question_answer 1) A car is moving at a speed of 72 km/hr. The diameter of its wheels is 0.5 m. If the wheels are stopped in 20 rotations applying brakes. Then angular retardation produced by the brake is:
A)
\[-45.5\text{ }rad/{{s}^{2}}\]
done
clear
B)
\[-33.5\text{ }rad/{{s}^{2}}\]
done
clear
C)
\[-29.5\text{ }rad/{{s}^{2}}\]
done
clear
D)
\[-25.5\text{ }rad/{{s}^{2}}\]
done
clear
View Answer play_arrow
question_answer 2) A bullet is fired from the gun with a speed of 1000 m/s m order to hit a target. s = 100 m away. At what height above the target should by gun be aimed? (The resistance of air is negligible and\[g=10\text{ }m/{{s}^{2}}\])
A)
23 cm
done
clear
B)
15 cm
done
clear
C)
9 cm
done
clear
D)
5 cm
done
clear
View Answer play_arrow
question_answer 3) If the cofficient of friction of a plane inclined at\[45{}^\circ \]is 0.5. Then acceleration of a body sliding freely on it will be:
A)
\[\frac{9.8}{1\sqrt{2}}m/{{s}^{2}}\]
done
clear
B)
\[\frac{9.8}{\sqrt{2}}m/{{s}^{2}}\]
done
clear
C)
\[9.8\text{ }m/{{s}^{2}}\]
done
clear
D)
\[4.8\text{ }m/{{s}^{2}}\]
done
clear
View Answer play_arrow
question_answer 4) Assuming earth to be sphere of uniform density what is the value of acceleration due to gravity at a point 100 km below the earth surface (Given\[R=6380\times {{10}^{3}}m\])
A)
\[3.10\text{ }m/s\]
done
clear
B)
\[5.06\text{ }m/{{s}^{2}}\]
done
clear
C)
\[7.64\text{ }m/{{s}^{2}}\]
done
clear
D)
\[9.66\text{ }m/{{s}^{2}}\]
done
clear
View Answer play_arrow
question_answer 5) Plancks constant has same dimension as:
A)
angular momentum
done
clear
B)
linear momentum
done
clear
C)
force
done
clear
D)
energy
done
clear
View Answer play_arrow
question_answer 6) The mass of ship is\[2\times {{10}^{7}}kg\]. On applying a fore of\[25\times {{10}^{5}}N,\]it is displaced through 25 m. After the displacement. The speed aquired by the ship will be:
A)
12.5 m/s
done
clear
B)
5m/s
done
clear
C)
3.7 m/s
done
clear
D)
2.5 m/s
done
clear
View Answer play_arrow
question_answer 7) A 500 kg car takes around turn of radius 50 m with a speed of 36 km/hr. The centripetel force acting on the car will be:
A)
1200 N
done
clear
B)
1000 N
done
clear
C)
750 N
done
clear
D)
250 N
done
clear
View Answer play_arrow
question_answer 8) An apple gives 21 kJ energy to a boy. How much height he can climb by using this energy. If his efficiency is 28%? (mass of boy 40 kg)
A)
22.5 m
done
clear
B)
15 m
done
clear
C)
10 m
done
clear
D)
5 m
done
clear
View Answer play_arrow
question_answer 9) Mass of the moon is\[\frac{1}{81}\]times that of earth and its radius is\[\frac{1}{4}\]. the radius of earth and the escape velocity at die surface of the earth is 11.2 km/sec:
A)
2.5km/s
done
clear
B)
5km/s
done
clear
C)
1.25km/s
done
clear
D)
0.25km/s
done
clear
View Answer play_arrow
question_answer 10) The moment of inertia of a body about a given axis 1.2 kg m2. To produce rotational K.E. of 1500 J an angular acceleration of\[25\text{ }rad/se{{c}^{2}}\]must be applied about that axis for:
A)
10 sec
done
clear
B)
8 sec
done
clear
C)
2 sec
done
clear
D)
4 sec
done
clear
View Answer play_arrow
question_answer 11) A moving body of mass m and velocity 3 km/hr. Collides with a body at rest and of mass 2m and then sticks to it. Now the combined mass starts to move, then the combined velocity will be:
A)
4 km/hr
done
clear
B)
3 km/hr
done
clear
C)
2 km/hr
done
clear
D)
1 km/hr
done
clear
View Answer play_arrow
question_answer 12) A 120 m long train is moving towards west with a speed of 10 m/s. A bird flying towards east with a speed of 3 m/s crosses the train. The time taken by the bird to cross the train will be:
A)
16 sec
done
clear
B)
12 sec
done
clear
C)
10 sec
done
clear
D)
8 sec
done
clear
View Answer play_arrow
question_answer 13) The velocity of all radio waves in free space is \[3\times {{10}^{8}}\]m/s. The frequency of a radio wave of wavelength 150 m is:
A)
20 kHz
done
clear
B)
2 kHz
done
clear
C)
2 MHz
done
clear
D)
1 MHz
done
clear
View Answer play_arrow
question_answer 14) A boy of 50 kg is standing in a left moving down with an acceleration\[9.8\text{ }m/{{s}^{2}}\]. The apparent weight of the boy is:
A)
\[\frac{50}{9.8}N\]
done
clear
B)
\[50\times 9.8N\]
done
clear
C)
50 N
done
clear
D)
zero
done
clear
View Answer play_arrow
question_answer 15) A particle executing SHM has amplitude 0.01 and frequency 60 Hz. The maximum acceleration of the particle is:
A)
\[60\text{ }{{\pi }^{2}}\text{ }m/{{s}^{2}}\]
done
clear
B)
\[88\,{{\pi }^{2}}m/{{s}^{2}}\]
done
clear
C)
\[140\,{{\pi }^{2}}m/{{s}^{2}}\]
done
clear
D)
\[144\,{{\pi }^{2}}m/{{s}^{2}}\]
done
clear
View Answer play_arrow
question_answer 16) If the equation of motion of standing waves is \[y=0.3\text{ }sin(314\text{ }t-1.57\text{ }x),\]then the velocity of standing wave will be:
A)
400 m/s
done
clear
B)
300 m/s
done
clear
C)
200in/s
done
clear
D)
100 m/s
done
clear
View Answer play_arrow
question_answer 17) In a thermodynamics process pressure of a fixed mass of a gas is changed in such a manner that the gas molecule give out 30 joule of heat and 10 joule of work is done on the gas. If the initial internal energy of the gas was 40 joule, then the final internal energy will be:
A)
\[-20J\]
done
clear
B)
\[20J\]
done
clear
C)
\[80J\]
done
clear
D)
\[3J\]
done
clear
View Answer play_arrow
question_answer 18) A black body is heated from\[27{}^\circ C\]to\[927{}^\circ C\] the ratio of radiations emitted will be:
A)
1:256
done
clear
B)
1:64
done
clear
C)
1:16
done
clear
D)
1: 4
done
clear
View Answer play_arrow
question_answer 19) The efficiency of a Cannot engine operating with reservoir temperature of \[100{}^\circ C\] and\[-23{}^\circ C\]will be:
A)
\[\frac{100-23}{373}\]
done
clear
B)
\[\frac{100+23}{373}\]
done
clear
C)
\[\frac{100+23}{100}\]
done
clear
D)
\[\frac{100-23}{100}\]
done
clear
View Answer play_arrow
question_answer 20) A body cools from\[60{}^\circ C\]to\[50{}^\circ C\]in 10 minutes. If the room temperature is\[25{}^\circ C\] and assuming newtons law of cooling to hold good, the temperature of the body at the end of the next 10 minutes will be:
A)
\[45{}^\circ C\]
done
clear
B)
\[42.85{}^\circ C\]
done
clear
C)
\[40{}^\circ C\]
done
clear
D)
\[38.5{}^\circ C\]
done
clear
View Answer play_arrow
question_answer 21) A particle having charge 100 times that of an electron is revolving in a circular path of radius 0.8 m with one rotation per second. Magnetic field produced at center of particle is:
