A) 1, 2-dinitrobenzene
B) 1, 3-dinitrobenzene
C) 1, 4-dinitrobenzene
D) 1, 2, 4-trinitrobenzene
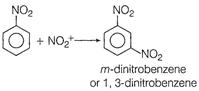
Correct Answer: B
Solution :
| [b] |
| Hence, now the mete-position becomes electron rich on which the electrophone (nitronium ion) attacks during nitration. |
| |
| |
 |
You need to login to perform this action.
You will be redirected in
3 sec
