question_answer 1) A circular thin disc of mass\[2kg\]has a diameter\[0.2m\]. Calculate its moment of inertia about an axis passing through the edge and perpendicular to the plane of the disc \[(in\text{ }kg-{{m}^{2}})\].
A)
\[0.01\]
done
clear
B)
\[0.03\]
done
clear
C)
\[0.02\]
done
clear
D)
\[3\]
done
clear
View Answer play_arrow
question_answer 2) In a step-up transformer the voltage in the primary is \[220V\] and die current is\[5A\]. The secondary voltage is found to be\[22000V\]. The current in the secondary (neglect losses) is
A)
\[5A\]
done
clear
B)
\[50A\]
done
clear
C)
\[500A\]
done
clear
D)
\[0.05A\]
done
clear
View Answer play_arrow
question_answer 3) An electron tube was sealed-off during manufacture at a pressure of \[1.2\times {{10}^{-7}}mm\] of mercury at\[{{27}^{o}}C\]. Its volume is \[100c{{m}^{3}}\]. The number of molecules that remain in the tube is
A)
\[2\times {{10}^{16}}\]
done
clear
B)
\[3\times {{10}^{15}}\]
done
clear
C)
\[3.86\times {{10}^{11}}\]
done
clear
D)
\[5\times {{10}^{11}}\]
done
clear
View Answer play_arrow
question_answer 4) The unit of specific conductivity is
A)
\[\Omega c{{m}^{-1}}\]
done
clear
B)
\[\Omega c{{m}^{-2}}\]
done
clear
C)
\[{{\Omega }^{-1}}cm\]
done
clear
D)
\[{{\Omega }^{-1}}c{{m}^{-1}}\]
done
clear
View Answer play_arrow
question_answer 5) If \[70cal\] of heat is required to raise the temperature of 2 mole of an ideal gas at constant pressure from\[{{30}^{o}}C\]to\[{{35}^{o}}C\], then the amount of heat required to raise the temperature of same gas through same range at constant volume is
A)
\[50cal\]
done
clear
B)
\[70cal\]
done
clear
C)
\[60cal\]
done
clear
D)
\[65cal\]
done
clear
View Answer play_arrow
question_answer 6) Power supplied to a particle of mass \[2kg\] varies with time as\[P=3{{t}^{2}}/2\]watt, where t is in second. If velocity of particle at \[t=0\] is\[v=0,\]the velocity of particle at\[t=2s\]will be
A)
\[1m{{s}^{-1}}\]
done
clear
B)
\[4m{{s}^{-1}}\]
done
clear
C)
\[2m{{s}^{-1}}\]
done
clear
D)
\[2\sqrt{2}m{{s}^{-1}}\]
done
clear
View Answer play_arrow
question_answer 7) A short bar magnet placed with its axis at\[{{30}^{o}}\]with a uniform external magnetic field of\[0.16\,T\]experiences a torque of magnitude\[0.032J\]. The magnetic moment of the bar magnet will be
A)
\[0.23\,\,J{{T}^{-1}}\]
done
clear
B)
\[0.40\,\,J{{T}^{-1}}\]
done
clear
C)
\[0.80\,\,J{{T}^{-1}}\]
done
clear
D)
zero
done
clear
View Answer play_arrow
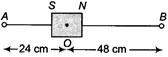
question_answer 8)
A bar magnet of length 3 cm has points\[A\]and\[B\]along its axis at distances of \[24cm\] and \[48cm\] on the opposite sides. Ratio of magnetic fields at these points will be
A)
\[8\]
done
clear
B)
\[\frac{1}{2\sqrt{2}}\]
done
clear
C)
\[3\]
done
clear
D)
\[4\]
done
clear
View Answer play_arrow
question_answer 9) A stone is dropped from a certain height which can reach the ground in 5s. If the stone is stopped after 3 s of its fall and then allowed to fall again, then the time taken by the stone to reach the ground for die remaining distance is
A)
\[2s\]
done
clear
B)
\[3s\]
done
clear
C)
\[4s\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 10) Two point-charges \[\pm 10\mu C\] are placed \[5.00mm\] apart, forming an electric dipole. Compute electric field at a point on the axis of the dipole \[15cm\] away from the centre on a line passing through the centre dipole.
