| The number of unpaired electrons in me complex ion \[{{[Co{{F}_{6}}]}^{3-}}\] is: [AIPMT 2003] |
| (Atomic number \[Co=27\]) |
A) 4
B) zero
C) 2
D) 3
Correct Answer: A
Solution :
| [a] In complex ion \[{{[Co{{F}_{6}}]}^{3-}}\,,\,Co\] is present at +3 oxidation state |
| \[_{27}Co=1{{s}^{2}},2{{s}^{2}}\,2{{p}^{6}},\,3{{s}^{2}}3{{p}^{6}}\,3{{d}^{7}},4{{s}^{2}}\] |
| \[C{{o}^{3+}}=1{{s}^{2}},2{{s}^{2}}\,2{{p}^{6}},3{{s}^{2}}3{{p}^{6}}\,3{{d}^{6}}\] |
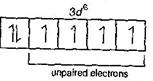 |
| Thus, the number of unpaired electrons in 3d sub-shell of \[{{[Co{{F}_{6}}]}^{3-}}\] is 4. |
You need to login to perform this action.
You will be redirected in
3 sec
