| For the following reactions :- [NEET - 2016] |
| \[~C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}Br\text{ }+\text{ }KOH\to \]\[C{{H}_{3}}CH=C{{H}_{2}}+KBr\text{ }+\text{ }{{H}_{2}}O\] |
 |
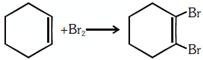 |
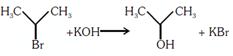
| Which of the following statements is correct? |
A) and are elimination reaction and is addition reaction
B) is elimination, is substitution and is addition reaction
C) is elimination, and are substitution reactions
D) is substitution, and are addition reaction
Correct Answer: B
Solution :
| [b] |
| \[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}Br+KOH\to C{{H}_{3}}CH\] \[=C{{H}_{2}}+KBr+\text{ }{{H}_{2}}O\] breaking of \[2\sigma \] bonds and formation of 1 \[1\pi \] bond so it is an example of elimination reaction. |
| |
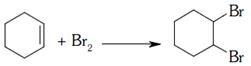 breaking of \[1\pi \] bond and formation of \[2\sigma \] bonds is addition reaction breaking of \[1\pi \] bond and formation of \[2\sigma \] bonds is addition reaction |
You need to login to perform this action.
You will be redirected in
3 sec