A)
\[{{10}^{-17}}{{\mu }_{0}}\]
done
clear
B)
\[{{10}^{-11}}{{\mu }_{0}}\]
done
clear
C)
\[{{10}^{-7}}{{\mu }_{0}}\]
done
clear
D)
\[{{10}^{-3}}{{\mu }_{0}}\]
done
clear
View Answer play_arrow
question_answer 22) A galvanometer can be changed into ammeter by connecting:
A)
high resistance in parallel
done
clear
B)
high resistance m series
done
clear
C)
low resistance in parallel
done
clear
D)
low resistance in series
done
clear
View Answer play_arrow
question_answer 23) A prism has a refracting angle\[60{}^\circ \]. A ray of given monochromatic light suffers minimum deviation of \[38{}^\circ \] in passing through prism refractive index of the material of prism is:
A)
1.5094
done
clear
B)
1.3056
done
clear
C)
0.7849
done
clear
D)
2.425
done
clear
View Answer play_arrow
question_answer 24) A metal corn is at the bottom of a beaker filled with a liquid of refractive index 4/3 to height of 6 cm. To an observer looking from above the surface of the liquid, coin will appear at a depth of:
A)
7.5 cm
done
clear
B)
6.75 cm
done
clear
C)
4.5 cm
done
clear
D)
1.5 cm
done
clear
View Answer play_arrow
question_answer 25) An astronomical telescope has two lenses of focal powers 0.5 D and 20 D. Then its magnifying power will be:
A)
8
done
clear
B)
20
done
clear
C)
30
done
clear
D)
40
done
clear
View Answer play_arrow
question_answer 26) The peak value of AC voltage on a 220V mains is:
A)
\[240\sqrt{2}V\]
done
clear
B)
\[230\sqrt{2}V\]
done
clear
C)
\[220\sqrt{2}V\]
done
clear
D)
\[200\sqrt{2}V\]
done
clear
View Answer play_arrow
question_answer 27) A resistance of a galvanometer\[G=6\,\Omega \] maximum current of 2 amp is measured by it. Then required resistance to convert it into an ammeter reading up to 6 A, will be:
A)
\[5\,\Omega \]
done
clear
B)
\[4\,\Omega \]
done
clear
C)
\[3\,\Omega \]
done
clear
D)
\[2\,\Omega \]
done
clear
View Answer play_arrow
question_answer 28) Two copper wires are of same length one of the twice as thick as the other. Then the resistance of the two wires are in the ratio of:
A)
1:16
done
clear
B)
1:8
done
clear
C)
1:4
done
clear
D)
1:2
done
clear
View Answer play_arrow
question_answer 29) A hot electric iron has a resistance of 80 n and is used on a 200 V source. The electrical energy spent, if it is used for hr, will be:
A)
8000Wt
done
clear
B)
2000Wt
done
clear
C)
1000Wt
done
clear
D)
800Wt
done
clear
View Answer play_arrow
question_answer 30)
In the given network capacitance\[{{C}_{2}}=10\mu F,{{C}_{1}}=5\mu F\]and\[{{C}_{3}}=4\mu F\]. The resultant capacitance between P and Q will be:
A)
\[4.7\mu F\]
done
clear
B)
\[1.2\mu F\]
done
clear
C)
\[3.2\mu F\]
done
clear
D)
\[2.2\mu F\]
done
clear
View Answer play_arrow
question_answer 31) The primary of a stepdown. Transformer used for ringing door bell has 2000 turns of fine wire and the secondary has 100 turns. The transformer when connected to 110 V AC source will deliver at its secondary at a potential difference of:
A)
5.5V
done
clear
B)
11V
done
clear
C)
55V
done
clear
D)
220V
done
clear
View Answer play_arrow
question_answer 32) From a point charge, there is a fixed point A. At A, there is an electric field of 500 volt/meter and potential difference of 3000 V. Distance between the point charge and A is:
A)
24m
done
clear
B)
16m
done
clear
C)
12m
done
clear
D)
6m
done
clear
View Answer play_arrow
question_answer 33)
In the inductive circuit given in the figure, the currents rises after the switch is closed. At instant when the current is
A)
zero
done
clear
B)
240 V
done
clear
C)
180V
done
clear
D)
60V
done
clear
View Answer play_arrow
question_answer 34)
From resistances of 100 ohm each are connected in the form of a square. The effective resistance along the diagonal points P R is:
A)
\[100\,\Omega \]
done
clear
B)
\[180\,\Omega \]
done
clear
C)
\[220\,\Omega \]
done
clear
D)
\[440\,\Omega \]
done
clear
View Answer play_arrow
question_answer 35) 700 pF capacitor is charged by 50 V battery. Electrostatic energy is stored by it will be:
A)
\[17.0\times {{10}^{-8}}J\]
done
clear
B)
\[13.0\times {{10}^{-9}}J\]
done
clear
C)
\[8.7\times {{10}^{-7}}J\]
done
clear
D)
\[6.7\times {{10}^{-7}}J\]
done
clear
View Answer play_arrow
question_answer 36) An aeroplane having a wing space of 35 m flies due north with the speed of 90 m/s given \[B\text{ }4\times {{10}^{-5}}\]tesla. The potential difference between the tips of the wings will be:
A)
0.013 V
done
clear
B)
1.26 V
done
clear
C)
12.6V
done
clear
D)
0.126V
done
clear
View Answer play_arrow
question_answer 37) Two coils have mutual inductance 0.005 H. The current changes in the first coil according to equation\[I={{I}_{0}}\sin \omega t\]. where\[{{I}_{0}}=10\]amp and \[\omega =100\text{ }\pi \] rad/sec. The maximum value of emf in the second coil is:
A)
\[12\,\pi \]
done
clear
B)
\[8\,\pi \]
done
clear
C)
\[5\,\pi \]
done
clear
D)
\[2\,\pi \]
done
clear
View Answer play_arrow
question_answer 38) A radioactive element has half life of 3.6 days. In what time will it be left 1/32nd undecayed?
A)
4 days
done
clear
B)
12 days
done
clear
C)
18 days
done
clear
D)
24 days
done
clear
View Answer play_arrow
question_answer 39) In a p type semiconductor gerpanium is doped with:
A)
gallium
done
clear
B)
aluminium
done
clear
C)
boron
done
clear
D)
all of above
done
clear
View Answer play_arrow
question_answer 40) The wavelength associated with an electron accelerated through a potential difference of 100 V is of the order of:
A)
\[1.2\overset{o}{\mathop{\text{A}}}\,\]
done
clear
B)
\[10.5\overset{o}{\mathop{\text{A}}}\,\]
done
clear
C)
\[100\overset{o}{\mathop{\text{A}}}\,\]
done
clear
D)
\[1000\overset{o}{\mathop{\text{A}}}\,\]
done
clear
View Answer play_arrow
question_answer 41) A stone is tied to one end of a spring 50 cm long is whirled in a horizontal circle with a constant speed. If the stone makes 10 revolutions in 20 second. The magnitude of acceleration of stone will be:
A)
\[990\,cm/{{s}^{2}}\]
done
clear
B)
\[860\,cm/{{s}^{2}}\]
done
clear
C)
\[720\,cm/{{s}^{2}}\]
done
clear
D)
\[493\,cm/{{s}^{2}}\]
done
clear
View Answer play_arrow
question_answer 42) A straight conductor of length 4m moves at a speed of 10 m/s. When the conductor makes an angle of\[30{}^\circ \]with the direction of magnetic field of induction of 0.1 wb. per\[{{m}^{2}}\]then induced emf. is:
