question_answer 1) If\[{{a}_{r}}\]and\[{{a}_{t}}\]represent radial and tangential accelerations, the motion of a particle will be uniformly circular if:
A)
\[{{a}_{r}}=0\]and \[{{a}_{t}}=0\]
done
clear
B)
\[{{a}_{r}}=0\]but \[{{a}_{t}}\ne 0\]
done
clear
C)
\[{{a}_{r}}\ne 0\]but \[{{a}_{t}}=0\]
done
clear
D)
\[{{a}_{r}}\ne 0\]and\[{{a}_{t}}\ne 0\]
done
clear
View Answer play_arrow
question_answer 2) The eccentricity of earths orbit is 0.0167. The ratio of its maximum speed in its orbit to its minimum speed is:
A)
2.507
done
clear
B)
1.0339
done
clear
C)
8.324
done
clear
D)
1.000
done
clear
View Answer play_arrow
question_answer 3) The radius R of the soap bubble is doubled under isothermal condition. If T be the surface tension of soap bubble, the required surface energy in doing so is given by:
A)
\[32\pi {{R}^{2}}T\]
done
clear
B)
\[24\pi {{R}^{2}}T\]
done
clear
C)
\[8\pi {{R}^{2}}T\]
done
clear
D)
\[4\pi {{R}^{2}}T\]
done
clear
View Answer play_arrow
question_answer 4) Mercury boils at\[367{}^\circ C\]. However, mercury thermometers are made such that they can measure temperature upto\[500{}^\circ C\]. This is done by:
A)
maintaining vacuum above mercury column in the stem of the thermometer
done
clear
B)
filling nitrogen gas at high pressure above the mercury column
done
clear
C)
filling oxygen gas at high pressure above the mercury column
done
clear
D)
filling nitrogen gas at low pressure above the mercury column
done
clear
View Answer play_arrow
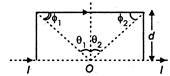
question_answer 5)
What is the magnetic field at 0 due to current in the infinite wire forming a loop as shown in the following figure?
A)
\[\frac{{{\mu }_{0}}I}{4\pi d}(\cos {{\phi }_{1}}+\cos {{\phi }_{2}})\]
done
clear
B)
\[\frac{{{\mu }_{0}}}{4\pi }\times \frac{2I}{d}\]
done
clear
C)
\[\frac{{{\mu }_{0}}I}{4\pi d}(\sin {{\phi }_{1}}+\sin {{\phi }_{2}})\]
done
clear
D)
\[\frac{{{\mu }_{0}}}{4\pi }\times \frac{I}{d}\]
done
clear
View Answer play_arrow
question_answer 6) A prism of refractive index \[\sqrt{2}\]has a refracting angle of\[60{}^\circ \]. At what angle a ray must be incident on it so that it suffers a minimum deviation?
A)
\[45{}^\circ \]
done
clear
B)
\[60{}^\circ \]
done
clear
C)
\[90{}^\circ \]
done
clear
D)
\[180{}^\circ \]
done
clear
View Answer play_arrow
question_answer 7) A cone filled with water is revolved in a vertical circle of radius 4 m and the water does not fall down. What must be the maximum period of revolution?
A)
2s
done
clear
B)
4s
done
clear
C)
1s
done
clear
D)
6s
done
clear
View Answer play_arrow
question_answer 8) A transparent cube of 15 cm edge contains a small air bubble. Its apparent depth when viewed through one face is 6 cm and when viewed through the opposite face is 4 cm. Then, the refractive index of the material of the cube is:
A)
2.0
done
clear
B)
2.5
done
clear
C)
1.6
done
clear
D)
1.5
done
clear
View Answer play_arrow
question_answer 9) A plane metallic sheet is placed with its face parallel to lines of magnetic induction B of a uniform field. A particle of mass m and charge q is projected with a velocity\[v\]from a distance \[d\]from the plane normal to the lines of induction. Then, the maximum velocity of projection for which the particle does not hit the plate is:
A)
\[\frac{2Bqd}{m}\]
done
clear
B)
\[\frac{Bqd}{m}\]
done
clear
C)
\[\frac{Bqd}{2m}\]
done
clear
D)
\[\frac{Bqm}{d}\]
done
clear
View Answer play_arrow
question_answer 10) A capacitor is connected to .a cell of emf E having someJntemal resistance r. The potential difference across the:
A)
cell is <E
done
clear
B)
cell is E
done
clear
C)
capacitor is > E
done
clear
D)
capacitor is < E
done
clear
View Answer play_arrow
question_answer 11) The length, breadth and thickness of a block are given by\[l=12\text{ }cm,\text{ }b=6\text{ }cm\]and\[t=2.45\]cm. The volume of the block according to the idea of significant figures should be:
A)
\[1\times {{10}^{2}}c{{m}^{3}}\]
done
clear
B)
\[2\times {{10}^{2}}c{{m}^{3}}\]
done
clear
C)
\[1.763\times {{10}^{2}}c{{m}^{2}}\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 12) Five particles of mass 2 kg are attached to the rim of a circular disc of radius 0.1 m and negligible mass. Moment of inertia of the system about the axis passing through the centre of the disc and perpendicular to its place is:
A)
1 kg\[{{m}^{2}}\]
done
clear
B)
0.1 kg\[{{m}^{2}}\]
done
clear
C)
2 kg\[{{m}^{2}}\]
done
clear
D)
0.2 kg\[{{m}^{2}}\]
done
clear
View Answer play_arrow
question_answer 13) The radius of the convex surface of plano-convex lens is 20 cm and the refractive index of the material of the lens is 1.5. The focal length is:
A)
30cm
done
clear
B)
50cm
done
clear
C)
20cm
done
clear
D)
40cm
done
clear
View Answer play_arrow
question_answer 14) An ice-cube of density\[900\text{ }kg/{{m}^{3}}\]is floating in water of density\[1000\text{ }kg/{{m}^{3}}\]. The percentage of, volume of ice-cube outside the water is:
A)
20%
done
clear
B)
35%
done
clear
C)
10%
done
clear
D)
25%
done
clear
View Answer play_arrow
question_answer 15) A sphere of diameter 0.2 m and mass 2 kg is rolling on an inclined plane with velocity\[v=0\]. 5 m/s. The kinetic energy of the sphere is:
A)
0.1 J
done
clear
B)
0.3J
done
clear
C)
0.5 J
done
clear
D)
0.42 J
done
clear
View Answer play_arrow
question_answer 16) An electron moves at right angle to a magnetic field of\[1.5\times {{10}^{-2}}\]tesia with a speed of\[6\times {{10}^{7}}\]m/s. If the specific charge of the electron is \[1.7\times {{10}^{11}}\]coulomb/kg, the radius of the circular path will be:
A)
2.9 cm
done
clear
B)
3.9 cm
done
clear
C)
2.35 cm
done
clear
D)
2 cm
done
clear
View Answer play_arrow
question_answer 17) Silver has a work function of 4.7 eV. When ultraviolet light of wavelength 100 nm is incident upon it, a potential of 7.7 V is required to stop the photoelectrons from reaching the collector late. How mcuh potential will be required to stop photoelectrons when light of wavelength 2p0 nm is incident upon silver?
A)
1.5V
done
clear
B)
3.85V
done
clear
C)
2.35V
done
clear
D)
15.4V
done
clear
View Answer play_arrow
question_answer 18) Velocity of sound waves in air is 330 m/s. For a particular sound in air, a path difference of 40 cm is equivalent to a phase difference of 1.6 tt. The frequency or the wave is:
A)
165 Hz
done
clear
B)
150 Hz
done
clear
C)
660 Hz
done
clear
D)
330 Hz
done
clear
View Answer play_arrow
question_answer 19) A particle is executing two different simple harmonic motions, mutually perpendicular, of different amplitudes and having phase difference of n/ 2. The path of the particle will be:
A)
circular
done
clear
B)
straight line
done
clear
C)
parabolic
done
clear
D)
elliptical
done
clear
View Answer play_arrow
question_answer 20) Equations of motion in the same direction are given by: \[{{y}_{1}}=2a\,\sin (\omega t-kx)\] \[{{y}_{2}}=2a\,\,\sin \,(\omega t-kx-\theta )\] The amplitude of the medium particle will be:
A)
\[2a\cos \theta \]
done
clear
B)
\[\sqrt{2}a\cos \theta \]
done
clear
C)
\[4a\cos \theta /2\]
done
clear
D)
\[\sqrt{2}a\cos \theta /2\]
done
clear
View Answer play_arrow
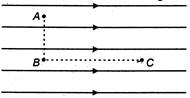
question_answer 21)
Figure shows three points A, B and C in a region of uniform electric field\[\overrightarrow{E}\]. The line AB is perpendicular and BC is parallel to the field lines. Then which of the following holds good?
A)
\[{{V}_{A}}={{V}_{B}}={{V}_{C}}\]
done
clear
B)
\[{{V}_{A}}={{V}_{B}}>{{V}_{C}}\]
done
clear
C)
\[{{V}_{A}}={{V}_{B}}<{{V}_{C}}\]
done
clear
D)
\[{{V}_{A}}>{{V}_{B}}={{V}_{C}}\]
done
clear
View Answer play_arrow
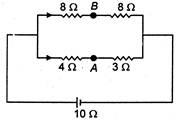
question_answer 22)
The potential difference between points A and B is:
A)
\[\frac{20}{7}V\]
done
clear
B)
\[\frac{40}{7}V\]
done
clear
C)
\[\frac{10}{7}V\]
done
clear
D)
zero
done
clear
View Answer play_arrow
question_answer 23) A short linear object of length b lies along the axis of a concave mirror of focal length\[f\]at a distance u from the pole of the mirror, what is the size of image?
