done
clear
C)
ICSH
done
clear
D)
Prolactin
done
clear
View Answer play_arrow
question_answer 160) What is another name for the wind pipe?
A)
Trachea
done
clear
B)
Larynx
done
clear
C)
Oesophagus
done
clear
D)
Lungs
done
clear
View Answer play_arrow
question_answer 161) People have little to consider ideas and objectives because
A)
they consider these ideals meaningless
done
clear
B)
they do not want to burden themselves with such ideas
done
clear
C)
they have no inclination for such things
done
clear
D)
they are excessively engaged in their routine activities
done
clear
View Answer play_arrow
question_answer 162) The burden of life's problems' in the fourth sentence refers to
A)
the incessant and feverish activities
done
clear
B)
the burden of family responsibilities
done
clear
C)
the onerous duties of life
done
clear
D)
the sorrows and sufferings
done
clear
View Answer play_arrow
question_answer 163) The World Wars IInd are the price that man paid due to
A)
the absence of wisdom and sagacity
done
clear
B)
his not caring to consider the life's problems
done
clear
C)
his ignoring the ideals and objectives of life
done
clear
D)
his excessive involvement in feverish activities
done
clear
View Answer play_arrow
question_answer 164) According to the writer the adoption of wrong means even for the right end would
A)
not let us attain our goal
done
clear
B)
bring us dishonor
done
clear
C)
impede our progress
done
clear
D)
deflect us from the right path
done
clear
View Answer play_arrow
question_answer 165) The word 'vitiate' used in the second paragraph means
A)
negate
done
clear
B)
debase
done
clear
C)
tarnish
done
clear
D)
destroy
done
clear
View Answer play_arrow
question_answer 166) Directions: In the following questions, sentences are given with blanks to be filled in with an appropriate word. Four alternatives are suggested for each questions. Choose the correct alternative out of the four. The little girl .......... for the light switch in the dark.
A)
groped
done
clear
B)
grappled
done
clear
C)
gripped
done
clear
D)
groveled
done
clear
View Answer play_arrow
question_answer 167) Directions: In the following questions, sentences are given with blanks to be filled in with an appropriate word. Four alternatives are suggested for each questions. Choose the correct alternative out of the four. The summit meeting provided him the much .......... shot in the arm.
A)
required
done
clear
B)
desired
done
clear
C)
needed
done
clear
D)
urgent
done
clear
View Answer play_arrow
question_answer 168) Directions: In the following questions, sentences are given with blanks to be filled in with an appropriate word. Four alternatives are suggested for each questions. Choose the correct alternative out of the four. We must .......... the tickets for the movie in advance.
A)
draw
done
clear
B)
buy
done
clear
C)
remove
done
clear
D)
take
done
clear
View Answer play_arrow
question_answer 169) Directions: In the following questions, sentences are given with blanks to be filled in with an appropriate word. Four alternatives are suggested for each questions. Choose the correct alternative out of the four. The State Transport Corporation has.......... a loss of Rs. 5 crore this year.
A)
obtained
done
clear
B)
derived
done
clear
C)
incurred
done
clear
D)
formulated
done
clear
View Answer play_arrow
question_answer 170) Directions: In the following questions, sentences are given with blanks to be filled in with an appropriate word. Four alternatives are suggested for each questions. Choose the correct alternative out of the four. One .......... and you know who among them is the culprit.
