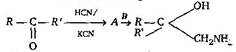question_answer 1) Aldol condensation will not take place in: [AIPMT 1999]
A)
\[HCHO\]
done
clear
B)
\[C{{H}_{3}}CHO\]
done
clear
C)
\[C{{H}_{3}}COC{{H}_{3}}\]
done
clear
D)
\[C{{H}_{3}}C{{H}_{2}}CHO\]
done
clear
View Answer play_arrow
question_answer 2) Which of the following is incorrect? [AIPMT 2001]
A)
\[FeC{{l}_{3}}\] is used in detection of phenol
done
clear
B)
Fehling solution is used in detection of glucose
done
clear
C)
Tollen's reagent is used in detection of unsaturation
done
clear
D)
\[NaHS{{O}_{3}}\] is used in detection of carbonyl compound
done
clear
View Answer play_arrow
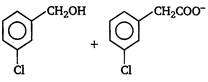
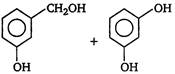
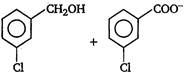
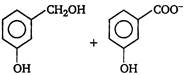
question_answer 3) Which one of the following is correct? [AIPMT 2001]
A)
On reduction of any aldehyde gives secondary alcohol
done
clear
B)
Reaction of vegetable oil with \[{{H}_{2}}S{{O}_{4}}\] gives glycerin
done
clear
C)
Alcoholic iodine with \[NaOH\] gives iodoform
done
clear
D)
Sucrose on reaction with \[NaCl\] gives invert sugar
done
clear
View Answer play_arrow
question_answer 4)
In the following reaction product 'P is: [AIPMT 2002] \[R-\underset{\begin{smallmatrix} |\,\,| \\ O \end{smallmatrix}}{\mathop{C}}\,-Cl\xrightarrow[Pd-BaS{{O}_{4}}]{{{H}_{2}}}P\]
A)
\[RC{{H}_{2}}OH\]
done
clear
B)
\[RCOOH\]
done
clear
C)
\[RCHO\]
done
clear
D)
\[RC{{H}_{3}}\]
done
clear
View Answer play_arrow
question_answer 5)
In this reaction: [AIPMT 2003] \[C{{H}_{3}}CHO+HCN\xrightarrow[{}]{{}}C{{H}_{3}}CH(OH)CN\]\[\xrightarrow{H.OH}C{{H}_{3}}CH(OH)COOH\] an asymmetric centre is generated. The acid obtained would be:
A)
50% D + 50% L-isomer
done
clear
B)
20% D + 80% L-isomer
done
clear
C)
D-isomer
done
clear
D)
L-isomer
done
clear
View Answer play_arrow
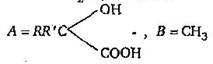
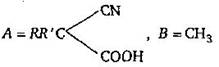
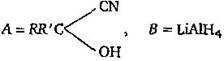
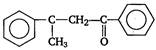
question_answer 6)
A and B in the following reactions are [AIPMT 2003]
A)
\[A=RR'C{{H}_{2}}CN,\,B=NaOH\]
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
question_answer 7) Which one of the following can be oxidised to the corresponding carbonyl compound? [AIPMT (S) 2004]
A)
2-hydroxy-propane
done
clear
B)
ortho-nitro -phenol
done
clear
C)
Phenol
done
clear
D)
2-methyl-2-hydroxy-propane
done
clear
View Answer play_arrow
question_answer 8)
The major organic product formed from the following reaction: [AIPMT (S) 2005]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
question_answer 9) A carbonyl compound reacts with hydrogen cyanide to form cyanohydrin which on hydrolysis forms a racemic mixture of \[\alpha \]-hydroxy acid. The carbonyl compound is: [AIPMT (S) 2006]
A)
acetaldehyde
done
clear
B)
acetone
done
clear
C)
diethyl ketone
done
clear
D)
formaldehyde
done
clear
View Answer play_arrow
question_answer 10) Nucleophilic addition reaction will be most favoured in: [AIPMT (S) 2006]
A)
\[C{{H}_{3}}-C{{H}_{2}}-C{{H}_{2}}\overset{\begin{smallmatrix} O \\ || \end{smallmatrix}}{\mathop{C}}\,-C{{H}_{3}}\]
done
clear
B)
\[{{(C{{H}_{3}})}_{2}}C=O\]
done
clear
C)
\[C{{H}_{3}}C{{H}_{2}}CHO\]
done
clear
D)
\[C{{H}_{3}}CHO\]
done
clear
View Answer play_arrow
question_answer 11) Reduction of aldehydes and ketones into hydrocarbons using zinc amalgam and cone. HCl is called: [AIPMT (S) 2007]
A)
Clemmensen reduction
done