A)
\[\left( \frac{f}{u-f} \right)b\]
done
clear
B)
\[{{\left( \frac{f}{u-f} \right)}^{2}}b\]
done
clear
C)
\[\left( \frac{f}{u-f} \right){{b}^{2}}\]
done
clear
D)
\[\left( \frac{f}{u-f} \right)\]
done
clear
View Answer play_arrow
question_answer 24) A closed organ pipe and an open organ pipe are tuned to the same fundamental frequency. What is the ratio of their lengths?
A)
\[1:2\]
done
clear
B)
\[2:1\]
done
clear
C)
\[2:3\]
done
clear
D)
\[4:3\]
done
clear
View Answer play_arrow
question_answer 25) Regarding a semiconductor which one of the following is wrong?
A)
There are no free electrons at room temperature
done
clear
B)
There are no free electrons at OK
done
clear
C)
The number of free electrons increases with rise of temperature
done
clear
D)
The charge carries are electrons and holes
done
clear
View Answer play_arrow
question_answer 26) A steel scale measures the length of a copper wire as 80.0 cm, when both are at\[20{}^\circ C,\]the calibration temperature for the scale. What would the scale read for the length of the wire when both are at\[40{}^\circ C\]? Given, \[{{a}_{S}}=11\times {{10}^{-6}}per{}^\circ C\] and\[{{a}_{Cu}}=17\times {{10}^{-6}}per{}^\circ C\]:
A)
80.0096 cm
done
clear
B)
80.0272 cm
done
clear
C)
1 cm
done
clear
D)
25.2 cm
done
clear
View Answer play_arrow
question_answer 27) A tank is filled with water upto height H. When a hole is made at a distance h below the level of water, what will be the horizontal range of water jet?
A)
\[2\sqrt{h(H-h)}\]
done
clear
B)
\[4\sqrt{h(H+h)}\]
done
clear
C)
\[4\sqrt{h(H-h)}\]
done
clear
D)
\[2\sqrt{h(H+h)}\]
done
clear
View Answer play_arrow
question_answer 28) A raft of wood of mass 120 kg floats in water. The weight that can be put on the raft to make it just sink, should be: \[({{d}_{raft}}=600kg/{{m}^{3}})\]
A)
80 kg
done
clear
B)
50 kg
done
clear
C)
60 kg
done
clear
D)
30 kg
done
clear
View Answer play_arrow
question_answer 29) Nuclear fusion is common to the pair:
A)
thermonuclear reactor, uranium based nuclear reactor
done
clear
B)
energy production is sun, uranium based nuclear reactor
done
clear
C)
energy production in sun, hydrogen bomb
done
clear
D)
disintegration of heavy nuclei, hydrogen bomb
done
clear
View Answer play_arrow
question_answer 30) Which of the following statements is true for an n-type semiconductor?
A)
The donor level lies closely below the bottom of the conduction band
done
clear
B)
The donor level lies closely above the top of the valence band
done
clear
C)
The donor level lies at the halfway mark of, the forbidden energy gap
done
clear
D)
None. of the above
done
clear
View Answer play_arrow
question_answer 31) The minimum wavelength of X-rays emitted by X-ray tube is\[0.4125\overset{o}{\mathop{\text{A}}}\,\]. The accelerating voltage is:
A)
30 kV
done
clear
B)
50 kV
done
clear
C)
80 kV
done
clear
D)
60 Kv
done
clear
View Answer play_arrow
question_answer 32) A monoatomic gas supplied the heat Q very slowly keeping the pressure constant. The work done by the gas will be:
A)
\[\frac{2}{3}Q\]
done
clear
B)
\[\frac{3}{5}Q\]
done
clear
C)
\[\frac{2}{5}Q\]
done
clear
D)
\[\frac{1}{5}Q\]
done
clear
View Answer play_arrow
question_answer 33) The plane face of a plano-convex lens of focal length 20 cm is silvered. What type of mirror will it become and of what focal length\[f\]?
A)
Convex,\[f=20\text{ }cm\]
done
clear
B)
Concave,\[f=20\text{ }cm\]
done
clear
C)
Convex,\[f=10\text{ }cm\]
done
clear
D)
Concave,\[f=10\text{ }cm\]
done
clear
View Answer play_arrow
question_answer 34) The temperature of the black body increases from T to 2T. The factor by which the rate of emission will increase, is:
A)
4
done
clear
B)
2
done
clear
C)
16
done
clear
D)
8
done
clear
View Answer play_arrow
question_answer 35) A police jeep is chasing with velocity of 45 km/h, a thief in another jeep moving with velocity 153 km/h. Police fires a bullet with muzzle velocity of 180 m/s. The velocity with which it will strike the car of the thief, is:
A)
150 m/s
done
clear
B)
27 m/s
done
clear
C)
450 m/s
done
clear
D)
250 m/s
done
clear
View Answer play_arrow
question_answer 36) In a sinusoidal wave, the time required for a particular point to move from maximum displacement to zero displacement is 0.17 s. The frequency of the wave is:
A)
1.47 Hz
done
clear
B)
2.94 Hz
done
clear
C)
0.73 Hz
done
clear
D)
0.36 Hz
done
clear
View Answer play_arrow
question_answer 37) An L-C circuit is in the state of resonance. If \[C=0.1\mu F\]and\[L=0.25\text{ }H,\]neglecting ohmic resistance of circuit, what is the frequency of oscillations?
A)
1007 Hz
done
clear
B)
100 Hz
done
clear
C)
109 Hz
done
clear
D)
500 Hz
done
clear
View Answer play_arrow
question_answer 38) A person who can see things most clearly at a distance of 10 cm, requires spectacles to enable to see clearly things at a distance of 30 cm. What should be the focal length of the spectacles?
A)
15 cm (concave)
done
clear
B)
15 cm (convex)
done
clear
C)
10 cm
done
clear
D)
0
done
clear
View Answer play_arrow
question_answer 39) A particle of mass\[m=5\]units is moving with a uniform speed\[v=3\sqrt{2}\]units in the XOY plane along the line\[y=x+4\]. The magnitude of the angular momentum of the particle about the origin is:
A)
60 unit
done
clear
B)
\[40\sqrt{2}\]unit
done
clear
C)
zero
done
clear
D)
7.5 unit
done
clear
View Answer play_arrow
question_answer 40) When temperature of an ideal gas is increased from\[27{}^\circ C\]to\[227{}^\circ C,\]its rms speed is changed from 400 m/s to\[{{v}_{s}}\]. The\[{{v}_{s}}\]is:
A)
516 m/s
done
clear
B)
450 m/s
done
clear
C)
310 m/s
done
clear
D)
746 m/s
done
clear
View Answer play_arrow
question_answer 41) A bar magnet of magnetic moment\[\overrightarrow{M}\]is placed in the magnetic field B. The torque acting on the magnet is:
A)
\[\overset{\to }{\mathop{M}}\,\times \overset{\to }{\mathop{B}}\,\]
done
clear
B)
\[\overset{\to }{\mathop{M}}\,-\overset{\to }{\mathop{B}}\,\]
done
clear
C)
\[\frac{1}{2}\overset{\to }{\mathop{M}}\,\times \overset{\to }{\mathop{B}}\,\]
done
clear
D)
\[\overset{\to }{\mathop{M}}\,+\overset{\to }{\mathop{B}}\,\]
done
clear
View Answer play_arrow
question_answer 42) A capacitor of capacitance 6uF is charged upto 100 V. The energy stored in the capacitor is:
A)
0.6 J
done
clear
B)
0.06 J
done
clear
C)
0.03 J
done
clear
D)
0.3 J
done
clear
View Answer play_arrow
question_answer 43) The radius of orbit of a planet is two times that of the earth. The time period of planet is:
A)
4.2 T
done
clear
B)
2.8 T
done
clear
C)
5.6 T
done
clear
D)
8.4 T
done
clear
View Answer play_arrow
question_answer 44) A body falls from a height\[h=200\text{ }m\]. The ratio of distance travelled in each 2 s, during\[t=0\]to \[t=6\]s of the journey is:
A)
\[1:4:9\]
done
clear
B)
\[1:2:4\]
done
clear
C)
\[1:3:5\]
done
clear
D)
\[1:2:3\]
done
clear
View Answer play_arrow
question_answer 45) To make the frequency double of a spring oscillator, we have to:
A)
reduce the mass to one-fourth
done
clear
B)
quardruple the mass
done
clear
C)
double the mass
done
clear
D)
half the mass
done
clear
View Answer play_arrow
question_answer 46) A particle executing SHM has amplitude 0.01 m and frequency 60 Hz. The maximum acceleration of particle is:
A)
\[60{{\pi }^{2}}m/{{s}^{2}}\]
done
clear
B)
\[80{{\pi }^{2}}m/{{s}^{2}}\]
done
clear
C)
\[120{{\pi }^{2}}m/{{s}^{2}}\]
done
clear
D)
\[144{{\pi }^{2}}m/{{s}^{2}}\]
done
clear
View Answer play_arrow
question_answer 47) When a wave travels in a medium the particles displacement is , given by the equation \[y=0.03\sin \pi (2t-0.01x),\]where x and y are in metre and t in second. The wavelength of the wave is:
A)
200m
done
clear
B)
100m
done
clear
C)
20 m
done
clear
D)
10 m
done
clear
View Answer play_arrow
question_answer 48) If 300 ml of a gas at\[27{}^\circ C\]is cooled to\[7{}^\circ C\]at constant pressure, then its final volume will be:
A)
540 ml
done
clear
B)
350 ml
done
clear
C)
280 ml
done
clear
D)
135 ml
done
clear
View Answer play_arrow
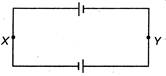
question_answer 49)
Two similar accumulators each of emf E and internal resistance r are connected as shown in the following figure. Then the potential difference between X and Y is:
A)
\[2E\]
done
clear
B)
\[E\]
done
clear
C)
zero
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 50) When sound waves travel from air to water, which one of the following remains constant?