A)
look
done
clear
B)
peep
done
clear
C)
sight
done
clear
D)
gaze
done
clear
View Answer play_arrow
question_answer 171) Directions: In the following questions, choose the word opposite in meaning to the given word. FILTHY
A)
Stainless
done
clear
B)
Shining
done
clear
C)
Sterilized
done
clear
D)
Clean
done
clear
View Answer play_arrow
question_answer 172) Directions: In the following questions, choose the word opposite in meaning to the given word. CROWDED
A)
Deserted
done
clear
B)
Lonely
done
clear
C)
Empty
done
clear
D)
Barren
done
clear
View Answer play_arrow
question_answer 173) Directions: In the following questions, choose the word opposite in meaning to the given word. VAGUE
A)
Known
done
clear
B)
Published
done
clear
C)
Popular
done
clear
D)
Definite
done
clear
View Answer play_arrow
question_answer 174) Directions: In the following questions, choose the word opposite in meaning to the given word. SUPERVISE
A)
Overlook
done
clear
B)
Misdirect
done
clear
C)
Neglect
done
clear
D)
Forget
done
clear
View Answer play_arrow
question_answer 175) Directions: In the following questions, choose the word opposite in meaning to the given word. MAGNANIMOUS
A)
Selfish
done
clear
B)
Naïve
done
clear
C)
Generous
done
clear
D)
Small
done
clear
View Answer play_arrow
question_answer 176) Ravi has got many friends because he has got much money.
A)
Enough money
done
clear
B)
A lot of money
done
clear
C)
Bags of money
done
clear
D)
No improvement
done
clear
View Answer play_arrow
question_answer 177) You must try making him to understand.
A)
Make him understand
done
clear
B)
To making him understand
done
clear
C)
To make him understand
done
clear
D)
No improvement
done
clear
View Answer play_arrow
question_answer 178) He has cooked that meal so often he can do it with his eyes closed.
A)
Mind blank
done
clear
B)
Eyes covered
done
clear
C)
Hands full
done
clear
D)
No improvement
done
clear
View Answer play_arrow
question_answer 179) Not a word they spoke to the unfortunate wife about it.
A)
They had spoken
done
clear
B)
Did they speak
done
clear
C)
They will speak
done
clear
D)
No improvement
done
clear
View Answer play_arrow
question_answer 180) There is sufficient fund to meet the requirement of the entire schools in our zone.
A)
Schools
done
clear
B)
All the schools
done
clear
C)
All of the schools
done
clear
D)
No improvement
done
clear
View Answer play_arrow
question_answer 181) Directions: In the following questions, out of the four alternatives, choose the one which best expresses the meaning of the given word. GAINSAY
A)
Advantage
done
clear
B)
Proposal
done
clear
C)
Contradict
done
clear
D)
Suggestion
done
clear
View Answer play_arrow
question_answer 182) Directions: In the following questions, out of the four alternatives, choose the one which best expresses the meaning of the given word. PROFOUND
A)
Profuse
done
clear
B)
Boundless
done
clear
C)
Deep
done
clear
D)
Fathomless
done
clear
View Answer play_arrow
question_answer 183) Directions: In the following questions, out of the four alternatives, choose the one which best expresses the meaning of the given word. FLAK
A)
Adventure
done
clear
B)
Advice
done
clear
C)
Criticism
done
clear
D)
Praise
done
clear
View Answer play_arrow
question_answer 184) Directions: In the following questions, out of the four alternatives, choose the one which best expresses the meaning of the given word. HOODLUM
A)
Pioneer
done
clear
B)
Criminal
done
clear
C)
Devotee
done
clear
D)
Scholar
done
clear
View Answer play_arrow
question_answer 185) Directions: In the following questions, out of the four alternatives, choose the one which best expresses the meaning of the given word. SPASMODIC
A)
Continuous
done
clear
B)
Gradual
done
clear
C)
Intermittent
done
clear
D)
Spontaneous
done
clear
View Answer play_arrow
question_answer 186)
(1) In reply to a question (P) that securing extradition (Q) operating from the UK soil remained (R) of anti-India elements (S) the spokesman said
(6) New Delhi's first priority.
A)
PRQS
done
clear
B)
QSPR
done
clear
C)
RQSP
done
clear
D)
SPRQ
done
clear
View Answer play_arrow
question_answer 187)
(1) The first component is (P) and vocational training (Q) so as to enable them (R) the provision of further technical (S) to both rural and urban youth
(6) to secure employment in industry and the services sector.