clear
B)
Cope reduction
done
clear
C)
Dow reduction
done
clear
D)
Wolff-Kishner reduction
done
clear
View Answer play_arrow
question_answer 12) Which one of the following on treatment with 50% aqueous sodium hydroxide yields the corresponding alcohol and acid? [AIPMT (S) 2007]
A)
C6H5CH2CHO
done
clear
B)
C6H5CHO
done
clear
C)
CH3CH2CH2CHO
done
clear
D)
\[\begin{align} & \,\,\,\,\,\,\,\,\,\,\,\,\,\,\,O \\ & C{{H}_{3}}-\overset{||}{\mathop{C}}\,-C{{H}_{3}} \\ \end{align}\]
done
clear
View Answer play_arrow
question_answer 13) The product formed in aldol condensation is: [AIPMT (S) 2007]
A)
a beta-hydroxy acid
done
clear
B)
a beta-hydroxy aldehyde or a beta-hydroxy ketone
done
clear
C)
an alpha-hydroxy aldehyde or ketone
done
clear
D)
an alpha, beta unsaturated ester
done
clear
View Answer play_arrow
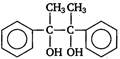
question_answer 14) Acetophenone when reacted with a base, \[{{C}_{2}}{{H}_{5}}ONa,\] yields a stable compound which has the structure [AIPMT (S) 2008]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
question_answer 15) A strong; base can abstract an \[\text{ }\!\!\alpha\!\!\text{ -}\]hydrogen from [AIPMT (S) 2008]
A)
alkene
done
clear
B)
amine
done
clear
C)
ketone
done
clear
D)
alkane
done
clear
View Answer play_arrow
question_answer 16) Which of the following reactions will not result in the formation of carbon-carbon bonds? [AIPMT (S) 2010]
A)
Reimer-Tiemann reaction
done
clear
B)
Cannizaro reaction
done
clear
C)
Wurtz reaction
done
clear
D)
Friedel-Crafts acylation
done
clear
View Answer play_arrow
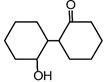
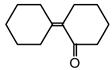
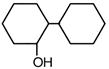
question_answer 17)
Following compounds are given [AIPMT (M) 2010] A.\[\text{C}{{\text{H}}_{\text{3}}}\text{C}{{\text{H}}_{\text{2}}}\text{OH}\] B.\[\text{C}{{\text{H}}_{\text{3}}}\text{COC}{{\text{H}}_{\text{3}}}\] C.\[\text{C}{{\text{H}}_{\text{3}}}-\underset{\begin{smallmatrix} \text{ }\!\!|\!\!\text{ } \\ \text{C}{{\text{H}}_{\text{3}}} \end{smallmatrix}}{\mathop{\text{C}}}\,\text{HOH}\] D\[\text{C}{{\text{H}}_{\text{3}}}\text{OH}\] Which of the above compound (s), on being warmed with iodine solution and NaOH, will give iodoform?
A)
A, C and D
done
clear
B)
Only B
done
clear
C)
A, B and C
done
clear
D)
A and B
done
clear
View Answer play_arrow
question_answer 18) Fructose reduces Tollen's reagent due to [AIPMT (M) 2010]
A)
asymmetric carbons
done
clear
B)
primary alcoholic group
done
clear
C)
secondary alcoholic group
done
clear
D)
enolisation of fructose followed by conversion to aldehyde by base
done
clear
View Answer play_arrow
question_answer 19) Clemmensen reduction of a ketone is carried out in the presence of which of the following? [AIPMT (S) 2011]
A)
Zn-Hg with HCl
done
clear
B)
LiAlH4
done
clear
C)
H2 and Pt as catalyst
done
clear
D)
Glycol with KOH
done
clear
View Answer play_arrow
question_answer 20) Which one is a nucleophilic substitution reaction among the following? [AIPMT (S) 2011]
A)
\[\text{RCHO+R }\!\!'\!\!\text{ MaX}\xrightarrow[{}]{{}}\text{R}-\underset{\begin{smallmatrix} \text{ }\!\!|\!\!\text{ } \\ \text{OH} \end{smallmatrix}}{\mathop{\text{CH}}}\,-\text{R}\]
done
clear
B)
\[\text{C}{{\text{H}}_{\text{3}}}-\text{C}{{\text{H}}_{\text{2}}}-\overset{\begin{smallmatrix} \text{C}{{\text{H}}_{\text{3}}} \\ \text{ }\!\!|\!\!\text{ } \end{smallmatrix}}{\mathop{\text{CH}}}\,-\text{C}{{\text{H}}_{\text{2}}}\text{Br}\,\text{+}\,\text{N}{{\text{H}}_{\text{3}}}\xrightarrow[{}]{{}}\]