A)
Time period
done
clear
B)
Frequency
done
clear
C)
Velocity
done
clear
D)
Wavelength
done
clear
View Answer play_arrow
question_answer 51) If error in measurement of radius of sphere is 1%, what will be the error in measurement of volume?
A)
1%
done
clear
B)
1/3%
done
clear
C)
3%
done
clear
D)
10%
done
clear
View Answer play_arrow
question_answer 52) Consider the following equation of Bernoullis theorem. \[P+\frac{1}{2}\rho {{v}^{2}}+\rho gh=K\](constant) The dimensions of K/P are same as that of which of the following?
A)
Thrust
done
clear
B)
Pressure
done
clear
C)
Angle
done
clear
D)
Viscosity
done
clear
View Answer play_arrow
question_answer 53) Why the dam of water reservoir is thick at the bottom?
A)
Quantity of water increases with depth
done
clear
B)
Density of water increases with depth
done
clear
C)
Pressure of water increases with depth
done
clear
D)
Temperature of water increases with depth
done
clear
View Answer play_arrow
question_answer 54) The length of seconds pendulum is 1 m on earth. If mass and diameter of a planet is doubled than that of earth, then its length becomes:
A)
1 m
done
clear
B)
2m
done
clear
C)
0.5 m
done
clear
D)
4 m
done
clear
View Answer play_arrow
question_answer 55) A beaker is completely filled with water at\[4{}^\circ C\] It will overflow if:
A)
heated above\[4{}^\circ C\]
done
clear
B)
cooled below\[4{}^\circ C\]
done
clear
C)
both heated and cooled above and below \[4{}^\circ C\]respectively
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 56) A coin is dropped in a lift. It takes time\[{{t}_{1}}\]to reach the floor when lift is stationary. It takes time\[{{t}_{2}}\]when lift is moving up with constant acceleration. Then:
A)
\[{{t}_{1}}>{{t}_{2}}\]
done
clear
B)
\[{{t}_{2}}>{{t}_{1}}\]
done
clear
C)
\[{{t}_{1}}={{t}_{2}}\]
done
clear
D)
\[{{t}_{1}}>>{{t}_{2}}\]
done
clear
View Answer play_arrow
question_answer 57) An object is placed at a distance equal to focal length of convex mirror. If the focal length of the mirror be\[f,\]then the distance of the image from the pole of the mirror is:
A)
less than\[f\]
done
clear
B)
equal to\[f\]
done
clear
C)
more than\[f\]
done
clear
D)
infinity
done
clear
View Answer play_arrow
question_answer 58) What is the angle between the electric dipole moment and the electric field strength due to it on the equatorial line?
A)
\[0{}^\circ \]
done
clear
B)
\[90{}^\circ \]
done
clear
C)
\[180{}^\circ \]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 59) A wire carrying current\[I\]and other carrying\[2I\] in the same direction produce a magnetic field B at the mid-point. What will be the field when\[2I\]wire is switched off?
A)
B/2
done
clear
B)
2B
done
clear
C)
B
done
clear
D)
4B
done
clear
View Answer play_arrow
question_answer 60) After 2 hours 1/16th of initial amount of a certain radioactive isotope remains undecayed. The half-life of the isotope is:
A)
15min
done
clear
B)
30 min
done
clear
C)
45 min
done
clear
D)
60 min
done
clear
View Answer play_arrow
question_answer 61) How many isomers are possible for the alkane\[{{C}_{4}}{{H}_{10}}\]?
A)
3
done
clear
B)
5
done
clear
C)
2
done
clear
D)
4
done
clear
View Answer play_arrow
question_answer 62) Geometrical isomerism is possible in case of:
A)
pentene-2
done
clear
B)
propane
done
clear
C)
pentane
done
clear
D)
ethane
done
clear
View Answer play_arrow
question_answer 63) The wrong IUPAC name of the compound \[C{{H}_{3}}CHOH.C{{H}_{2}}C{{H}_{3}}\]is:
A)
butan-2-ol
done
clear
B)
2-butanol
done
clear
C)
butanol-2
done
clear
D)
1-methyl-propanol-1
done
clear
View Answer play_arrow
question_answer 64) The number of isomers of the compound with molecular formula\[{{C}_{2}}{{H}_{2}}B{{r}_{2}}\]is:
A)
4
done
clear
B)
3
done
clear
C)
5
done
clear
D)
2
done
clear
View Answer play_arrow
question_answer 65) The number of ether isomers possible for \[{{C}_{4}}{{H}_{10}}O\]are:
A)
2
done
clear
B)
5
done
clear
C)
4
done
clear
D)
3
done
clear
View Answer play_arrow
question_answer 66) Correct gas equation is:
A)
\[\frac{{{P}_{1}}{{T}_{1}}}{{{V}_{1}}}=\frac{{{P}_{2}}{{T}_{2}}}{{{V}_{2}}}\]
done
clear
B)
\[\frac{{{V}_{1}}{{T}_{2}}}{{{P}_{1}}}=\frac{{{V}_{2}}{{T}_{1}}}{{{P}_{2}}}\]
done
clear
C)
\[\frac{{{P}_{1}}{{V}_{1}}}{{{P}_{2}}{{V}_{2}}}=\frac{{{T}_{1}}}{{{T}_{2}}}\]
done
clear
D)
\[\frac{{{V}_{1}}{{V}_{2}}}{{{T}_{1}}{{T}_{2}}}={{P}_{1}}{{P}_{2}}\]
done
clear
View Answer play_arrow
question_answer 67) Which combinations of quantum numbers\[n,l,\] \[m\]and s for the electron in an atom does not provide a permissible solution of the wave equation?
A)
\[3,2,1,\frac{1}{2}\]
done
clear
B)
\[3,1,1,-\frac{1}{2}\]
done
clear
C)
\[3,3,1,-\frac{1}{2}\]
done
clear
D)
\[3,2,-2,\frac{1}{2}\]
done
clear
View Answer play_arrow
question_answer 68) \[C{{a}^{2+}}\text{ }\]is isoelectronic with:
A)
\[N{{a}^{+}}\]
done
clear
B)
\[Ar\]
done
clear
C)
\[M{{g}^{2+}}\]
done
clear
D)
\[Kr\]
done
clear
View Answer play_arrow
question_answer 69) Which element is used for making a transistor?
A)
\[Sn\]
done
clear
B)
\[Sb\]
done
clear
C)
\[Si\]
done
clear
D)
\[Mg\]
done
clear
View Answer play_arrow
question_answer 70) The rate constant for a first order reaction whose half-life, is 480 s is:
A)
\[2.88\times {{10}^{-3}}{{s}^{-1}}\]
done
clear
B)
\[2.72\times {{10}^{-3}}{{s}^{-1}}\]
done
clear
C)
\[1.44\times {{10}^{-3}}{{s}^{-1}}\]
done
clear
D)
\[1.44{{s}^{-1}}\]
done
clear
View Answer play_arrow
question_answer 71) For the following homogeneous reaction, the unit of rate constant is: \[A+B\xrightarrow[{}]{k}C\]
A)
\[mo{{l}^{-1}}L\,{{s}^{-1}}\]
done
clear
B)
\[{{s}^{-1}}\]
done
clear
C)
\[s\]
done
clear
D)
\[{{s}^{-1}}mol\,{{L}^{-1}}\]
done
clear
View Answer play_arrow
question_answer 72) A sample of rock from moon contains equal number of atoms of uranium and lead. \[{{t}_{1/2}}\]for \[U=4.5\times {{10}^{9}}\]years. The age of the rock would be:
A)
\[2.25\times {{10}^{9}}\]years
done
clear
B)
\[13.5\times {{10}^{9}}\]years
done
clear
C)
\[9.0\times {{10}^{9}}\]years
done
clear
D)
\[4.5\times {{10}^{9}}\]years
done
clear
View Answer play_arrow
question_answer 73) The half-life period of a radioactive material is 15 min. What per cent of radioactivity of that material will remain after 45 minutes?