A)
8V
done
clear
B)
4V
done
clear
C)
1V
done
clear
D)
2V
done
clear
View Answer play_arrow
question_answer 43) The work function for aluminium surface is 4.2 eV. The cutoff wavelength for photoelectric effect is:
A)
\[1000\overset{o}{\mathop{\text{A}}}\,\]
done
clear
B)
\[2000\overset{o}{\mathop{\text{A}}}\,\]
done
clear
C)
\[2955\overset{o}{\mathop{\text{A}}}\,\]
done
clear
D)
\[4200\overset{o}{\mathop{\text{A}}}\,\]
done
clear
View Answer play_arrow
question_answer 44) The ionisation potential of hydrogen atom is 13.6 volt. In the lowest energy level, this atom is ionised by absorbing a photon of\[800\overset{o}{\mathop{\text{A}}}\,\]. The kinetic energy of the released electron will be:
A)
15.51 eV
done
clear
B)
2.91 eV
done
clear
C)
13.6 eV
done
clear
D)
1.91 Ev
done
clear
View Answer play_arrow
question_answer 45) Penetrating power is minimum for:
A)
X-rays
done
clear
B)
\[\gamma -\]rays
done
clear
C)
\[\beta -\]rays
done
clear
D)
\[\alpha -\]rays
done
clear
View Answer play_arrow
question_answer 46) The functions of moderators in nuclear reactor is to:
A)
decrease the speed of neutrons
done
clear
B)
increase the speed of neutrons
done
clear
C)
decrease the speed of electrons
done
clear
D)
increase the speed of electrons
done
clear
View Answer play_arrow
question_answer 47) A chain reaction in fission of uranium is possible, because:
A)
Two intermediate sized nuclear fragments are formed
done
clear
B)
Three neutrons are given out in each fission
done
clear
C)
fragments in fission are radioactive
done
clear
D)
large amount of energy is released
done
clear
View Answer play_arrow
question_answer 48) A nuclei X with mass number A and charge number Z, disintegrates into one\[\alpha -\]particle and one\[\beta -\]particle. The resulting nuclide R has atomic mass and atomic number, equal to:
A)
(A - Z) and (Z - 1)
done
clear
B)
(A - Z) and (Z - 2)
done
clear
C)
(A -4) and (A -2)
done
clear
D)
(A-4) and (Z - 1)
done
clear
View Answer play_arrow
question_answer 49) Two inductors each of inductance L are joined in parallel. Their equivalent inductance will be:
A)
zero
done
clear
B)
\[\frac{L}{2}\]
done
clear
C)
L
done
clear
D)
\[2L\]
done
clear
View Answer play_arrow
question_answer 50) What will be the amount of energy absorbed when an electron jumps from first orbit to second orbits? (If the value of energy m nth orbit of N-atom is expressed as\[\left. E=-\frac{13.6}{{{n}^{2}}}eV \right)\]
A)
\[-3.4eV\]
done
clear
B)
\[-6.6eV\]
done
clear
C)
\[-8.1eV\]
done
clear
D)
\[-10.2eV\]
done
clear
View Answer play_arrow
question_answer 51) The number of neutrons in the element\[_{4}B{{e}^{9}}\]is:
A)
3
done
clear
B)
5
done
clear
C)
7
done
clear
D)
9
done
clear
View Answer play_arrow
question_answer 52) The first ionisation potential of\[Na,Mg,Al\]and\[Si\]are in the order:
A)
\[Na<Mg<Al<Si\]
done
clear
B)
\[Na<Al<Mg<Si\]
done
clear
C)
\[Na>Mg>Al>Si\]
done
clear
D)
\[Na<Si<Al<Mg\]
done
clear
View Answer play_arrow
question_answer 53) Which of the following is not a colligative property?
A)
Depression in freezing point
done
clear
B)
Osmotic pressure
done
clear
C)
Elevation of boiling point
done
clear
D)
Increase in freezing point
done
clear
View Answer play_arrow
question_answer 54) The main product obtained from phenol with \[PC{{l}_{5}}\] is:
A)
BHC
done
clear
B)
hexachlorobenzene
done
clear
C)
chlorobenzene
done
clear
D)
triphenyl phosphate
done
clear
View Answer play_arrow
question_answer 55) 50 ml. of\[{{H}_{2}}S{{O}_{4}}\]require 10 gm\[CaC{{O}_{3}}\]for complete decomposition. The normality of acid is:
A)
2
done
clear
B)
0.30
done
clear
C)
4
done
clear
D)
0.20
done
clear
View Answer play_arrow
question_answer 56) The isomerism represented by ethyl acetoacetate is:
A)
keto-enol isomerism
done
clear
B)
geometrical isomerism
done
clear
C)
enantiomerism
done
clear
D)
diasterioisomerism
done
clear
View Answer play_arrow
question_answer 57) The compound that does not produce blue colour in Lassaignes test is:
A)
urea
done
clear
B)
hydrazine
done
clear
C)
aniline
done
clear
D)
glycine
done
clear
View Answer play_arrow
question_answer 58) No evolution of oxygen takes place by the reaction of ozone with:
A)
\[PbS\]
done
clear
B)
\[{{H}_{2}}{{O}_{2}}\]
done
clear
C)
\[S{{O}_{2}}\]
done
clear
D)
\[KI\]
done
clear
View Answer play_arrow
question_answer 59) Conversion of ethyl alcohol into acetaldehyde is an example of:
A)
hydrolysis
done
clear
B)
oxidation
done
clear
C)
reduction
done
clear
D)
molecular rearrangement
done
clear
View Answer play_arrow
question_answer 60) Transition elements does not show:
A)
paramagnetism
done
clear
B)
colour
done
clear
C)
fixed valency
done
clear
D)
all the above
done
clear
View Answer play_arrow
question_answer 61) The compound known as oil of wintergreen is:
A)
phenyl acetate
done
clear
B)
phenyl salicylate
done
clear
C)
methyl salicylate
done
clear
D)
methyl acetate
done
clear
View Answer play_arrow
question_answer 62) Which is not correct?
A)
\[n=2,\text{ }l=0,\text{ }m=-1\]
done
clear
B)
\[n=2,\text{ }l=1,\text{ }m=0\]
done
clear
C)
\[n=3,\text{ }l=1,\text{ }m=-1\]
done
clear
D)
\[n=3,\text{ }l=0,\text{ }m=0\]
done
clear
View Answer play_arrow
question_answer 63) Which of the following-is an intensive property?
A)
Density
done
clear
B)
Volume
done
clear
C)
Mass
done
clear
D)
Energy
done
clear
View Answer play_arrow
question_answer 64) The pH of the solution obtained by mixing 40 ml of\[0.10\text{ }M\text{ }HCl\]with 10 ml of 0.45 of\[NaOH\]is:
A)
4
done
clear
B)
8
done
clear
C)
12
done
clear
D)
14
done
clear
View Answer play_arrow
question_answer 65) The pairs of bases in DNA are held together by:
A)
ionic bonds
done
clear
B)
hydrogen bond
done
clear
C)
phosphate group
done
clear
D)
deoxyribose group
done
clear
View Answer play_arrow
question_answer 66) \[2Ag+2{{H}_{2}}S{{O}_{4}}\to A{{g}_{2}}S{{O}_{4}}+2{{H}_{2}}O+S{{O}_{2}}\] In the above reaction\[{{H}_{2}}S{{O}_{4}}\]acts as a:
A)
catalyst
done
clear
B)
oxidising agent
done
clear
C)
reducing agent
done
clear
D)
acid as well as oxidant
done
clear
View Answer play_arrow
question_answer 67) Lyophilic colloids are stable due to:
A)
large size of the particle
done
clear
B)
small size of the particle
done
clear
C)
charge on particle
done
clear
D)
dispersion layer on the particle
done
clear
View Answer play_arrow
question_answer 68) Which is not present in chlorophyll?