done
clear
C)
\[\text{C}{{\text{H}}_{\text{3}}}-\text{C}{{\text{H}}_{\text{2}}}-\overset{\begin{smallmatrix} \text{C}{{\text{H}}_{\text{3}}} \\ \text{ }\!\!|\!\!\text{ } \end{smallmatrix}}{\mathop{\text{CH}}}\,-\text{C}{{\text{H}}_{\text{2}}}\text{N}{{\text{H}}_{2}}\]
done
clear
D)
\[\text{C}{{\text{H}}_{\text{3}}}\text{CHO}\,\text{+}\,\text{HCN}\xrightarrow[{}]{{}}\text{C}{{\text{H}}_{\text{3}}}\text{CH(OH)CN}\] \[\text{C}{{\text{H}}_{\text{3}}}-\text{CH}\,\text{=}\,\text{C}{{\text{H}}_{\text{2}}}+{{\text{H}}_{\text{2}}}\text{O}\xrightarrow[{}]{{{\text{H}}^{\text{+}}}}\] \[\text{C}{{\text{H}}_{\text{3}}}-\underset{\begin{smallmatrix} | \\ \text{OH} \end{smallmatrix}}{\mathop{\text{CH}\,}}\,-\text{C}{{\text{H}}_{\text{3}}}\]
done
clear
View Answer play_arrow
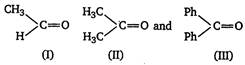
question_answer 21)
The order of reactivity of phenyl magnesium bromide (PhMgBr) with the following compounds [AIPMT (M) 2011]
A)
\[\text{III}\,\text{}\,\text{II}\,\text{}\,\text{I}\]
done
clear
B)
\[\text{II}\,\text{}\,\text{II}\,\text{}\,\text{III}\]
done
clear
C)
\[\text{I}\,\text{}\,\text{III}\,\text{}\,\text{II}\]
done
clear
D)
\[\text{I}\,\text{}\,\text{II}\,\text{}\,\text{III}\]
done
clear
View Answer play_arrow
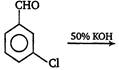
question_answer 22)
Predict the products in the given reaction, [AIPMT (S) 2012]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
question_answer 23) Acetone is treated with excess of ethanol in the presence of hydrochloric acid. The product obtained is [AIPMT (S) 2012]
A)
\[\text{C}{{\text{H}}_{\text{3}}}\text{C}{{\text{H}}_{\text{2}}}\text{C}{{\text{H}}_{\text{2}}}\text{-}\overset{\begin{smallmatrix} \text{O} \\ \text{ }\!\!|\!\!\text{ }\!\!|\!\!\text{ } \end{smallmatrix}}{\mathop{\text{C}}}\,\text{-C}{{\text{H}}_{\text{3}}}\]
done
clear
B)
\[\text{C}{{\text{H}}_{\text{3}}}\text{C}{{\text{H}}_{\text{2}}}\text{C}{{\text{H}}_{\text{2}}}\text{-}\overset{\begin{smallmatrix} \text{O} \\ \text{ }\!\!|\!\!\text{ }\!\!|\!\!\text{ } \end{smallmatrix}}{\mathop{\text{C}}}\,\text{-C}{{\text{H}}_{2}}\text{C}{{\text{H}}_{\text{2}}}\text{C}{{\text{H}}_{\text{3}}}\]
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
question_answer 24) \[\text{C}{{\text{H}}_{\text{3}}}\text{CHO}\]and \[{{\text{C}}_{\text{6}}}{{\text{H}}_{\text{5}}}\text{C}{{\text{H}}_{\text{2}}}\text{CHO}\] can be distinguished chemically by [AIPMT (S) 2012]
A)
Benedict test
done
clear
B)
Iodofrom test
done
clear
C)
Tollen's reagent test
done
clear
D)
Fehling solution test
done
clear
View Answer play_arrow
question_answer 25) An organic compound \[({{C}_{3}}{{H}_{9}}N)\] , when treated with nitrous acid, gave an alcohol and N2 gas was evolved. on warming with \[CHC{{l}_{3}}\] and caustic potash gave (C which on reduction gave iso-propylmethylamine. Predict the structure of.[AIPMT (M) 2012]
A)
done
clear
B)
\[C{{H}_{3}}C{{H}_{2}}-NH-C{{H}_{3}}\]
done
clear
C)
\[\text{C}{{\text{H}}_{\text{3}}}-\underset{\begin{smallmatrix} \text{ }\!\!|\!\!\text{ } \\ \text{C}{{\text{H}}_{\text{3}}} \end{smallmatrix}}{\mathop{\text{N}}}\,-\text{C}{{\text{H}}_{\text{3}}}\]
done
clear
D)
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}-N{{H}_{2}}\]
done
clear
View Answer play_arrow
question_answer 26)
Consider the reaction, [AIPMT (M) 2012] \[RCHO+N{{H}_{2}}N{{H}_{2}}\xrightarrow[{}]{{}}RCH=N-N{{H}_{2}}\]What sort of reaction is it?