A)
17.5%
done
clear
B)
15%
done
clear
C)
12.5%
done
clear
D)
10%
done
clear
View Answer play_arrow
question_answer 74) In terms of energy 1 amu is equal to:
A)
100 J
done
clear
B)
931.1 kcal
done
clear
C)
931.1 MeV
done
clear
D)
\[{{10}^{7}}erg\]
done
clear
View Answer play_arrow
question_answer 75) A biological catalyst is:
A)
the\[{{N}_{2}}\]molecule
done
clear
B)
an enzyme
done
clear
C)
an amino acid
done
clear
D)
a carbohydrate.
done
clear
View Answer play_arrow
question_answer 76) The action of enzymes in living system is to:
A)
supply energy to tissues
done
clear
B)
create immunity
done
clear
C)
circulate oxygen
done
clear
D)
enhance the rate of biochemical reactions
done
clear
View Answer play_arrow
question_answer 77) Amongst the following, the molecule that is linear is:
A)
\[S{{O}_{2}}\]
done
clear
B)
\[C{{O}_{2}}\]
done
clear
C)
\[Cl{{O}_{2}}\]
done
clear
D)
\[N{{O}_{2}}\]
done
clear
View Answer play_arrow
question_answer 78) The hydrogen bond is. strongest in:
A)
\[FH...O\]
done
clear
B)
\[FH-.....F\]
done
clear
C)
\[SH...S\]
done
clear
D)
\[OH-....O\]
done
clear
View Answer play_arrow
question_answer 79) Dipole moment is shown by:
A)
cis-1, 2-dichloro ethene
done
clear
B)
trans-1, 2-dichloro ethene
done
clear
C)
trans-1, 2-dichloro-2 pentene
done
clear
D)
both [a] and [c]
done
clear
View Answer play_arrow
question_answer 80) The compound 1, 2-butadiene has:
A)
\[sp,s{{p}^{2}}\]and\[s{{p}^{3}}-\]hybridised carbon atoms
done
clear
B)
only\[s{{p}^{2}}-\]hybridised carbon atoms
done
clear
C)
only\[sp\]hybridised carbon atoms
done
clear
D)
Bbth\[sp\]and\[s{{p}^{2}}-\]hybridised carbon atoms
done
clear
View Answer play_arrow
question_answer 81) The\[C-H\]bond distance is the longest in
A)
\[{{C}_{2}}{{H}_{6}}\]
done
clear
B)
\[{{C}_{2}}{{H}_{2}}\]
done
clear
C)
\[{{C}_{2}}{{H}_{2}}B{{r}_{2}}\]
done
clear
D)
\[{{C}_{2}}{{H}_{4}}\]
done
clear
View Answer play_arrow
question_answer 82) Which one of the following solutions has the highest\[bp\]?
A)
\[0.1\text{ }M\,NaCl\]
done
clear
B)
\[0.1\,J\text{ }M\text{ }urea\]
done
clear
C)
\[0.1\,M\,BaC{{l}_{2}}\]
done
clear
D)
\[0.1\,M\text{ }glucose\]
done
clear
View Answer play_arrow
question_answer 83) The freezing point of 1% solution of lead nitrate in water will be:
A)
\[2{}^\circ C\]
done
clear
B)
\[1{}^\circ C\]
done
clear
C)
\[0{}^\circ C\]
done
clear
D)
below \[0{}^\circ C\]
done
clear
View Answer play_arrow
question_answer 84) The amount of urea to be dissolved in 500 cc of water\[(K=1.86{}^\circ C)\]to produce a depression of \[0.186{}^\circ C\]in the freezing point is:
A)
\[9g\]
done
clear
B)
\[6g\]
done
clear
C)
\[3g\]
done
clear
D)
\[0.3g\]
done
clear
View Answer play_arrow
question_answer 85) The solubility of\[AgI\]in\[NaI\]solution is less than that in pure water because:
A)
the temperature of the solution decreases
done
clear
B)
solubility product of\[AgI\]is less than that of \[NaI\]
done
clear
C)
of common ion effect
done
clear
D)
\[AgI\]forms complex with\[NaI\]
done
clear
View Answer play_arrow
question_answer 86) Which is chemically most active non-metal?
A)
\[S\]
done
clear
B)
\[{{O}_{2}}\]
done
clear
C)
\[{{F}_{2}}\]
done
clear
D)
\[{{N}_{2}}\]
done
clear
View Answer play_arrow
question_answer 87) Which one of the given transition metal ions is diamagnetic?
A)
\[C{{u}^{2+}}\]
done
clear
B)
\[C{{o}^{2+}}\]
done
clear
C)
\[C{{r}^{3+}}\]
done
clear
D)
\[Z{{n}^{2+}}\]
done
clear
View Answer play_arrow
question_answer 88) Which of the following isoelectronic species has the largest size?
A)
\[N{{a}^{+}}\]
done
clear
B)
\[{{O}^{2-}}\]
done
clear
C)
\[{{N}^{3-}}\]
done
clear
D)
\[Z{{n}^{2+}}\]
done
clear
View Answer play_arrow
question_answer 89) Molecular formula of Glaubers salt is:
A)
\[N{{a}_{2}}S{{O}_{4}}.10{{H}_{2}}O\]
done
clear
B)
\[FeS{{O}_{4}}.7{{H}_{2}}O\]
done
clear
C)
\[CuS{{O}_{4}}.5{{H}_{2}}O\]
done
clear
D)
\[MgS{{O}_{4}}.7{{H}_{2}}O\]
done
clear
View Answer play_arrow
question_answer 90) Cyanide process is used for the extraction of
A)
\[Au\]
done
clear
B)
\[Ag\]
done
clear
C)
\[Cu\]
done
clear
D)
both [a] and [b]
done
clear
View Answer play_arrow
question_answer 91) Which of the following metal is correctly matched with its ore?
A)
done
clear
B)
Metal Ore Silver Ilmenite
done
clear
C)
Metal Ore Magnesium Cassiterite
done
clear
D)
done
clear
View Answer play_arrow
question_answer 92) An example of a Lewis acid is:
A)
\[MgC{{l}_{2}}\]
done
clear
B)
\[SnC{{l}_{4}}\]
done
clear
C)
\[AlC{{l}_{3}}\]
done
clear
D)
\[NaCl\]
done
clear
View Answer play_arrow
question_answer 93) Which solution has pH equal to 10?
A)
\[{{10}^{-4}}\]M KOH
done
clear
B)
\[{{10}^{-10}}M\text{ }KOH\]
done
clear
C)
\[{{10}^{-10}}\]M \[\text{HCl}\]
done
clear
D)
\[{{10}^{-4}}M\text{ HCl}\]
done
clear
View Answer play_arrow
question_answer 94) Solubility product of\[BaC{{l}_{2}}\]is\[4\times {{10}^{-9}}\]. Its solubility would be:
A)
\[1\times {{10}^{-27}}\]
done
clear
B)
\[1\times {{10}^{-3}}\]
done
clear
C)
\[1\times {{10}^{-7}}\]
done
clear
D)
\[1\times {{10}^{-2}}\]
done
clear
View Answer play_arrow
question_answer 95) Molecular orbital theory was given by:
A)
Kossel
done
clear
B)
Mosley
done
clear
C)
Mulliken
done
clear
D)
Werner
done
clear
View Answer play_arrow
question_answer 96) In methane the bond angle is:
A)
\[180{}^\circ \]
done
clear
B)
\[90{}^\circ \]
done
clear
C)
\[109{}^\circ \]
done
clear
D)
\[120{}^\circ \]
done
clear
View Answer play_arrow
question_answer 97) Oxidation number of\[N\]in\[N{{H}_{3}}\](hydrazoic acid) is:
A)
\[-\frac{1}{3}\]
done
clear
B)
\[+3\]
done
clear
C)
\[0\]
done
clear
D)
\[-3\]
done
clear
View Answer play_arrow
question_answer 98) Phosphorus has the oxidation state of\[+3\]in:
A)
phosphorous acid
done
clear
B)
pyrophosphoric acid
done
clear
C)
orthophosphoric acid
done
clear
D)
metaphosphoric acid
done
clear
View Answer play_arrow
question_answer 99) Which of the following has highest pH?