A)
Carbon
done
clear
B)
Calcium
done
clear
C)
Magnesium
done
clear
D)
Hydrogen
done
clear
View Answer play_arrow
question_answer 69) White lead is:
A)
\[PbS{{O}_{4}}-PbO\]
done
clear
B)
\[PbC{{O}_{3}}-PbO\]
done
clear
C)
\[PbC{{O}_{3}}\]
done
clear
D)
\[Pb{{(OH)}_{2}}-2PbC{{O}_{3}}\]
done
clear
View Answer play_arrow
question_answer 70) By passing air over red hot coke the gas obtained is:
A)
coal gas
done
clear
B)
water gas
done
clear
C)
oil gas
done
clear
D)
producer gas
done
clear
View Answer play_arrow
question_answer 71) The basic unit not present m DNA is:
A)
uracil
done
clear
B)
adenine
done
clear
C)
cytosine
done
clear
D)
guanine
done
clear
View Answer play_arrow
question_answer 72) \[{{P}_{4}}{{H}_{10}}\]on treatment with water produce:
A)
hypophosphoric acid
done
clear
B)
hypophosphorous acid
done
clear
C)
ortho phosphorous acid
done
clear
D)
ortho phosphoric acid
done
clear
View Answer play_arrow
question_answer 73) The resistance of 1 N solution of acetic acid is 250 ohm, when measured in a cell of cell constant\[1.15\text{ }c{{m}^{-1}}\]. The equivalent conductivity in\[oh{{m}^{-1}}\]cm\[equi{{v}^{-1}}\]1N acetic acid is:
A)
3.8
done
clear
B)
4.6
done
clear
C)
9.2
done
clear
D)
12.6
done
clear
View Answer play_arrow
question_answer 74) \[{{N}_{2(g)}}+3{{H}_{2(g)}}\xrightarrow[{}]{{}}2N{{H}_{3(g)}}\]. For the above reaction the correct statement is:
A)
\[\Delta H<\Delta E\]
done
clear
B)
\[\Delta H=\Delta E\]
done
clear
C)
\[\Delta H>\Delta E\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 75) The densities of two gases are in the ratio of\[1:16\]. The ratio of their rate of diffusion is:
A)
\[16:1\]
done
clear
B)
\[1:4\]
done
clear
C)
\[4:1\]
done
clear
D)
\[1:16\]
done
clear
View Answer play_arrow
question_answer 76) The molecular formula of cryollite is:
A)
\[N{{a}_{2}}F.Al{{F}_{6}}\]
done
clear
B)
\[N{{a}_{3}}Al{{F}_{6}}\]
done
clear
C)
\[N{{a}_{3}}Al{{F}_{5}}\]
done
clear
D)
\[N{{a}_{2}}Al{{F}_{3}}\]
done
clear
View Answer play_arrow
question_answer 77) 60 gm of a compound on analysis produce 24 gm carbon, 4 gm hydrogen and 32 gm oxygen. The empirical formula of the compound is:
A)
\[C{{H}_{2}}{{O}_{2}}\]
done
clear
B)
\[C{{H}_{2}}O\]
done
clear
C)
\[{{H}_{2}}\]
done
clear
D)
\[C{{H}_{4}}\]
done
clear
View Answer play_arrow
question_answer 78) Both ionic and covalent bond is present in:
A)
\[KCN\]
done
clear
B)
\[KCl\]
done
clear
C)
\[{{H}_{2}}\]
done
clear
D)
\[C{{H}_{4}}\]
done
clear
View Answer play_arrow
question_answer 79) On dissolving\[N{{H}_{4}}Cl\]to water the solution becomes cold. This is due to:
A)
exothermic
done
clear
B)
endothermic
done
clear
C)
supercooling
done
clear
D)
none
done
clear
View Answer play_arrow
question_answer 80) Hydrolysis of sucrose is:
A)
inversion
done
clear
B)
hydradon
done
clear
C)
sappnification
done
clear
D)
inhibition
done
clear
View Answer play_arrow
question_answer 81) \[{{C}_{2}}{{H}_{5}}N{{H}_{2}}\]and\[{{C}_{6}}{{H}_{5}}N{{H}_{2}}\]can be best distinguished by:
A)
\[NaOH\]
done
clear
B)
\[\beta -\]naphthol
done
clear
C)
\[CHC{{l}_{3}}/KOH\]
done
clear
D)
Hinsbergs reagent
done
clear
View Answer play_arrow
question_answer 82) \[{{C}_{3}}{{H}_{9}}N\]cannot represent:
A)
\[1{}^\circ \] Famine
done
clear
B)
\[2{}^\circ \]amine
done
clear
C)
\[3{}^\circ \]amine
done
clear
D)
quaternary ammonium salt
done
clear
View Answer play_arrow
question_answer 83) The function of moderator in a nuclear reactor is:
A)
to produce more neutrons
done
clear
B)
to stop nuclear reaction
done
clear
C)
to increase speed of neutron
done
clear
D)
to slow down speed of neutron
done
clear
View Answer play_arrow
question_answer 84) The weight of 112 ml of oxygen at NTP is:
A)
0.64gm
done
clear
B)
0.96gm
done
clear
C)
0.32gm
done
clear
D)
0.16gm
done
clear
View Answer play_arrow
question_answer 85) The pH of pure water at\[80{}^\circ C\]will be:
A)
>7
done
clear
B)
<7
done
clear
C)
=7
done
clear
D)
none
done
clear
View Answer play_arrow
question_answer 86) The most acidic oxide is:
A)
\[{{P}_{2}}{{O}_{5}})\]
done
clear
B)
\[{{N}_{2}}{{O}_{5}}\]
done
clear
C)
\[S{{b}_{2}}{{O}_{5}}\]
done
clear
D)
\[A{{S}_{2}}{{O}_{5}}\]
done
clear
View Answer play_arrow
question_answer 87) At 400 K sodium phenoxide reacts with\[C{{O}_{2}}\]to produce:
A)
catechol
done
clear
B)
sodium salicylate
done
clear
C)
benzoic acid
done
clear
D)
salicylaldehyde
done
clear
View Answer play_arrow
question_answer 88) The concentration units independent of temperature is:
A)
molarity
done
clear
B)
molality
done
clear
C)
normality
done
clear
D)
formality
done
clear
View Answer play_arrow
question_answer 89) Sold is an:
A)
antipyretic
done
clear
B)
analgesics
done
clear
C)
antiseptic
done
clear
D)
antibiotic
done
clear
View Answer play_arrow
question_answer 90) Which is not a Lewis acid?
A)
\[FeC{{l}_{3}}\]
done
clear
B)
\[AlC{{l}_{3}}\]
done
clear
C)
\[SnC{{l}_{4}}\]
done
clear
D)
\[AlC{{l}_{3}}.6{{H}_{2}}O\]
done
clear
View Answer play_arrow
question_answer 91) The reaction in which primary amines is not formed:
A)
\[C{{H}_{3}}NC\xrightarrow[{}]{LiAl{{H}_{4}}}\]
done
clear
B)
\[C{{H}_{3}}CON{{H}_{2}}\xrightarrow[{}]{LiAl{{H}_{4}}}\]
done
clear
C)
\[C{{H}_{3}}CN\xrightarrow[{}]{LiAl{{H}_{4}}}\]
done
clear
D)
\[C{{H}_{3}}CON{{H}_{2}}\xrightarrow[{}]{KOH/B{{r}_{2}}}\]
done
clear
View Answer play_arrow
question_answer 92) The reagent used to estimate\[{{I}_{2}}\]volumetrically is:
A)
\[KMn{{O}_{4}}\]
done
clear
B)
hypo
done
clear
C)
\[{{K}_{2}}C{{r}_{2}}{{O}_{7}}\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 93) Aluminium appears like gold when mixed with:
A)
90% Cu
done
clear
B)
75% Ni
done
clear
C)
80% Sn
done
clear
D)
80% CO
done
clear
View Answer play_arrow
question_answer 94) The abundantly found noble gas in atomosphere is:
A)
\[Kr\]
done
clear
B)
\[Xe\]
done
clear
C)
\[He\]
done
clear
D)
\[Ar\]
done
clear
View Answer play_arrow
question_answer 95) Distribution law was given by:
A)
Ostwald
done
clear
B)
Nernst
done
clear
C)
Henery
done
clear
D)
Vant Hoff
done
clear
View Answer play_arrow
question_answer 96) 8.2 litre of an ideal gas weight 9.0 gm at 300 K and 1 atm pressure. The molecular mass of the gas is:
A)
9
done
clear
B)
18
done
clear
C)
27
done
clear
D)
36
done
clear
View Answer play_arrow
question_answer 97) Variation of heat of reaction with temperature is known as:
A)
Kirchoffs equation
done
clear
B)
Vant Hoffs isotherm
done
clear
C)
Vant Hoffs isochore
done
clear
D)
none of the above .