A)
PRSQ
done
clear
B)
RPSQ
done
clear
C)
RSQP
done
clear
D)
SRPQ
done
clear
View Answer play_arrow
question_answer 188)
(1) The move to revert to a six-day week (P) among the employees (Q) while the leaders represented to the Chef Minister (R) that they be taken into confidence (S) led to an animated decision
(6) before any decision was taken.
A)
QPSR
done
clear
B)
RSPQ
done
clear
C)
SPQR
done
clear
D)
SQPR
done
clear
View Answer play_arrow
question_answer 189)
(1) It was obvious (P) made by him (Q) submitted at the meeting (R) from the comments (S) on the draft proposals
(6) that he was not satisfied with them
A)
PSRQ
done
clear
B)
QRSP
done
clear
C)
RPSQ
done
clear
D)
SQRP
done
clear
View Answer play_arrow
question_answer 190)
(1) The Minister of state for power (P) laying emphasis (Q) in conservation of electricity in industries (R) has written to his counterparts in State Government (S) on bringing about improvement
(6) by introduction of energy efficient equipment.
A)
QPSR
done
clear
B)
RPSQ
done
clear
C)
SPQR
done
clear
D)
SQPR
done
clear
View Answer play_arrow
question_answer 191) We have to keep our fingers crossed till the final result is declared.
A)
keep praying
done
clear
B)
feel suspicious
done
clear
C)
wait expectantly
done
clear
D)
feel scared
done
clear
View Answer play_arrow
question_answer 192) The members of the group were at odds over the selection procedure.
A)
acting foolishly
done
clear
B)
in dispute
done
clear
C)
unanimous
done
clear
D)
behaving childishly
done
clear
View Answer play_arrow
question_answer 193) The popularity of the yesteryears' superstar is on the wane.
A)
growing more
done
clear
B)
at its peak
done
clear
C)
growing less
done
clear
D)
at rock-bottom
done
clear
View Answer play_arrow
question_answer 194) His father advised him to be fair and square in his dealings lest he should fall into trouble.
A)
considerate
done
clear
B)
upright
done
clear
C)
careful
done
clear
D)
polite
done
clear
View Answer play_arrow
question_answer 195) There is no love lost between the two neighbors.
A)
close friendship
done
clear
B)
intense dislike
done
clear
C)
a love-hate relationship
done
clear
D)
cool indifference
done
clear
View Answer play_arrow
question_answer 196) Directions: In the following questions, out of the four alternatives, choose the one which can be substituted for the given words/sentence. Word for word reproduction.
A)
Copying
done
clear
B)
Mugging
done
clear
C)
Verbatim
done
clear
D)
Photostat
done
clear
View Answer play_arrow
question_answer 197) Directions: In the following questions, out of the four alternatives, choose the one which can be substituted for the given words/sentence. A person who collects coins.
A)
Philatelist
done
clear
B)
Numismatist
done
clear
C)
Narcissist
done
clear
D)
Fatalist
done
clear
View Answer play_arrow
question_answer 198) Directions: In the following questions, out of the four alternatives, choose the one which can be substituted for the given words/sentence. That which is perceptible by touch
A)
Tangible
done
clear
B)
Tenacious
done
clear
C)
Contagious
done
clear
D)
Contingent
done
clear
View Answer play_arrow
question_answer 199) Directions: In the following questions, out of the four alternatives, choose the one which can be substituted for the given words/sentence. One who possesses many talents.
A)
Versatile
done
clear
B)
Gifted
done
clear
C)
Exceptional
done
clear
D)
Nubile
done
clear
View Answer play_arrow
question_answer 200) Directions: In the following questions, out of the four alternatives, choose the one which can be substituted for the given words/sentence. A person who studies the formation of the Earth.
A)
Meteorologist
done
clear
B)
Anthropologist
done
clear
C)
Geologist
done
clear
D)
Seismologist
done
clear
View Answer play_arrow