A)
\[\frac{M}{4}KOH\]
done
clear
B)
\[\frac{M}{4}NaOH\]
done
clear
C)
\[\frac{M}{4}N{{H}_{4}}OH\]
done
clear
D)
\[\frac{M}{4}Ca{{(OH)}_{2}}\]
done
clear
View Answer play_arrow
question_answer 100) The heat of neutralisation of\[HCl\]by\[NaOH\]is \[-55.9\text{ }kJ/mol,\]the energy of dissociation of \[HCN\]is:
A)
43.8 kJ
done
clear
B)
\[-43.8\text{ }kJ\]
done
clear
C)
\[-68kJ\]
done
clear
D)
68kJ
done
clear
View Answer play_arrow
question_answer 101) Bronze is an alloy of:
A)
\[Pb+Sn+Zn\]
done
clear
B)
\[Cu+Sn\]
done
clear
C)
\[Pb+Zn\]
done
clear
D)
\[Cu+Zn\]
done
clear
View Answer play_arrow
question_answer 102) In photography we use:
A)
\[AgI\]
done
clear
B)
\[N{{H}_{3}}\]
done
clear
C)
\[AgCl\]
done
clear
D)
\[AgBr\]
done
clear
View Answer play_arrow
question_answer 103) The treatment of Cu with dilute\[HN{{O}_{3}}\]gives:
A)
\[{{N}_{2}}O\]
done
clear
B)
\[NO\]
done
clear
C)
\[N{{H}_{4}}\]
done
clear
D)
\[N{{O}_{2}}\]
done
clear
View Answer play_arrow
question_answer 104) A mixture of camphor and benzoic acid can be separated by:
A)
sublimation
done
clear
B)
extraction with a solvent
done
clear
C)
chemical method
done
clear
D)
fractional crystallization
done
clear
View Answer play_arrow
question_answer 105) The empirical formula of a compound is\[C{{H}_{2}}\]. One mole of this compound has a mass of 42 g. Its molecular formula is:
A)
\[{{C}_{3}}{{H}_{6}}\]
done
clear
B)
\[{{C}_{3}}{{H}_{8}}\]
done
clear
C)
\[C{{H}_{2}}\]
done
clear
D)
\[{{C}_{2}}{{H}_{2}}\]
done
clear
View Answer play_arrow
question_answer 106) The lightest particle is:
A)
\[\alpha -\]particle
done
clear
B)
positron
done
clear
C)
proton
done
clear
D)
neutron
done
clear
View Answer play_arrow
question_answer 107) In a gaseous reversible reaction \[{{N}_{2}}+{{O}_{2}}=2NO+heat\] If pressure is increased then the equilibrium constant would be:
A)
unchanged
done
clear
B)
increased
done
clear
C)
decreased
done
clear
D)
sometimes increased, sometimes decreased
done
clear
View Answer play_arrow
question_answer 108) Bohr model can explain:
A)
the solar spectrum
done
clear
B)
the spectrum of hydrogen molecule
done
clear
C)
spectrum of any atom or ion containing one electron only
done
clear
D)
the spectrum of hydrogen atom only
done
clear
View Answer play_arrow
question_answer 109) Gamma rays have:
A)
no mass and no charge
done
clear
B)
mass only
done
clear
C)
charge only
done
clear
D)
mass and charge both
done
clear
View Answer play_arrow
question_answer 110) Zeeman effect refers to the:
A)
splitting up of the lines in an emission spectrum in the presence of an external electrostatic field
done
clear
B)
random scattering of light by colloidal particles
done
clear
C)
splitting up of the lines in an emission spectrum in a magnetic field
done
clear
D)
emission of electrons from metals when light falls upon them
done
clear
View Answer play_arrow
question_answer 111) The extraction of which of the following metals involves bessemerisation?
A)
Fe
done
clear
B)
Ag
done
clear
C)
\[Al\]
done
clear
D)
Cu
done
clear
View Answer play_arrow
question_answer 112) The composition of the common glass is:
A)
\[N{{a}_{2}}O.CaO.6Si{{O}_{3}}\]
done
clear
B)
\[N{{a}_{2}}O.A{{l}_{2}}{{O}_{3}}.Si{{O}_{2}}\]
done
clear
C)
\[CaO.A{{l}_{2}}{{O}_{3}}.Si{{O}_{2}}\]
done
clear
D)
\[N{{a}_{2}}O.CaO.6Si{{O}_{2}}\]
done
clear
View Answer play_arrow
question_answer 113) \[{{H}_{2}}{{O}_{2}}\]is manufactured these days:
A)
by the action of\[{{H}_{2}}{{O}_{2}}\]on\[Ba{{O}_{2}}\]
done
clear
B)
by the action of\[{{H}_{2}}S{{O}_{4}}\]on \[N{{a}_{2}}{{O}_{2}}\]
done
clear
C)
by electrolysis of 50% \[{{H}_{2}}S{{O}_{4}}\]
done
clear
D)
by burning hydrogen in excess of oxygen
done
clear
View Answer play_arrow
question_answer 114) \[{{K}_{a}}\]of\[{{H}_{2}}{{O}_{2}}\]is of the order of:
A)
\[{{10}^{-12}}\]
done
clear
B)
\[{{10}^{-14}}\]
done
clear
C)
\[{{10}^{-16}}\]
done
clear
D)
\[{{10}^{-10}}\]
done
clear
View Answer play_arrow
question_answer 115) Which of the following oxy-acids of phosphorus is a reducing agent and monobasic?
A)
\[{{H}_{3}}P{{O}_{2}}\]
done
clear
B)
\[{{H}_{3}}P{{O}_{3}}\]
done
clear
C)
\[{{H}_{3}}P{{O}_{4}}\]
done
clear
D)
\[{{H}_{4}}{{P}_{2}}{{O}_{6}}\]
done
clear
View Answer play_arrow
question_answer 116) The hybrid state ofhsulphurinS03 molecule is:
A)
\[s{{p}^{3}}d\]
done
clear
B)
\[s{{p}^{3}}\]
done
clear
C)
\[s{{p}^{3}}{{d}^{2}}\]
done
clear
D)
\[s{{p}^{2}}\]
done
clear
View Answer play_arrow
question_answer 117) Solubility of iodine in water may be increased by adding:
A)
chloroform
done
clear
B)
potassium iodide
done
clear
C)
carbon disulphide
done
clear
D)
sodium thiosulphate
done
clear
View Answer play_arrow
question_answer 118) Noble gases are adsorbed by:
A)
anhydrous calcium chloride
done
clear
B)
ferric hydroxide
done
clear
C)
cone. \[{{H}_{2}}S{{O}_{4}}\]
done
clear
D)
activated coconut charcoal
done
clear
View Answer play_arrow
question_answer 119) Which one of the following is not present in RNA?
A)
Uracil
done
clear
B)
Thymine
done
clear
C)
Ribose
done
clear
D)
Phosphate
done
clear
View Answer play_arrow
question_answer 120) An organic compound has an empirical formula \[C{{H}_{2}}O,\]its vapour density is 45. The molecular formula of the compound is:
A)
\[C{{H}_{2}}O\]
done
clear
B)
\[{{C}_{2}}{{H}_{5}}O\]
done
clear
C)
\[{{C}_{2}}{{H}_{2}}O\]
done
clear
D)
\[{{C}_{3}}{{H}_{6}}{{O}_{3}}\]
done
clear
View Answer play_arrow
question_answer 121) Rat carries rat fleas which are the causal organism of:
A)
typhus fever
done
clear
B)
tick fever
done
clear
C)
bubonic plague
done
clear
D)
yellow fever
done
clear
View Answer play_arrow
question_answer 122) Which of the blood vessel is the largest in earthworm and possess valves?
A)
Dorsal blood vessel
done
clear
B)
Sub-neural blood vessel
done
clear
C)
Ventral blood vessel
done
clear
D)
Supra oesophageal blood vessels
done
clear
View Answer play_arrow
question_answer 123) Which of the two parts in cockroach are fundamentally similar in structure?
A)
Anal styees and labrum
done
clear
B)
Maxillae and legs
done
clear
C)
Mandibles and antennae
done
clear
D)
Wings and anal cerci
done
clear
View Answer play_arrow
question_answer 124) The integument of the frog is always kept moist because:
A)
it cannot move with dry skin
done
clear
B)
it performs cutaneous respiration
done
clear
C)
it cannot catch food with dry skin
done
clear
D)
it cannot jump better with moist skin
done
clear
View Answer play_arrow
question_answer 125) In Rattus rattvs, internally cerebral hemisphere are connected by:
A)
corpus striatum
done
clear
B)
corpus cardiacum
done
clear
C)
corpus callosum
done
clear
D)
corpus allatum
done
clear
View Answer play_arrow
question_answer 126) The function of typhlosole in earthworm is to:
A)
secrete digestive juice
done
clear
B)
slowdown rate of passage of food
done
clear
C)
increase absorptive area of intestinal epithelium
done
clear
D)
have no function
done
clear
View Answer play_arrow
question_answer 127) Blood of cockroach does not contains haemoglobin because:
A)
it respires through atmosphere
done
clear
B)
it respires through book-lungs
done
clear
C)
it does not respire i
done
clear
D)
it has some other means to cany oxygen direct into the tissue
done
clear
View Answer play_arrow
question_answer 128) In the pectoral girdle of frog there is a cup like cavity on each side called:
A)
acetabulum
done
clear
B)
sigmoid arc
done
clear
C)
glenoid cavity
done
clear
D)
thoracic cavity
done
clear
View Answer play_arrow
question_answer 129) Chloragogen cells are present in Pheretima. These are specialized for performing function of:
A)
nutrition
done
clear
B)
reproduction
done
clear
C)
excretion
done
clear
D)
respiration
done
clear
View Answer play_arrow
question_answer 130) Part of the right lung of rat which is not distinguishable due to passage of post caval through it is:
A)
anterior
done
clear
B)
middle
done
clear
C)
posterior
done
clear
D)
post caval
done
clear
View Answer play_arrow
question_answer 131) In earthworm, the circular muscles are highly develop in:
A)
body wall
done
clear
B)
typhlosole
done
clear
C)
gizzard
done
clear
D)
pharynx
done
clear
View Answer play_arrow
question_answer 132) The rate of absorption of ion is slow at high temperature are to:
A)
low transpiration rate
done
clear
B)
low photosynthetic rate
done
clear
C)
enzyme inactivation
done
clear
D)
low rate of cell elongation
done
clear
View Answer play_arrow
question_answer 133) Which is related to feminism?