done
clear
View Answer play_arrow
question_answer 98) A colloidal solution is subjected to an electric field. The colloidal particles more towards anode. The coagulation of die same solution studied using\[NaCl,BaCl\]and\[AlC{{l}_{3}}\] solutions. The coagulation power is in the order of:
A)
\[BaCl>NaCl>AlC{{l}_{3}}\]
done
clear
B)
\[NaCl>BaCl>AlC{{l}_{3}}\]
done
clear
C)
\[AlC{{l}_{3}}>BaCl>NaCl\]
done
clear
D)
\[NaCl>AlC{{l}_{3}}>BaC{{l}_{2}}\]
done
clear
View Answer play_arrow
question_answer 99) The water soluble compound is:
A)
\[AgI\]
done
clear
B)
\[AgF\]
done
clear
C)
\[AgCl\]
done
clear
D)
\[AgBr\]
done
clear
View Answer play_arrow
question_answer 100) Lithophone is a mixture of:
A)
\[CuS{{O}_{4}}+ZnS\]
done
clear
B)
\[CaS{{O}_{4}}+ZnS\]
done
clear
C)
\[BaS{{O}_{4}}+CaS{{O}_{4}}\]
done
clear
D)
\[BaS{{O}_{4}}+ZnS\]
done
clear
View Answer play_arrow
question_answer 101) Haemoglobin is a:
A)
protein
done
clear
B)
fibrous protein
done
clear
C)
globular protein
done
clear
D)
enzyme
done
clear
View Answer play_arrow
question_answer 102) LPP-lisa:
A)
bacterium
done
clear
B)
fungus
done
clear
C)
bacteriophage
done
clear
D)
cyanophage
done
clear
View Answer play_arrow
question_answer 103) Hormogonia are vegetative reporoductive structures of:
A)
Ulothrix
done
clear
B)
Oscillatoria
done
clear
C)
Spirogyra
done
clear
D)
Chara
done
clear
View Answer play_arrow
question_answer 104) Which of the following organisms are useful in sewage treatment?
A)
Eiiglena
done
clear
B)
Chlamydomonas
done
clear
C)
Chlorella
done
clear
D)
All of these
done
clear
View Answer play_arrow
question_answer 105) In earthworm, self fertilization cannot occur due to:
A)
hypogyny
done
clear
B)
protogyny
done
clear
C)
protandry
done
clear
D)
epigyny
done
clear
View Answer play_arrow
question_answer 106) Insectivorous plants can survive in:
A)
nitrogen deficient soils
done
clear
B)
carbonate deficient soils
done
clear
C)
soil rich in water and minerals
done
clear
D)
soil deficient in carbon
done
clear
View Answer play_arrow
question_answer 107) Coleoptile is:
A)
covering of root
done
clear
B)
covering of floral bud
done
clear
C)
covering of plumule
done
clear
D)
covering of seed
done
clear
View Answer play_arrow
question_answer 108) Tocopherol stands for:
A)
vitamin A
done
clear
B)
vitamin B complex
done
clear
C)
vitamin E
done
clear
D)
vitamin C
done
clear
View Answer play_arrow
question_answer 109) Which of the following is characteristic feature of fishes?
A)
venous heart and gills
done
clear
B)
epidermal scales
done
clear
C)
epidermal gills
done
clear
D)
tail and heart
done
clear
View Answer play_arrow
question_answer 110) Territoriality occurs as a result of:
A)
predation
done
clear
B)
competition
done
clear
C)
parasitism
done
clear
D)
cooperation
done
clear
View Answer play_arrow
question_answer 111) Who proposed mutation theory of evolution?
A)
Lamarck
done
clear
B)
Darwin
done
clear
C)
Hugo de Vries
done
clear
D)
Wiesmann
done
clear
View Answer play_arrow
question_answer 112) Mosquito:
A)
omnivors
done
clear
B)
sanguivores
done
clear
C)
frugivores
done
clear
D)
carrion eater
done
clear
View Answer play_arrow
question_answer 113) Goiter is caused due to deficiency of:
A)
vitamin D
done
clear
B)
vitamin C
done
clear
C)
iodine
done
clear
D)
iron
done
clear
View Answer play_arrow
question_answer 114) The terminal end of a chromosome is called:
A)
metamere
done
clear
B)
telomere
done
clear
C)
centromere
done
clear
D)
basal granule
done
clear
View Answer play_arrow
question_answer 115) The main function of Henles loop is:
A)
conservation of water
done
clear
B)
passage of urine
done
clear
C)
filteration of blood
done
clear
D)
absorption of nutrients
done
clear
View Answer play_arrow
question_answer 116) Regeneration of tail in lizard is an example of:
A)
morphogenesis
done
clear
B)
parthenogenesis
done
clear
C)
epimorphosis
done
clear
D)
heteromorphosis
done
clear
View Answer play_arrow
question_answer 117) The chemical substances produced by host plants to protect themselves against fungal infection is:
A)
pathogen
done
clear
B)
phytotoxin
done
clear
C)
phytoalexin
done
clear
D)
phytohormone
done
clear
View Answer play_arrow
question_answer 118) Clad odes are found in:
A)
Opuntia
done
clear
B)
Asparagus
done
clear
C)
Riiscus
done
clear
D)
Ficus
done
clear
View Answer play_arrow
question_answer 119) In\[{{C}_{4}}\]plants, the first\[C{{O}_{2}}\]acceptor is:
A)
pyruvate
done
clear
B)
phosphoenol pyruvate
done
clear
C)
ribulose biphosphate
done
clear
D)
ribulose phosphate
done
clear
View Answer play_arrow
question_answer 120) The sound producing organ in birds is:
A)
Glothis
done
clear
B)
larynx
done
clear
C)
syrinx
done
clear
D)
oesophagus
done
clear
View Answer play_arrow
question_answer 121) Lymphoid tissue is found in:
A)
thymus
done
clear
B)
tonsils
done
clear
C)
lymph nodes
done
clear
D)
all of the above
done
clear
View Answer play_arrow
question_answer 122) The symbiotic association of fungus and alga is called:
A)
mycorrhiza
done
clear
B)
lichen
done
clear
C)
exomycorrhiza
done
clear
D)
endomvcorrhiza
done
clear
View Answer play_arrow
question_answer 123) Veiamen is found in:
A)
aerial root of orchid
done
clear
B)
clinging root of orchid
done
clear
C)
leaf of orchid
done
clear
D)
leaf of angiosperm
done
clear
View Answer play_arrow
question_answer 124) In moss, the middle sterile part of capsule is called:
A)
foot
done
clear
B)
protonema
done
clear
C)
columella
done
clear
D)
spore case
done
clear
View Answer play_arrow
question_answer 125) In Cycas, endosperm is:
A)
diploid
done
clear
B)
triploid
done
clear
C)
polyploid
done
clear
D)
haploid
done
clear
View Answer play_arrow
question_answer 126) Peyers patches produce:
A)
mucous
done
clear
B)
trypsin.
done
clear
C)
lymphocytes
done
clear
D)
enterokinase
done
clear
View Answer play_arrow
question_answer 127) The life span of thrombocyte is:
A)
3-7 days
done
clear
B)
3-4 weeks
done
clear
C)
4-5 weeks
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 128) The term protobiogenesis used for theory of:
A)
panspermia
done
clear
B)
special creation
done
clear
C)
biochemical origin of life
done
clear
D)
spontaneous generation
done
clear
View Answer play_arrow
question_answer 129) The dominant animal of mesozoic era was:
A)
Climatius
done
clear
B)
Plesiosaurus
done
clear
C)
Ceratodiis
done
clear
D)
Latimeria
done
clear
View Answer play_arrow
question_answer 130) The cavity of gastrula is called:
A)
blastocoel
done
clear
B)
coelom
done
clear
C)
archanteron
done
clear
D)
haemocoel
done
clear
View Answer play_arrow
question_answer 131) Most important use of earthworm is:
A)
to catch fish
done
clear
B)
to make soil porous and fertile
done
clear
C)
to make soil more saline
done
clear
D)
to make dish
done
clear
View Answer play_arrow
question_answer 132) In which of the following animal, circulatory system is not involved in gas transport:
A)
trout
done
clear
B)
mouse
done
clear
C)
dragonfly
done
clear
D)
sparrow
done
clear
View Answer play_arrow
question_answer 133) Osphredium of Pila globosa is a:
A)
photoreceptor
done
clear
B)
thermoreceptor
done
clear
C)
tangoreceptor
done
clear
D)
chemoreceptor
done
clear
View Answer play_arrow
question_answer 134) Spindle fibres are made up of:
A)
actin
done
clear
B)
myosin
done
clear
C)
collagen
done
clear
D)
tubulin
done
clear
View Answer play_arrow
question_answer 135) Cristae of mitochondria help in:
A)
respiration
done
clear
B)
transpiration
done
clear
C)
photooxidation
done
clear
D)
photosynthesis
done
clear
View Answer play_arrow
question_answer 136) Which of the following is a sesamoid bone?