A)
\[1.66\times {{10}^{5}}N{{C}^{-1}}\]
done
clear
B)
\[3.66\times {{10}^{5}}N{{C}^{-1}}\]
done
clear
C)
\[2.66\times {{10}^{-5}}N{{C}^{-1}}\]
done
clear
D)
\[2.66\times {{10}^{5}}N{{C}^{-1}}\]
done
clear
View Answer play_arrow
question_answer 11) In an\[AC\]circuit the potential difference V and current i are given respectively by \[V=100\sin (100t)volt\]and\[i=100\sin \left( 100t+\frac{\pi }{3} \right)mA\] The power dissipated in the circuit will be
A)
\[{{10}^{4}}W\]
done
clear
B)
\[10W\]
done
clear
C)
\[2.5W\]
done
clear
D)
\[5W\]
done
clear
View Answer play_arrow
question_answer 12) An inductor\[L,\,\,a\]capacitor of\[20\mu F\]and a resistor of \[10\Omega \] are connected in series with an AC source of frequency\[50Hz\]. If the current is in phase with the voltage, then the inductance of the inductor is
A)
\[2.00H\]
done
clear
B)
\[0.51H\]
done
clear
C)
\[1.5H\]
done
clear
D)
\[0.99H\]
done
clear
View Answer play_arrow
question_answer 13) A rifle shoots a bullet with a muzzle velocity of\[500m{{s}^{-1}}\]at a small target \[50m\] away. To hit the target the rifle must be aimed\[(take\]\[g=10m{{s}^{-2}})\]
A)
exactly at the target
done
clear
B)
\[10cm\] below the target
done
clear
C)
\[10cm\] above the target
done
clear
D)
\[5cm\] above the target
done
clear
View Answer play_arrow
question_answer 14) An \[\alpha -\]particle of mass \[m\] suffers one dimensional elastic collision with a nucleus of unknown mass. After the collision the \[\alpha -\]particle is scattered directly backwards losing\[75%\]of its kinetic energy. Then the mass of the nucleus is
A)
\[m\]
done
clear
B)
\[2m\]
done
clear
C)
\[3m\]
done
clear
D)
\[\frac{3}{2}m\]
done
clear
View Answer play_arrow
question_answer 15) Mark the correct one.
A)
Our eyes can distinguish between real and virtual image
done
clear
B)
Virtual image can also be taken on screen
done
clear
C)
If the incident rays are converging at a point, then the object is real
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 16) A body is suspended from a spring balance kept in a satellite. The reading of the balance is\[{{W}_{1}}\] when the satellite goes in an orbit of radius\[R\] and is\[{{W}_{2}}\]when it goes in an orbit of radius\[2R\]. Which of the following will be correct?
A)
\[{{W}_{1}}={{W}_{2}}\]
done
clear
B)
\[{{W}_{1}}<{{W}_{2}}\]
done
clear
C)
\[{{W}_{1}}>{{W}_{2}}\]
done
clear
D)
\[{{W}_{1}}\ne {{W}_{2}}\]
done
clear
View Answer play_arrow
question_answer 17) The force required to separate two glass plates of area\[{{10}^{-2}}{{m}^{2}}\]with a film of water\[0.05mm\] thick between them, is (surface tension of water is\[70\times {{10}^{-3}}N/m\])
A)
\[28N\]
done
clear
B)
\[14N\]
done
clear
C)
\[50N\]
done
clear
D)
\[38N\]
done
clear
View Answer play_arrow
question_answer 18) A body of mass m is placed on the earths surface. It is taken from earths surface to a height\[h=3R\]. The change in gravitational potential energy of the body is
A)
\[\frac{2}{3}mgR\]
done
clear
B)
\[\frac{3}{4}mgR\]
done
clear
C)
\[\frac{mgR}{2}\]
done
clear
D)
\[\frac{mgR}{4}\]
done
clear
View Answer play_arrow
question_answer 19) The only property possessed by ferromagnetic substance is
A)
hysteresis
done
clear
B)
susceptibility
done
clear
C)
directional property
done
clear
D)
attracting magnetic substances
done
clear
View Answer play_arrow
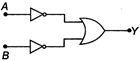
question_answer 20)
Which logic gate is represented by the following combination of logic gates?