A)
\[DNA\to RNA\to Protein\]
done
clear
B)
\[RNA\to DNA\to RNA\to Protein\]
done
clear
C)
\[\underset{\begin{smallmatrix} \uparrow \\ DNA \end{smallmatrix}}{\mathop{DNA}}\,\to RNA\to Protein\to \]
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 134) Number of meiotic divisions required to produce 100 microspore in angiosperm egg is:
A)
125
done
clear
B)
50
done
clear
C)
100
done
clear
D)
25
done
clear
View Answer play_arrow
question_answer 135) Number of meiotic division required to produce 100 macrospore in angiosperm/egg is:
A)
125
done
clear
B)
50
done
clear
C)
100
done
clear
D)
25
done
clear
View Answer play_arrow
question_answer 136) An egg cell has 5 pico gram of DNA in its nucleus. How much amount of DNA will be in this animal at the end of\[{{G}_{2}}\]phase mitosis?
A)
2.5 pico gram
done
clear
B)
5 pico gram
done
clear
C)
5gm
done
clear
D)
20 pico gram
done
clear
View Answer play_arrow
question_answer 137) The hypothesis that the early atmosphere, combined with an energy source, produced organic monomer was developed in the 1920s by:
A)
Miller and Urey
done
clear
B)
Oparin and Haldane
done
clear
C)
Fox and Pauling
done
clear
D)
Curiee and Pasteur
done
clear
View Answer play_arrow
question_answer 138) The determination of climatic condition of the geological past by study of the fossil is known as:
A)
Sedimentology
done
clear
B)
Palaeoecology
done
clear
C)
Palaeontology
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 139) Plants of the Galapagos islands resembles most closely to the plants of:
A)
Asia
done
clear
B)
Australia
done
clear
C)
North America
done
clear
D)
South America
done
clear
View Answer play_arrow
question_answer 140) Directional selection favour:
A)
one extreme form over the other extreme form and over intermediate form of a trait
done
clear
B)
both extremes forms of a trait
done
clear
C)
environmental differences
done
clear
D)
intermediate form of a trait
done
clear
View Answer play_arrow
question_answer 141) Sudden appearance of an ancestral character is known as:
A)
mutation
done
clear
B)
heredity
done
clear
C)
atavism
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 142) Resemblance between widely different groups due to a common adaptation is known as:
A)
retrogressive evolution
done
clear
B)
parallel evolution
done
clear
C)
convergent evolution
done
clear
D)
divergent evolution
done
clear
View Answer play_arrow
question_answer 143) The peak concentration ozone above surface of earth is at:
A)
23 km
done
clear
B)
13 km
done
clear
C)
15 km
done
clear
D)
30 km
done
clear
View Answer play_arrow
question_answer 144) One of the following plants found in India is an escape from the quarantine:
A)
coffee plant
done
clear
B)
Eichhornia
done
clear
C)
congress weed
done
clear
D)
cocoa
done
clear
View Answer play_arrow
question_answer 145) The percentage of soil volume occupied by pore space is called porosity of soil. It is minimum in:
A)
sandy soil
done
clear
B)
clay soil
done
clear
C)
loamy soil
done
clear
D)
silt
done
clear
View Answer play_arrow
question_answer 146) Biotic potential or potential natality means:
A)
natural increase of population under ideal/optimum concentration
done
clear
B)
potential of organism in a bioirie
done
clear
C)
number of organism in a biome
done
clear
D)
number of maximum number in a population
done
clear
View Answer play_arrow
question_answer 147) The storage of energy at consumer level is known as:
A)
net primary productivity
done
clear
B)
gross primary productivity
done
clear
C)
net productivity
done
clear
D)
secondary productivity
done
clear
View Answer play_arrow
question_answer 148) Detritus food chains law account for more energy flow than grazing food chain because:
A)
most organisms die without having being eaten
done
clear
B)
most organisms do not die
done
clear
C)
most organisms having being eaten
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 149) The sequence of communities showing a gradual change in composition is called:
A)
continuum
done
clear
B)
bio indicator
done
clear
C)
succession
done
clear
D)
pyramid of number
done
clear
View Answer play_arrow
question_answer 150) Association of animals belonging to different species, where both partness are benefitted, is called:
A)
commensalism
done
clear
B)
mutualism
done
clear
C)
colony
done
clear
D)
sympathy
done
clear
View Answer play_arrow
question_answer 151) The amount of living matter present in an ecosystem at a given time is called:
A)
biomass
done
clear
B)
standing crop
done
clear
C)
standing state
done
clear
D)
productivity
done
clear
View Answer play_arrow
question_answer 152) Tyioses an outgrowth from ray or axial parenchyma cell into the lumen of a vessel which partially or complete blocks the cavity are present in?
A)
Periderm
done
clear
B)
Heartwood
done
clear
C)
Sapwood
done
clear
D)
Secondary cortex
done
clear
View Answer play_arrow
question_answer 153) The chief function of sieve tube element is:
A)
to conduct minerals
done
clear
B)
to help the plant in forming wood
done
clear
C)
to transport water from roots to leaves
done
clear
D)
to translocate the organic material from source to sink
done
clear
View Answer play_arrow
question_answer 154) Chi-square test is:
A)
calculated on percentage
done
clear
B)
calculated on frequency
done
clear
C)
both [a] and [b]
done
clear
D)
calculated on original data
done
clear
View Answer play_arrow
question_answer 155) Which of the following processes/technique was used by Watson and Crick for giving double helical DNA model?
A)
Differential staining
done
clear
B)
X-ray diffraction
done
clear
C)
Auto radiography
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 156) The exchange of chromosomal parts between non-homologous chromosomes is known as:
A)
translocation
done
clear
B)
transcription
done
clear
C)
transduction
done
clear
D)
translation
done
clear
View Answer play_arrow
question_answer 157) Which of the following chromosomal mutation are most likely to take place when homologous chromosomes are undergoing synapsis?
A)
Inversion and translocation
done
clear
B)
Deletion and duplication
done
clear
C)
Inversion and deletion
done
clear
D)
Translocation and duplication
done
clear
View Answer play_arrow
question_answer 158) Polyploidy leads to rapid formation of new species because of:
A)
isolation
done
clear
B)
development of multiple sets of chromosomes
done
clear
C)
mutation
done
clear
D)
genetic recombination
done
clear
View Answer play_arrow
question_answer 159) Which of the following commonly known as Jumping genes?
A)
Transgenic genes
done
clear
B)
Recon
done
clear
C)
Plasmid
done
clear
D)
Transposon
done
clear
View Answer play_arrow
question_answer 160) Mendel observed that certain character did not assort independently. Later, scientist found that this is due to:
A)
linkage in traits
done
clear
B)
crossing over
done
clear
C)
both [a] and [b]
done
clear
D)
dominance of one trait over the other
done
clear
View Answer play_arrow
question_answer 161) Plants having similar genotypes produced by plant breeding are called:
A)
haploid
done
clear
B)
autoploid
done
clear
C)
clone
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 162) The recent technique which is used to separate DNA fragment is:
A)
Northern blotting
done
clear
B)
Easthern blotting
done
clear
C)
Southern blotting
done
clear
D)
Western blotting
done
clear
View Answer play_arrow
question_answer 163) A test cross (cross between\[{{F}_{1}}\]and parents both dominant and recessive) is performed to. distinguish between:
A)
two homozygous forms
done
clear
B)
two heterozygous form
done
clear
C)
a homozygous dominant and a heterozygous form
done
clear
D)
a homozygous recessive and the heterozygous form
done
clear
View Answer play_arrow
question_answer 164) Chromophil cells are found in:
A)
anterior pituitary
done
clear
B)
adrenal cortex
done
clear
C)
thymus
done
clear
D)
testes
done
clear
View Answer play_arrow
question_answer 165) Eyes are ............ in origin:
A)
ectoderm
done
clear
B)
endoderm
done
clear
C)
mesoderm
done
clear
D)
both a and c
done
clear
View Answer play_arrow
question_answer 166) In human beings placenta is formed by:
A)
amnion
done
clear
B)
chorion
done
clear
C)
allantois
done
clear
D)
all of these
done
clear
View Answer play_arrow
question_answer 167) Cytoplasm surrounding mitochondria present in the middle piece of sperm is:
A)
manchette
done
clear
B)
centroplasm
done
clear
C)
microplasm
done
clear
D)
acrosome
done
clear
View Answer play_arrow
question_answer 168) Neubenkern is a part of:
A)
human ovum
done
clear
B)
foetus
done
clear
C)
human sperm
done
clear
D)
Graafian follicle
done
clear
View Answer play_arrow
question_answer 169) Oxyntic cells are found in:
A)
kidneys and secrete renin
done
clear
B)
gastric epithelium and secrete\[HCl\]
done
clear
C)
gastric epithelium and secrete pepsin
done
clear
D)
islets of Langerhans and secrete glucagon
done
clear
View Answer play_arrow
question_answer 170) The epithelial cells lining the stomach of vertebrates is protected from damage by\[HCl\] because:
A)
\[HCl\]acid is too dilute
done
clear
B)
the epithelial cells are resistant to the action of\[HCl\]
done
clear
C)
\[HCl\]is neutralised by alkaline gastric juice
done
clear
D)
the epithelial cells are covered with a mucous secretion
done
clear
View Answer play_arrow
question_answer 171) Kattha is obtained from the heart wood of:
A)
Acacia arabica
done
clear
B)
Acacia fornesiana
done
clear
C)
Acacia auriculiformis
done
clear
D)
Acacia catechu
done
clear
View Answer play_arrow
question_answer 172) The timber yielding plant Shorea robusta belongs to the family:
A)
Fabaceae
done
clear
B)
Rubiaceae
done
clear
C)
Dipterocarpaceae
done
clear
D)
Verbenaceae
done
clear
View Answer play_arrow
question_answer 173) Cricket bat is made from the wood of:
A)
Pinus walichiana
done
clear
B)
Shorea robusta
done
clear
C)
Salix sp
done
clear
D)
Cedrus deodara
done
clear
View Answer play_arrow
question_answer 174) Cocaine an alkaloid is obtained from the leaves of:
A)
Erythroxylon coca
done
clear
B)
Ephedra sp.
done
clear
C)
Digitalis purpurea
done
clear
D)
Papaver somniferum
done
clear
View Answer play_arrow
question_answer 175) In which one of the following stages of cell division, the DNA content doubled?