A)
Patella
done
clear
B)
palatine
done
clear
C)
pterygoid
done
clear
D)
presphenoid
done
clear
View Answer play_arrow
question_answer 137) Which of the following cranial nerve purely motor?
A)
vagus
done
clear
B)
trigeminal
done
clear
C)
facial
done
clear
D)
spinal accessory
done
clear
View Answer play_arrow
question_answer 138) Auxanometer is used to measure:
A)
length
done
clear
B)
ascept of sap
done
clear
C)
toot pressure
done
clear
D)
respiration
done
clear
View Answer play_arrow
question_answer 139) The study related to the structure and functions of cells is called as:
A)
palynology
done
clear
B)
histology
done
clear
C)
anatomy
done
clear
D)
cytology
done
clear
View Answer play_arrow
question_answer 140) Antiviral substance is:
A)
antibody
done
clear
B)
antigen
done
clear
C)
interferpn
done
clear
D)
all of the above
done
clear
View Answer play_arrow
question_answer 141) Lomentumisa:
A)
infloresence
done
clear
B)
seed
done
clear
C)
fruit
done
clear
D)
flower
done
clear
View Answer play_arrow
question_answer 142) Who discovered penicillin?
A)
Waksman
done
clear
B)
Darwin
done
clear
C)
Alexander Fleming
done
clear
D)
Benzman
done
clear
View Answer play_arrow
question_answer 143) Aspergillosis is caused by Aspergillns. Which of the following part is involved in this disease?
A)
heart
done
clear
B)
brain
done
clear
C)
lung
done
clear
D)
skin
done
clear
View Answer play_arrow
question_answer 144) Which of the following is a unicellular fungus?
A)
Yeast
done
clear
B)
Acetabiilaria
done
clear
C)
E. coli
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 145) Choanocytes perform:
A)
excretion
done
clear
B)
reproduction
done
clear
C)
nutrition
done
clear
D)
secretion
done
clear
View Answer play_arrow
question_answer 146) A condition of failure of kidney to form urine is:
A)
hematuria
done
clear
B)
creatinum
done
clear
C)
ketouria
done
clear
D)
aneuria
done
clear
View Answer play_arrow
question_answer 147) Root hairs originates from:
A)
cortex
done
clear
B)
hyperdermis
done
clear
C)
pericycle
done
clear
D)
epidermis
done
clear
View Answer play_arrow
question_answer 148) Radial bundles are found in:
A)
stem of angiosperms
done
clear
B)
root of angiosperms
done
clear
C)
monocot stem
done
clear
D)
dicotstem
done
clear
View Answer play_arrow
question_answer 149) Binomial nomenclature indicates:
A)
genus and species
done
clear
B)
genus and family
done
clear
C)
phylum and genus
done
clear
D)
species and subspecies
done
clear
View Answer play_arrow
question_answer 150) Which of the following is an excretory organ in Mollusca?
A)
nephridia
done
clear
B)
flame cells
done
clear
C)
kebers organ
done
clear
D)
malphigian organ
done
clear
View Answer play_arrow
question_answer 151) The structure of DNA is:
A)
triple helix
done
clear
B)
single helix
done
clear
C)
double helix
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 152) Who discovered double fertilization?
A)
R. Brown
done
clear
B)
R.Koch
done
clear
C)
Strassburger
done
clear
D)
Nawaschin
done
clear
View Answer play_arrow
question_answer 153) Tube feets are found in:
A)
Echinoderms
done
clear
B)
Arthropoda
done
clear
C)
Mollusca
done
clear
D)
Annelida
done
clear
View Answer play_arrow
question_answer 154) Bone is connected to muscle with the help of:
A)
tendon
done
clear
B)
ligament
done
clear
C)
cartilage
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 155) The contraction of gall bladder is due to:
A)
secretin
done
clear
B)
cholecystokinin
done
clear
C)
entergastrone
done
clear
D)
gastris
done
clear
View Answer play_arrow
question_answer 156) Transposon was discovered by:
A)
Sutton
done
clear
B)
B. McClintock
done
clear
C)
Watson and Crick
done
clear
D)
N. Ehle
done
clear
View Answer play_arrow
question_answer 157) Plasmids are found in:
A)
yeast
done
clear
B)
cyanobacteria
done
clear
C)
bacteria
done
clear
D)
Acetabularia
done
clear
View Answer play_arrow
question_answer 158) Grams staining was introduced by:
A)
Christian Gram
done
clear
B)
T. Gram
done
clear
C)
Pasteur
done
clear
D)
A. Fleming
done
clear
View Answer play_arrow
question_answer 159) Rabies is a:
A)
fungal disease
done
clear
B)
bacterial disease
done
clear
C)
viral disease
done
clear
D)
environmental disease
done
clear
View Answer play_arrow
question_answer 160) The nuclear material of T.M.V. is:
A)
DNA
done
clear
B)
RNA
done
clear
C)
both a and b
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 161) The term antibiotic was coined by:
A)
Fleming
done
clear
B)
Waksman
done
clear
C)
Darwin
done
clear
D)
Fohn
done
clear
View Answer play_arrow
question_answer 162) Transpiration is a necessary evil, this statement belongs to:
A)
Burgerstein
done
clear
B)
Reschke
done
clear
C)
Curtis
done
clear
D)
Willmer
done
clear
View Answer play_arrow
question_answer 163) Nodules with nitrigen fixing bacteria are found in the roots of:
A)
wheat
done
clear
B)
mustard
done
clear
C)
cotton
done
clear
D)
gram
done
clear
View Answer play_arrow
question_answer 164) Who discovered auxin?
A)
Went
done
clear
B)
Lethan
done
clear
C)
Miller
done
clear
D)
Fischer
done
clear
View Answer play_arrow
question_answer 165) Hypothetical plant hormones are:
A)
florigen
done
clear
B)
venalin
done
clear
C)
florigen and vemalin
done
clear
D)
none
done
clear
View Answer play_arrow
question_answer 166) Coiling of tendril around base is due to:
A)
thigmotropism
done
clear
B)
phototropism
done
clear
C)
geotropism
done
clear
D)
chemotropism
done
clear
View Answer play_arrow
question_answer 167) Who performed experiments on Chlorela to study photosynthesis?
A)
R. Hill
done
clear
B)
Warburg
done
clear
C)
Blackmann
done
clear
D)
Mortenson
done
clear
View Answer play_arrow
question_answer 168) Who coined the term chemiosmosis?
A)
P. Mitchell
done
clear
B)
Hill
done
clear
C)
Priestley
done
clear
D)
Calvin
done
clear
View Answer play_arrow
question_answer 169) The complete enzyme is called:
A)
apoenzyme
done
clear
B)
cofactcr
done
clear
C)
prosthetic group
done
clear
D)
holoenzyme
done
clear
View Answer play_arrow
question_answer 170) Zymase was discovered by:
A)
Buchner
done
clear
B)
Fischer
done
clear
C)
Kuhne
done
clear
D)
Koshland
done
clear
View Answer play_arrow
question_answer 171) Which of the following surrounds the vacuole?
A)
Cell wall
done
clear
B)
Mucopep tide layer
done
clear
C)
Tonoplast
done
clear
D)
Murein layer
done
clear
View Answer play_arrow
question_answer 172) Lomasomes are found in:
A)
algal cell
done
clear
B)
bacterial cell
done
clear
C)
cyanobacterial cell
done
clear
D)
fungal cell
done
clear
View Answer play_arrow
question_answer 173) Clover leaf model belongs to:
A)
\[t-RNA\]
done
clear
B)
DNA
done
clear
C)
centriole
done
clear
D)
flagella
done
clear
View Answer play_arrow
question_answer 174) Krebs7 cycle occurs in:
A)
chloroplast
done
clear
B)
cytoplasm
done
clear
C)
mitochondria
done
clear
D)
grana
done
clear
View Answer play_arrow
question_answer 175) Savannah is:
A)
grass land with scattered trees
done
clear
B)
part of ocean
done
clear
C)
part of tropical forest
done
clear
D)
alpine forest
done
clear
View Answer play_arrow
question_answer 176) Vegetation of tundra consists of:
A)
mosses
done
clear
B)
sedges
done
clear
C)
grasses
done
clear
D)
all of the above
done
clear
View Answer play_arrow
question_answer 177) The term biocoenosis was first of all used by:
A)
Forbes
done
clear
B)
Mobius
done
clear
C)
Reiter
done
clear
D)
Lindmann
done
clear
View Answer play_arrow
question_answer 178) Renewable resources are:
A)
forest
done
clear
B)
wild life
done
clear
C)
aquatic life
done
clear
D)
all of the above
done
clear
View Answer play_arrow
question_answer 179) Paul Millar discovered:
A)
B.H.C.
done
clear
B)
D.D.T.
done
clear
C)
Rotenone
done
clear
D)
Silicosis
done
clear
View Answer play_arrow
question_answer 180) World environment day is celebrated on.