A)
\[OR\]
done
clear
B)
\[NAND\]
done
clear
C)
\[AND\]
done
clear
D)
\[NOR\]
done
clear
View Answer play_arrow
question_answer 21) Minimum light intensity that can be perceived by normal human eye is about\[{{10}^{-10}}Wb\,\,{{m}^{-2}}\]. What is the minimum number of photons of wavelength 660 nm that must enter the pupil in\[1s\]for one to see the object? Area of cross-section of the pupil is\[{{10}^{-4}}{{m}^{2}}\].
A)
\[3.3\times {{10}^{2}}\]
done
clear
B)
\[3.3\times {{10}^{3}}\]
done
clear
C)
\[3.3\times {{10}^{4}}\]
done
clear
D)
\[3.3\times {{10}^{5}}\]
done
clear
View Answer play_arrow
question_answer 22) A thin convergent glass lens \[\mu =1.5\] has a power of\[+5.0D\]. When this lens is immersed in a liquid of refractive index \[{{\mu }_{1}}\] it acts as a diverging lens of focal length\[100cm\]. The value of\[{{\mu }_{l}}\]should be
A)
\[\frac{3}{2}\]
done
clear
B)
\[\frac{4}{3}\]
done
clear
C)
\[\frac{5}{3}\]
done
clear
D)
\[2\]
done
clear
View Answer play_arrow
question_answer 23) The phase difference between two points separated by\[0.8m\], in a wave of frequency\[120Hz\], is\[\frac{\pi }{2}\]. The velocity of wave is
A)
\[720m{{s}^{-1}}\]
done
clear
B)
\[384m{{s}^{-1}}\]
done
clear
C)
\[250m{{s}^{-1}}\]
done
clear
D)
\[1m{{s}^{-1}}\]
done
clear
View Answer play_arrow
question_answer 24) A ball of mass\[m\]and density\[\rho \]is immersed in a liquid of density\[\rho \]at a depth\[h\]and released. To what height will the ball jump up above the surface of liquid? (Neglect the resistance of water and air)
A)
\[h\]
done
clear
B)
\[\frac{h}{2}\]
done
clear
C)
\[3h\]
done
clear
D)
\[2h\]
done
clear
View Answer play_arrow
question_answer 25) Dimensions of resistance in an electrical circuit, in terms of dimensions of mass\[M\], of length\[L\], of time\[T\]and of current\[I\], would be
A)
\[[M{{L}^{2}}{{T}^{-3}}{{I}^{-1}}]\]
done
clear
B)
\[[M{{L}^{2}}{{T}^{-2}}]\]
done
clear
C)
\[[M{{L}^{2}}{{T}^{-1}}{{I}^{-1}}]\]
done
clear
D)
\[[M{{L}^{2}}{{T}^{-3}}{{I}^{-2}}]\]
done
clear
View Answer play_arrow
question_answer 26) The plane of a dip circle is set in the geographic meridian and the apparent dip is\[{{\delta }_{1}}\]. It is then set in a vertical plane perpendicular to the geographic meridian. The apparent dip angle is \[{{\delta }_{2}}\].The decimation\[\theta \]at the plane is
A)
\[\theta ={{\tan }^{-1}}(\tan {{\delta }_{1}}\tan {{\delta }_{2}})\]
done
clear
B)
\[\theta ={{\tan }^{-1}}(\tan {{\delta }_{1}}+\tan {{\delta }_{2}})\]
done
clear
C)
\[\theta ={{\tan }^{-1}}\left( \frac{\tan {{\delta }_{1}}}{\tan {{\delta }_{2}}} \right)\]
done
clear
D)
\[\theta ={{\tan }^{-1}}(\tan {{\delta }_{1}}-\tan {{\delta }_{2}})\]
done
clear
View Answer play_arrow
question_answer 27) A body starts from the origin and moves along the axis such that the velocity at any instant is given by\[v=4{{t}^{3}}-2t\]where t is in second and the velocity is in\[m/s\]. Find the acceleration of the particle when it is at a distance of \[2m\] from the origin.