A)
Electrophilic addition elimination reaction
done
clear
B)
Free radical addition elimination reaction
done
clear
C)
Electrophilic substitution elimination reaction
done
clear
D)
Nucleophilic addition elimination reaction
done
clear
View Answer play_arrow
question_answer 27) Which one is most reactive towards nucleophilic addition reaction? [AIPMT 2014]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
question_answer 28)
Treatment of cyclopentanone
A)
Cyclopentanonyl anion
done
clear
B)
Cyclopentanonyl cation
done
clear
C)
Cyclopentanonyl radical
done
clear
D)
Cyclopentanonyl biradical
done
clear
View Answer play_arrow
question_answer 29) An organic compound X having molecular formula \[{{C}_{5}}{{H}_{10}}O\] yields phenyl hydrazone and gives negative response to the iodoform test and Tollen's test. It produces n-pentane on reduction. X could be [NEET 2015]
A)
pentanal
done
clear
B)
2-pentanone
done
clear
C)
3-pentanone
done
clear
D)
n-amyl alcohol
done
clear
View Answer play_arrow
question_answer 30) Reaction of a carbonyl compound with one of the following reagents involves nucleophilic addition followed by the elimination of water. The reagents is [NEET 2015 (Re)]
A)
a Grignard reagent
done
clear
B)
hydrazine in presence of feebly acidic solution
done
clear
C)
hydrocyanic acid
done
clear
D)
sodium hydrogen sulphite
done
clear
View Answer play_arrow
question_answer 31) The correct statement regarding a carbonyl compound with a hydrogen atom on its alpha carbon, is :-[NEET - 2016]
A)
a carbonyl compound with a hydrogen atom on its alpha-carbon never equilibrates with its corresponding enol.
done
clear
B)
a carbonyl compound with a hydrgen atom on its alpha-carbon rapidly equilibrates with its corresponding enol and this process is known as aldehyde-ketone equilibration.
done
clear
C)
a carbonyl compound with a hydrogen atom on its alpha-carbon rapidly equilibrates with its corresponding enol and this process is known as carbonylation.
done
clear
D)
a carbonyl compound with a hydrogen atom on its alpha-carbon rapidly equilibrates with its corresponding enol and this process is known as keto-enol tautomerism.
done
clear
View Answer play_arrow
question_answer 32) The product formed by the reaction of an aldehyde with a primary amine is :- [NEET - 2016]
A)
Schiff base
done
clear
B)
Ketone
done
clear
C)
Carboxylic acid
done
clear
D)
Aromatic acid
done
clear
View Answer play_arrow
question_answer 33) Of the following, which is the product formed when cyclohexanone undergoes aldol condensation followed by heating? [NEET-2017]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
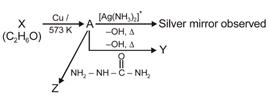
question_answer 34)
Consider the reactions: [NEET-2017]
A)
A-Ethanol, X-Acetaldehyde, Y-Butanone, Z-Hydrazone
done
clear
B)
A-Methoxymethane, X-Ethanoic acid, Y-Acetate ion, Z-hydrazine
done
clear
C)
A-Methoxymethane, X-Ethanol, Y-Ethanoic acid, Z-Semicarbazide
done
clear
D)
A-Ethanal, X-Ethanol, Y-But-2-enal, Z-Semicarbazone
done
clear
View Answer play_arrow
A)
II < I < III
done
clear
B)
II < III < I
done
clear
C)
III < I < II
done
clear
D)
III < II < I
done
clear
View Answer play_arrow
question_answer 36) Carboxylic acids have higher boiling points than aldehydes, ketones and even alcohols of comparable molecular mass. It is due to their [NEET - 2018]
A)
More extensive association of carboxylic acid via van der Waals force of attraction
done
clear
B)
Formation of carboxylate ion
done
clear
C)
Formation of intramolecular H-bonding
done
clear
D)
Formation of intermolecular H-bonding
done
clear
View Answer play_arrow
question_answer 37) Reaction between benzaldehyde and acetophenone in presence of dilute NaOH is known as [NEET 2020]
A)
Cannizzaro's reaction
done
clear
B)
Cross Cannizzaros reaction
done
clear
C)
Cross Aldol condensation
done
clear
D)
Aldol condensation
done
clear
View Answer play_arrow