A)
28th February
done
clear
B)
5th June
done
clear
C)
16th September
done
clear
D)
15th October
done
clear
View Answer play_arrow
question_answer 181) Terminating codons are:
A)
UAG
done
clear
B)
UAA
done
clear
C)
UGA
done
clear
D)
all of the above
done
clear
View Answer play_arrow
question_answer 182) Hydra captures the prey by injecting the chemical:
A)
toxoplasmin
done
clear
B)
hypnotoxin
done
clear
C)
kaliotoxin
done
clear
D)
Sarafotoxin
done
clear
View Answer play_arrow
question_answer 183) Leydig cells secrete:
A)
estrogen
done
clear
B)
testosterone
done
clear
C)
progesterone
done
clear
D)
corticosterone
done
clear
View Answer play_arrow
question_answer 184) The edible part of coconut:
A)
Epicarp
done
clear
B)
mesocarp
done
clear
C)
endocarp
done
clear
D)
endosperm
done
clear
View Answer play_arrow
question_answer 185) Larva of house fly lacks:
A)
eyes
done
clear
B)
wings
done
clear
C)
both a and b
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 186) Bat belongs to:
A)
chiroptera
done
clear
B)
dermoptera
done
clear
C)
edentats
done
clear
D)
insectivore
done
clear
View Answer play_arrow
question_answer 187) Protein, maintaining osmotic concentration of blood plasma isotonic with liter cellular body fluid and help in transport of\[{{O}_{2}},\]is:
A)
actin
done
clear
B)
haemoglobin
done
clear
C)
myoglobin
done
clear
D)
myosin
done
clear
View Answer play_arrow
question_answer 188) Melanin is a:
A)
pigment
done
clear
B)
hormone
done
clear
C)
enzyme
done
clear
D)
waste substances
done
clear
View Answer play_arrow
question_answer 189) Reproduction in Pcimrnnecnim is controlled by:
A)
micronucleus
done
clear
B)
macronucleus
done
clear
C)
cell wall
done
clear
D)
flagella
done
clear
View Answer play_arrow
question_answer 190) The egg case in female cockroach is formed by secretions of:
A)
collaterial gland
done
clear
B)
conglobate gland
done
clear
C)
prothoracic gland
done
clear
D)
mushroom gland
done
clear
View Answer play_arrow
question_answer 191) The type of veretebrae in sub order ophidia is:
A)
acoelous
done
clear
B)
amphicoelous
done
clear
C)
procoelous
done
clear
D)
heterocoelous
done
clear
View Answer play_arrow
question_answer 192) ABA is involved in:
A)
shoot elongation
done
clear
B)
root elongation
done
clear
C)
dormancy of seeds
done
clear
D)
increase in cell division
done
clear
View Answer play_arrow
question_answer 193) Balbiani rings are found in:
A)
polysomes
done
clear
B)
autosomes
done
clear
C)
polytenic chromosomes
done
clear
D)
sex chromosomes
done
clear
View Answer play_arrow
question_answer 194) The cytoplasmic units of inheritance are called as:
A)
genome
done
clear
B)
lethal gene
done
clear
C)
plasmagenes
done
clear
D)
all of the above
done
clear
View Answer play_arrow
question_answer 195) Acetabularia used in nucleocytoplasmic experiments by Hammerling, is:
A)
unicellular, uninucleate green alga
done
clear
B)
unicellular, anucleates blue-green alga
done
clear
C)
multicellular, multinucleate green alga
done
clear
D)
multicellular, uninucleate green alga
done
clear
View Answer play_arrow
question_answer 196) Bateson and Punnet (1909) discovered linkage while working on:
A)
Pisum saHvimi
done
clear
B)
Latli yrus odoratus
done
clear
C)
Drosoplula nielanogasfer
done
clear
D)
Lychiins alba
done
clear
View Answer play_arrow
question_answer 197) Maximum chromosome number (631) in haploid cell is found in:
A)
Dog
done
clear
B)
Ophioglossiim
done
clear
C)
Allium
done
clear
D)
Cat
done
clear
View Answer play_arrow
question_answer 198) The largest corpuscles in mammalian blood is:
A)
monocytes
done
clear
B)
erythrocytes
done
clear
C)
lymphocytes
done
clear
D)
basophils
done
clear
View Answer play_arrow
question_answer 199) Gastric juice secreted in stomach of men contains the enzymes:
A)
trypsin, rennin, lipase
done
clear
B)
pepsin, trypsin, rennin
done
clear
C)
pepsin, rennin, lipase
done
clear
D)
gastric, pepsin, rennin
done
clear
View Answer play_arrow
question_answer 200) Brunners glands and crypts of Lieberkuhn are situated in the:
A)
mucosa of intestine
done
clear
B)
mucosa of duodenum
done
clear
C)
mucosa of stomach
done
clear
D)
mucosa of pancreas
done
clear
View Answer play_arrow
question_answer 201) Directions: In each of the following questions one word in caps letters is followed by four words. Choose the word or phrase nearest in meaning: INSOLVENT:
A)
instalment
done
clear
B)
bankrupt
done
clear
C)
disastrous
done
clear
D)
deficient
done
clear
View Answer play_arrow
question_answer 202) Directions: In each of the following questions one word in caps letters is followed by four words. Choose the word or phrase nearest in meaning: BLASPHEMOUS:
A)
enormous
done
clear
B)
profane
done
clear
C)
insane
done
clear
D)
sceptic
done
clear
View Answer play_arrow
question_answer 203) Directions: In each of the following questions one word in caps letters is followed by four words. Choose the word or phrase nearest in meaning: FASCINATE:
A)
impress
done
clear
B)
express
done
clear
C)
suppress
done
clear
D)
attract
done
clear
View Answer play_arrow
question_answer 204) Directions: In each of the following questions one word in caps letters is followed by four words. Choose the word or phrase nearest in meaning: ARROGANT:
A)
shy
done
clear
B)
shameful
done
clear
C)
superior
done
clear
D)
proud
done
clear
View Answer play_arrow
question_answer 205) Directions: In each of the following questions one word in caps letters is followed by four words. Choose the word or phrase nearest in meaning: ORDINARY:
A)
common
done
clear
B)
uncommon
done
clear
C)
primary
done
clear
D)
ordinary
done
clear
View Answer play_arrow
question_answer 206) Directions: In each of the following questions one word in caps letters is followed by four words. Choose the word or phrase nearest in meaning: ANONYMOUS:
A)
generous
done
clear
B)
well-known
done
clear
C)
famous
done
clear
D)
unknown
done
clear
View Answer play_arrow
question_answer 207) Directions: In each of the following questions one word in caps letters is followed by four words. Choose the word or phrase nearest in meaning: FERVOUR:
A)
anger
done
clear
B)
pride
done
clear
C)
passion
done
clear
D)
compassion
done
clear
View Answer play_arrow
question_answer 208) Directions: In each of the following questions find out which part of the sentence has an error. If there is no mistake the answer is no error.
A)
I fell of my bicycle
done
clear
B)
When I was there and
done
clear
C)
I still have a scar on my knee
done
clear
D)
No error
done
clear
View Answer play_arrow
question_answer 209) Directions: In each of the following questions find out which part of the sentence has an error. If there is no mistake the answer is no error.
A)
I have
done
clear
B)
five
done
clear
C)
ten-rupees
done
clear
D)
notes
done
clear
View Answer play_arrow
question_answer 210) Directions: In each of the following questions find out which part of the sentence has an error. If there is no mistake the answer is no error.