A)
\[28m{{s}^{-2}}\]
done
clear
B)
\[22m{{s}^{-2}}\]
done
clear
C)
\[12m{{s}^{-2}}\]
done
clear
D)
\[10m{{s}^{-2}}\]
done
clear
View Answer play_arrow
question_answer 28) The resistance of a wire at\[{{20}^{o}}C\]is \[20\Omega \] and at \[{{500}^{o}}C\]is\[60\Omega \]. At which temperature its resistance will be\[25\Omega \]?
A)
\[{{50}^{o}}C\]
done
clear
B)
\[{{60}^{o}}C\]
done
clear
C)
\[{{70}^{o}}C\]
done
clear
D)
\[{{80}^{o}}C\]
done
clear
View Answer play_arrow
question_answer 29) A voltmeter of resistance\[1000\Omega \]gives full scale deflection when a current of \[100mA\] flows through it. The shunt resistance required across it to enable it to be used as an ammeter reading \[1A\] at full scale deflection is
A)
\[10000\Omega \]
done
clear
B)
\[9000\Omega \]
done
clear
C)
\[222\Omega \]
done
clear
D)
\[111\Omega \]
done
clear
View Answer play_arrow
question_answer 30) A particle having a mass of \[0.5g\] carries a charge of\[2.5\times {{10}^{-8}}C\]. The particle is given an initial horizontal velocity of\[6\times {{10}^{4}}m{{s}^{-1}}\]. The minimum magnitude of the magnetic field that is required so that, particle will keep moving in a horizontal direction is [take\[g=10m{{s}^{-2}}\]]
A)
\[8.46T\]
done
clear
B)
\[3.33T\]
done
clear
C)
\[8.64\times {{10}^{-2}}T\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 31) A wire in the form of a circular loop of radius\[r\]lies with its plane normal to a magnetic field\[B.\] If the wire is pulled to take a square shape in the same plane in time\[t\], the emf induced in the loop is given by
A)
\[\frac{\pi B{{r}^{2}}}{t}\left( 1-\frac{\pi }{10} \right)\]
done
clear
B)
\[\frac{\pi B{{r}^{2}}}{t}\left( 1-\frac{\pi }{8} \right)\]
done
clear
C)
\[\frac{\pi B{{r}^{2}}}{t}\left( 1-\frac{\pi }{6} \right)\]
done
clear
D)
\[\frac{\pi B{{r}^{2}}}{t}\left( 1-\frac{\pi }{4} \right)\]
done
clear
View Answer play_arrow
question_answer 32) 25 tuning forks are arranged in series in the order of decreasing frequency. Any two successive forks produce 3 beats/s. If the frequency of the first tuning fork is the octave of the last fork, then the frequency of the 21st fork is
A)
\[72Hz\]
done
clear
B)
\[288Hz\]
done
clear
C)
\[84Hz\]
done
clear
D)
\[87Hz\]
done
clear
View Answer play_arrow
question_answer 33) If the red light is replaced by blue light illuminating the object in a microscope the resolving power of the microscope
A)
decreases
done
clear
B)
increases
done
clear
C)
gets halved
done
clear
D)
remains unchanged
done
clear
View Answer play_arrow
question_answer 34) Five identical lamps grouped together produce a certain illumination on a screen kept 5 m from the lamps. If three of the lamps are switched-off, through what distance should the group of lamps be moved to obtain the same illumination on the screen? (Assume normal incidence)
A)
\[\sqrt{10}m\]towards the screen
done
clear
B)
\[(5+\sqrt{10})m\]towards the screen
done
clear
C)
\[(5-\sqrt{10})m\]towards the screen
done
clear
D)
\[(5-\sqrt{10})m\]away from the screen
done
clear
View Answer play_arrow
question_answer 35) A body of mass \[2kg\] makes an elastic collision with another body at rest and continues to move in the original direction with one-fourth its original speed. The mass of the second body which collides with the first body is
A)
\[2kg\]
done
clear
B)
\[1.2kg\]
done
clear
C)
\[3kg\]
done
clear
D)
\[1.5kg\]
done
clear
View Answer play_arrow