A)
Interphase
done
clear
B)
Metaphase
done
clear
C)
Prophase
done
clear
D)
Anaphase
done
clear
View Answer play_arrow
question_answer 176) At which one of the following stages of meiosis, crossing over take place?
A)
Diplotene
done
clear
B)
Diakinesis
done
clear
C)
Leptotene
done
clear
D)
Pachytene
done
clear
View Answer play_arrow
question_answer 177) Out of the given substances which one is present in abundance in lysosomes?
A)
Isomerase
done
clear
B)
Heme
done
clear
C)
Acid phosphate
done
clear
D)
Cytochrome
done
clear
View Answer play_arrow
question_answer 178) Explant is defined as:
A)
excised part of plant used in tissue culture
done
clear
B)
plant part taken from outside India
done
clear
C)
tissue taken to space for exobiology
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 179) The statement omnis cellula e cellula of Rudolf Virchow has been taken from his book:
A)
Cellular pathology
done
clear
B)
Cellular potency
done
clear
C)
Micrographia
done
clear
D)
Scala naturae
done
clear
View Answer play_arrow
question_answer 180) Robert Hooke thought of the cells, he observed that:
A)
something similar to veins and arteries of animals for conducting fluid
done
clear
B)
smallest structural unit
done
clear
C)
smallest functional unit
done
clear
D)
unit of heredity
done
clear
View Answer play_arrow
question_answer 181) Manipulation of DNA in genetic engineering become easy due to the invention of:
A)
polymerase chain reaction
done
clear
B)
dot blot
done
clear
C)
enzyme linked immuno asorbant assay
done
clear
D)
eastern blotting
done
clear
View Answer play_arrow
question_answer 182) Gel electrophoresis is used to:
A)
measure the size of plasmids
done
clear
B)
find out whether viruses are infeteous
done
clear
C)
measure the enlarge and size of protein and DNA fragment
done
clear
D)
production of steroids
done
clear
View Answer play_arrow
question_answer 183) Which one of the following families has unilocular superior ovary?
A)
Asteraceae
done
clear
B)
Solanaceae
done
clear
C)
Papaveraceae
done
clear
D)
Cucurbitaceae
done
clear
View Answer play_arrow
question_answer 184) Identify the mismatch among the following pairs of trees and families:
A)
Psidium gujava - Myrtaceae
done
clear
B)
Swietenia mahogni - Meliaceae
done
clear
C)
Pistacia vraa--Anacardeaceae
done
clear
D)
Murraya koenigii - Meleaicae
done
clear
View Answer play_arrow
question_answer 185) Name the hormone which stimulates growth and development of breast is preparation for lactation?
A)
Oestrogen
done
clear
B)
Human placental lactogen
done
clear
C)
Progesterone
done
clear
D)
Chorionic gonadotrophin
done
clear
View Answer play_arrow
question_answer 186) Multi carpellary, apocarpous, gynoecium with superior ovary is characteristic feature of the family:
A)
Papaveraceae
done
clear
B)
Mystaceae
done
clear
C)
Ranunculaceae
done
clear
D)
Rutaceae
done
clear
View Answer play_arrow
question_answer 187) Free-central placentation is found in:
A)
Brassicaceae
done
clear
B)
Caryophyllaceae
done
clear
C)
Asteraceae
done
clear
D)
Malvaceae
done
clear
View Answer play_arrow
question_answer 188) Which of the following in moss capsule is haploid/gametophytic tissue?
A)
Anriulus and peristome
done
clear
B)
Calyptra and spore
done
clear
C)
Columella and theca
done
clear
D)
Operculum foot and seta
done
clear
View Answer play_arrow
question_answer 189) In capsule of moss, shock absorbers are:
A)
trabeculae
done
clear
B)
peristome teeth
done
clear
C)
seta
done
clear
D)
annulus
done
clear
View Answer play_arrow
question_answer 190) At the base of seta of capsule of moss, there is a haploid brownish growth called:
A)
calyptea
done
clear
B)
perigonium
done
clear
C)
vaginula
done
clear
D)
perichaetial
done
clear
View Answer play_arrow
question_answer 191) Human female possess\[44+XX\]chromosomes. The secondary oocyte shall have:
A)
\[44+XX\]
done
clear
B)
\[22+X\]
done
clear
C)
22
done
clear
D)
44
done
clear
View Answer play_arrow
question_answer 192) Who isolated rennet a cheese producing enzyme from calfs stomach?
A)
Flemming
done
clear
B)
Hensen
done
clear
C)
Waksman
done
clear
D)
Smith and Norths
done
clear
View Answer play_arrow
question_answer 193) Biopiracy is related to which of the following?
A)
Traditional knowledge
done
clear
B)
Biomolecules and regarding bioresources, genes, isolated from bioresources
done
clear
C)
Bioresources
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 194) A genetic code is a:
A)
sequence of nitrogenous bases on\[m-RNA\]
done
clear
B)
sequence of nitrogenous bases on\[c-RNA\]
done
clear
C)
sequence of nitrogenous bases on\[r-RNA\]
done
clear
D)
sequence of nitrogenous bases on DNA
done
clear
View Answer play_arrow
question_answer 195) Who among the following established the scientific basis of vaccination?
A)
Edward Jenner
done
clear
B)
George Kohler
done
clear
C)
Louis Pasteur
done
clear
D)
Von Behring
done
clear
View Answer play_arrow
question_answer 196) The main reason, why antibiotics could not solve all the problem of bacterial disease, is:
A)
enactivation of antibiotics of bacterial enzyme
done
clear
B)
decreased deficiency of the immune system
done
clear
C)
the development of mutant strains resistant to antibiotics
done
clear
D)
insensitivity of the individual following long exposure to antibiotics
done
clear
View Answer play_arrow
question_answer 197) Lactic acid formation is a two step anaerobic process. Both steps are carried at one stage by:
A)
Streptococcus
done
clear
B)
Rhizopus
done
clear
C)
Lactobacillus
done
clear
D)
Aspergillus
done
clear
View Answer play_arrow
question_answer 198) Which one of the following combination of microbes is responsible for the formation and flavour of yoghurt?
A)
Lactobacillus casei and Streptococcus thermophillus
done
clear
B)
Rhizobium meliloti and Azotobacter species
done
clear
C)
Edoboiquerilluers rubum and Sciencealla typhosa
done
clear
D)
Bacillus subtilis and Escherichia coli
done
clear
View Answer play_arrow
question_answer 199) Meiosisi in Spirogyra Ulothrix, Chlamydomonas and most of the algae/thallophytes is:
A)
sporic
done
clear
B)
zygotic
done
clear
C)
gametic
done
clear
D)
unequal
done
clear
View Answer play_arrow
question_answer 200) An industrial product obtained from a keep having haemostatic property (stopping of internal bleeding) is:
A)
alginic acid
done
clear
B)
agai
done
clear
C)
funeran
done
clear
D)
all of these
done
clear
View Answer play_arrow
question_answer 201) It is nice if everyone an have a own car.
A)
can has a own car
done
clear
B)
can has his own car
done
clear
C)
can have his own car
done
clear
D)
no correction required
done
clear
View Answer play_arrow
question_answer 202) In ancient time one believes in witches.
A)
people believes in witches
done
clear
B)
people believed in witches
done
clear
C)
one believed in witches
done
clear
D)
no correction required
done
clear
View Answer play_arrow
question_answer 203) He said that he has forgotten his umbrella at home.
A)
had forgotten
done
clear
B)
had left
done
clear
C)
has left
done
clear
D)
no correction required
done
clear
View Answer play_arrow
question_answer 204) I am not having seen her anywhere.
A)
have not seeing
done
clear
B)
have not being seen
done
clear
C)
have not seen
done
clear
D)
no correction required
done
clear
View Answer play_arrow
question_answer 205) Having been there before, he knew what to expect.
A)
have been
done
clear
B)
has been
done
clear
C)
had been
done
clear
D)
no correction required
done
clear
View Answer play_arrow
question_answer 206) Directions: Select the word which is most nearly the same in meaning to the word in capital letters. DEMISE:
A)
Result
done
clear
B)
Default
done
clear
C)
Death
done
clear
D)
Apprehension
done
clear
View Answer play_arrow
question_answer 207) Directions: Select the word which is most nearly the same in meaning to the word in capital letters. DISPARITY:
A)
Distaste
done
clear
B)
Dissimilarity
done
clear
C)
Criticism
done
clear
D)
Distinction
done
clear
View Answer play_arrow
question_answer 208) Directions: Select the word which is most nearly the same in meaning to the word in capital letters. FORUM:
A)
An assembly
done
clear
B)
Place of public discussion
done
clear
C)
An application
done
clear
D)
Recesses
done
clear
View Answer play_arrow
question_answer 209) Directions: Select the word which is most nearly the same in meaning to the word in capital letters. DISMANTLE:
A)
Take apart
done
clear
B)
Destroy
done
clear
C)
Shatter
done
clear
D)
Upset
done
clear
View Answer play_arrow
question_answer 210) Directions: Select the word which is most nearly the same in meaning to the word in capital letters. CURTAIL:
A)
Decorate
done
clear
B)
Celebrate
done
clear
C)
Cut short
done
clear
D)
Deprive
done
clear
View Answer play_arrow
question_answer 211) Directions: Choose the correct pair of words out of the alternatives given to fill in the blanks in each sentence. He went........here........five days ago.