A)
Ravi told me
done
clear
B)
that his wife was ill
done
clear
C)
for fifteen days
done
clear
D)
No error
done
clear
View Answer play_arrow
question_answer 211) Directions: In each of the following questions find out which part of the sentence has an error. If there is no mistake the answer is no error.
A)
I have
done
clear
B)
not
done
clear
C)
yet taken
done
clear
D)
my meals
done
clear
View Answer play_arrow
question_answer 212) Directions: In each of the following questions find out which part of the sentence has an error. If there is no mistake the answer is no error.
A)
He is not happy
done
clear
B)
because he is getting
done
clear
C)
a less salary
done
clear
D)
No error
done
clear
View Answer play_arrow
question_answer 213) Directions: In each of the following questions find out which part of the sentence has an error. If there is no mistake the answer is no error.
A)
Ravi and
done
clear
B)
I cannot
done
clear
C)
bear
done
clear
D)
your separation
done
clear
View Answer play_arrow
question_answer 214) Directions: In each of the following questions find out which part of the sentence has an error. If there is no mistake the answer is no error.
A)
ten new members
done
clear
B)
have been enrolled
done
clear
C)
and seven resigned
done
clear
D)
No error
done
clear
View Answer play_arrow
question_answer 215) It is high time you must leam to behave properly:
A)
learned
done
clear
B)
will learn
done
clear
C)
should learn
done
clear
D)
no improvement
done
clear
View Answer play_arrow
question_answer 216) I am use to hard work:
A)
work hard
done
clear
B)
work hardly
done
clear
C)
hard working
done
clear
D)
No improvement
done
clear
View Answer play_arrow
question_answer 217) I must has forgotten to switch off the light:
A)
forget
done
clear
B)
have forget
done
clear
C)
have forgotten
done
clear
D)
No improvement
done
clear
View Answer play_arrow
question_answer 218) If I was God I should eliminate all sufferings:
A)
am
done
clear
B)
were
done
clear
C)
been
done
clear
D)
No improvement
done
clear
View Answer play_arrow
question_answer 219) I knew Mr. Kumar since 1981:
A)
I have been knowing
done
clear
B)
know
done
clear
C)
have known
done
clear
D)
No improvement
done
clear
View Answer play_arrow
question_answer 220) Please blow up the candle:
A)
turn off
done
clear
B)
blow off
done
clear
C)
put off
done
clear
D)
No improvement
done
clear
View Answer play_arrow
question_answer 221) In which year the. Life Insurance Corporation of India was established?
A)
1964
done
clear
B)
1963
done
clear
C)
1956
done
clear
D)
1949
done
clear
View Answer play_arrow
question_answer 222) Hieun Tsang- came in India in the reign
A)
Chandragupta Mauraya
done
clear
B)
Harsh
done
clear
C)
Ashoka
done
clear
D)
Kanishka
done
clear
View Answer play_arrow
question_answer 223) Which state is the highest producer of cotton in India?
A)
Maharashtra
done
clear
B)
Uttar Pradesh
done
clear
C)
Tamil Nadu
done
clear
D)
Gujarat
done
clear
View Answer play_arrow
question_answer 224) Which country is known as the Playground of Europe?
A)
Germany
done
clear
B)
Spain
done
clear
C)
Denmark
done
clear
D)
Switzerland
done
clear
View Answer play_arrow
question_answer 225) In which year OPEC was constituted?
A)
1960
done
clear
B)
1965
done
clear
C)
1966
done
clear
D)
1967
done
clear
View Answer play_arrow
question_answer 226) Who wrote the famous book Urvashi?
A)
Bishnu Dev
done
clear
B)
Yogesh Chaddha
done
clear
C)
Munshi Prem Chand
done
clear
D)
Ramdhari Singh Dinkar.
done
clear
View Answer play_arrow
question_answer 227) The name of the first newspaper in India was:
A)
Times of India
done
clear
B)
Bengal Gazette
done
clear
C)
Hindustan Times
done
clear
D)
Bombay Samachar
done
clear
View Answer play_arrow
question_answer 228) In which country the largest temple of world is situated?
A)
India
done
clear
B)
Cambodia
done
clear
C)
Bhutan
done
clear
D)
Nepal
done
clear
View Answer play_arrow
question_answer 229) In which year National School of Drama (NSD) was set up?
A)
1960
done
clear
B)
1959
done
clear
C)
1970
done
clear
D)
1976
done
clear
View Answer play_arrow
question_answer 230) Which article of constitution bans untouchability?
A)
Art. 14
done
clear
B)
Art. 15
done
clear
C)
Art. 16
done
clear
D)
Art. 17
done
clear
View Answer play_arrow
question_answer 231) The solution of equation\[x-y=0\]and
A)
\[x=2\]and\[y=2\]
done
clear
B)
\[x=-2\]and \[y=1\]
done
clear
C)
\[x=-1\]and \[y=2\]
done
clear
D)
\[x=2\]and\[y=-1\]
done
clear
View Answer play_arrow
question_answer 232) lf\[A=\frac{x+1}{x-1},\]the the value of\[A-\frac{1}{A}\]is:
A)
\[\frac{1}{{{x}^{2}}-1}\]
done
clear
B)
\[\frac{4x}{{{x}^{2}}-1}\]
done
clear
C)
\[\frac{x}{x-1}\]
done
clear
D)
\[\frac{x}{{{x}^{2}}-1}\]
done
clear
View Answer play_arrow
question_answer 233) In equilateral triangle of. side 2a, the length of its altitude will be:
A)
\[a\sqrt{3}\]
done
clear
B)
\[a\]
done
clear
C)
\[3a\]
done
clear
D)
\[{{a}^{2}}\]
done
clear
View Answer play_arrow
question_answer 234) A car covers 420 km with a constant speed If its speed were 10 km/h more it would have taken one hour less to cover the distance. The speed of the car is:
A)
60
done
clear
B)
70
done
clear
C)
80
done
clear
D)
120
done
clear
View Answer play_arrow
question_answer 235) A merchant lent out Rs. 1,000 in two parts one at 8% and the other at 10% interest. The yearly averge comes out to be 9.2% .The amount lent in two parts is:
A)
400, 800
done
clear
B)
400, 600
done
clear
C)
700, 800
done
clear
D)
900, 1000
done
clear
View Answer play_arrow
question_answer 236) Melt is related to -Liquid in the same way Freeze is related to:
A)
Ice
done
clear
B)
Crystal
done
clear
C)
Water
done
clear
D)
Cubes
done
clear
View Answer play_arrow
question_answer 237) Find out the wrong number \[864,\,\,420,\,\,200,\,\,96,\,\,40,\,\,16,\,\,6\]
A)
420
done
clear
B)
200
done
clear
C)
96
done
clear
D)
40
done
clear
View Answer play_arrow
question_answer 238)
A)
4
done
clear
B)
8
done
clear
C)
20
done
clear
D)
14
done
clear
View Answer play_arrow
question_answer 239)
Statements: (i) Some cars are trains (ii) Some trains are cats. Conclusion: (I) No car is cat (II) Some cats are trains (III) Some cars are cats (IV) All trains are cars
A)
only either I or II or III follows
done
clear
B)
only either I or III and II follows
done
clear
C)
only either I or III follows
done
clear
D)
only I, II and III follows
done
clear
View Answer play_arrow
question_answer 240)
Consider the venn diagram given below: The number in the venn diagram indicates the number of persons reading
A)
5000
done
clear
B)
5400
done
clear
C)
6000
done
clear
D)
6250
done
clear
View Answer play_arrow
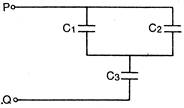
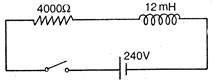 15 mA, then potential difference across the inductor will be:
15 mA, then potential difference across the inductor will be:
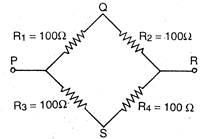
 the newspaper. The diagram is drawn after surveying 50 persons. In a population of 10,000 how many can be expected to seed at least two newspaper?
the newspaper. The diagram is drawn after surveying 50 persons. In a population of 10,000 how many can be expected to seed at least two newspaper?