A)
from, about
done
clear
B)
in, into
done
clear
C)
into, by
done
clear
D)
out, of
done
clear
View Answer play_arrow
question_answer 212) Directions: Choose the correct pair of words out of the alternatives given to fill in the blanks in each sentence. This college is affiliated........ Kanpur University ........ Uttar Pradesh.
A)
by, of
done
clear
B)
with, by
done
clear
C)
to, in
done
clear
D)
in, into
done
clear
View Answer play_arrow
question_answer 213) Directions: Choose the correct pair of words out of the alternatives given to fill in the blanks in each sentence. He belongs........an ancient family........ Hyderabad.
A)
upon, by
done
clear
B)
into, of
done
clear
C)
to, of
done
clear
D)
of, of
done
clear
View Answer play_arrow
question_answer 214) Directions: Choose the correct pair of words out of the alternatives given to fill in the blanks in each sentence. He insists ........ going ........ train.
A)
on, by
done
clear
B)
in, with
done
clear
C)
by, in
done
clear
D)
to, in
done
clear
View Answer play_arrow
question_answer 215) Directions: Choose the correct pair of words out of the alternatives given to fill in the blanks in each sentence. He called ........ me ........ Tuesday.
A)
on, by
done
clear
B)
upon, on
done
clear
C)
by, before
done
clear
D)
to, on
done
clear
View Answer play_arrow
question_answer 216) Directions: Choose the one suitable word for each of the sentences out of the four responses. Mania for stealing articles:
A)
Logamania
done
clear
B)
Kleptomania
done
clear
C)
Hymphomania
done
clear
D)
Lypomania
done
clear
View Answer play_arrow
question_answer 217) Directions: Choose the one suitable word for each of the sentences out of the four responses. A person living permanently in a certain place:
A)
Native
done
clear
B)
Resident
done
clear
C)
pomicile
done
clear
D)
Subject
done
clear
View Answer play_arrow
question_answer 218) Directions: Choose the one suitable word for each of the sentences out of the four responses. Witty clever retort:
A)
Repartee
done
clear
B)
Sarcasm
done
clear
C)
Platitude
done
clear
D)
Invective
done
clear
View Answer play_arrow
question_answer 219) Directions: Choose the one suitable word for each of the sentences out of the four responses. A person who actively opposes somebody of something:
A)
Antagonist
done
clear
B)
Activist
done
clear
C)
Incendiary
done
clear
D)
Terrorist
done
clear
View Answer play_arrow
question_answer 220) Directions: Choose the one suitable word for each of the sentences out of the four responses. A story in which ideas are symbolised as people:
A)
Fable
done
clear
B)
Allegory
done
clear
C)
Legend
done
clear
D)
Parable
done
clear
View Answer play_arrow
question_answer 221) Directions: In the questions, choose the group of words that shows the same relationship as given at the top of every question. Ink.-Pen : Paper
A)
Watch : Dial: Strap
done
clear
B)
Book : Paper : Words
done
clear
C)
Colour: Brush : Canvas
done
clear
D)
Farmer : Plough : Field
done
clear
View Answer play_arrow
question_answer 222) Directions: In the questions, choose the group of words that shows the same relationship as given at the top of every question. Mouth : Teeth : Chew
A)
Eyes : Lanses : See
done
clear
B)
Head : Brain : Think
done
clear
C)
Skin : Sweat: Touch
done
clear
D)
Feet: Dance : Toe
done
clear
View Answer play_arrow
question_answer 223) Directions: In the questions, choose the group of words that shows the same relationship as given at the top of every question. Music : Guitar : Performer
A)
Trick : Rope : Acrobat
done
clear
B)
Dance : Tune : Instrument
done
clear
C)
Food : Recipe : Cook
done
clear
D)
Patient: Medicine : Doctor
done
clear
View Answer play_arrow
question_answer 224) Directions: In the questions, choose the group of words that shows the same relationship as given at the top of every question. Correspondent: News : Newspaper
A)
Farmer: Crops : Food
done
clear
B)
Mason : Cement: Construction
done
clear
C)
Cloud : Water : Ponds
done
clear
D)
Road : Vehicle : Destination
done
clear
View Answer play_arrow
question_answer 225) Directions: In the questions, choose the group of words that shows the same relationship as given at the top of every question. Talk : Whishper : Shout
A)
Boredom : Tiredness : Rest
done
clear
B)
Touch : Hold : Embrace
done
clear
C)
See : Look : Watch
done
clear
D)
Create : Form: Make
done
clear
View Answer play_arrow
question_answer 226) Directions: In the-following questions, choose the option which shows common feature in the relationship given in each question. Water : Glass : Mirror
A)
They are without colour
done
clear
B)
Reflection can be seen in them
done
clear
C)
They are used in day to day life
done
clear
D)
They are transparent
done
clear
View Answer play_arrow
question_answer 227) Directions: In the-following questions, choose the option which shows common feature in the relationship given in each question. Blunder : Bungle : Jumble
A)
They are mistakes that cannot be remedied
done
clear
B)
The terms are used for unskilful handling
done
clear
C)
The terms are connected with fury
done
clear
D)
They mean sudden arrival
done
clear
View Answer play_arrow
question_answer 228) Directions: In the-following questions, choose the option which shows common feature in the relationship given in each question. Chlorine : Fluorine : Iodine
A)
These are names of inert gases
done
clear
B)
These are gases at room temperature
done
clear
C)
These are transition elements
done
clear
D)
These are halogens
done
clear
View Answer play_arrow
question_answer 229) Directions: In the-following questions, choose the option which shows common feature in the relationship given in each question. Fraction : Decimal: Ratio
A)
They are part of a number
done
clear
B)
The terms are connected with equations
done
clear
C)
The terms are connected with mathematics
done
clear
D)
They show, relationship between numbers
done
clear
View Answer play_arrow
question_answer 230) Directions: In the-following questions, choose the option which shows common feature in the relationship given in each question. Flood : Cyclone : Earthquakes
A)
They take toll of many lives
done
clear
B)
They move from one place to another
done
clear
C)
They are caused by low pressure
done
clear
D)
They are natural calamities
done
clear
View Answer play_arrow
question_answer 231) Which of the following is/are wrong regarding the Brahmos Missile-?
A)
It has been developed jointly by Russia
done
clear
B)
Its range is about 300 km.
done
clear
C)
It is cruise missile
done
clear
D)
All are correct
done
clear
View Answer play_arrow
question_answer 232) Nobel Prizes are distributed annually at:
A)
Manila
done
clear
B)
Stockholm
done
clear
C)
Geneva
done
clear
D)
New York
done
clear
View Answer play_arrow
question_answer 233) The Nobel Prize for Economics was instituted in 1967, but was first awarded in:
A)
1967
done
clear
B)
1988
done
clear
C)
1969
done
clear
D)
1970
done
clear
View Answer play_arrow
question_answer 234) To win a Grand-Slam in Tennis, a player has to win:
A)
Australian Open, Wimbeldon, French Open US Open
done
clear
B)
Wimbeldon, French Open, US Open
done
clear
C)
Wimbeldon, French Open, Pegas Czeck Open, US Open
done
clear
D)
Davis Cup, Wimbeldon, French Open
done
clear
View Answer play_arrow
question_answer 235) The international Court of Justice is located at:
A)
Geneva
done
clear
B)
Hague
done
clear
C)
Amsterdam
done
clear
D)
Vienna
done
clear
View Answer play_arrow
question_answer 236) The first talkie film of India was:
A)
Harishchandra
done
clear
B)
Kismat
done
clear
C)
AlamAra
done
clear
D)
Kangan
done
clear
View Answer play_arrow
question_answer 237) Folk painting Madhubani is famous in:
A)
West Bengal
done
clear
B)
Orissa
done
clear
C)
Bihar
done
clear
D)
Assam
done
clear
View Answer play_arrow
question_answer 238) Who among the following is the worlds first woman cosmonaut?
A)
Bachendri Pal
done
clear
B)
Junko Tabei
done
clear
C)
Valentina Tereshkova
done
clear
D)
Sally Ride
done
clear
View Answer play_arrow
question_answer 239) The first person to conduct heart transplantation in India is:
A)
Dr. P.K.K. Ayyangar
done
clear
B)
Dr. R. Valiathan
done
clear
C)
Dr. Venugopal
done
clear
D)
Dr. R. Kesavan Nair
done
clear
View Answer play_arrow
question_answer 240) The parliament of Japan is known as:
A)
Assembly
done
clear
B)
Key
done
clear
C)
Senate
done
clear
D)
Diet
done
clear
View Answer play_arrow