question_answer 36) In Youngs double slit experiment, the intensity of light coming from one of the slits is doubled the intensity from the other slit. The ratio of the maximum intensity to the minimum intensity in the interference fringe pattern observed is
A)
34
done
clear
B)
40
done
clear
C)
25
done
clear
D)
38
done
clear
View Answer play_arrow
question_answer 37) A rocket of initial mass \[1000kg\] ejects mass at a constant rate of \[10kg/s\] with constant relative speed of\[11m{{s}^{-1}}\]. Neglecting gravity, the acceleration of the rocket 1 min after the blast is
A)
\[11/40m{{s}^{-2}}\]
done
clear
B)
\[22/40m{{s}^{-2}}\]
done
clear
C)
\[1.1/40m{{s}^{-2}}\]
done
clear
D)
\[1m{{s}^{-2}}\]
done
clear
View Answer play_arrow
question_answer 38) An elastic ball is dropped from a height\[h\]and it rebounds many times from the floor. If the coefficient of restitution is\[e\], the time interval between the second and the third impacts, is
A)
\[ev/g\]
done
clear
B)
\[{{e}^{2}}v/g\]
done
clear
C)
\[{{e}^{2}}\sqrt{\left( \frac{8h}{g} \right)}\]
done
clear
D)
\[{{e}^{2}}\sqrt{\left( \frac{h}{g} \right)}\]
done
clear
View Answer play_arrow
question_answer 39) An adulterated sample of milk has a density of\[1032kg{{m}^{-3}}\], while pure milk has a density of\[1080kg{{m}^{-3}}\]. Then the volume of pure milk in a sample of \[10L\] of adulterated milk is
A)
\[0.5L\]
done
clear
B)
\[1.0L\]
done
clear
C)
\[2.0L\]
done
clear
D)
\[4.0L\]
done
clear
View Answer play_arrow
question_answer 40) Typical silt (hard mud) particle of radius\[20\mu m\]is on the top of lake water, its density is \[2000kg/{{m}^{3}}\] and the viscosity of lake water is\[1.0mPa\], density is\[1000\text{ }kg/{{m}^{3}}\]. If the lake is still (has no internal fluid motion) the terminal speed with which the particle hits the bottom of the lake is in\[mm/s\]
A)
\[0.67\]
done
clear
B)
\[0.77\]
done
clear
C)
\[0.87\]
done
clear
D)
\[0.97\]
done
clear
View Answer play_arrow
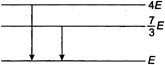
question_answer 41)
The following diagram indicates the energy levels of a certain atom when the system moves from \[4E\] level to\[E\]. A photon of wavelength\[\lambda \], is emitted. The wavelength of photon produced during its transition from\[\frac{7}{3}E\] level to\[E\]is\[{{\lambda }_{2}}\]. The ratio\[\frac{{{\lambda }_{1}}}{{{\lambda }_{2}}}\]will be
A)
\[\frac{9}{4}\]
done
clear
B)
\[\frac{4}{9}\]
done
clear
C)
\[\frac{3}{2}\]
done
clear
D)
\[\frac{7}{3}\]
done
clear
View Answer play_arrow
question_answer 42) A thin ring of mass \[2.7kg\] and radius \[8cm\] rotates about an axis through its centre and perpendicular to the plane of the ring at\[1.5rev/s\]. Calculate the kinetic energy of the ring.
A)
\[0.763\,\,J\]
done
clear
B)
\[0.345\,\,J\]
done
clear
C)
\[1.5\,\,J\]
done
clear
D)
zero
done
clear
View Answer play_arrow
question_answer 43) Two bodies at different temperatures are mixed in a calorimeter. Which of the following quantities remains conserved?
A)
Sum of the temperatures of the two bodies
done
clear
B)
Total heat of the two bodies
done
clear
C)
Total internal energy of the two bodies
done
clear
D)
Internal energy of each body
done
clear
View Answer play_arrow
question_answer 44) Mark the correct option.
A)
In electrostatics, there is no motion of charge at all in conductors bulk
done
clear
B)
In electrostatics, there is a motion of charged particle in conductors bulk
done
clear
C)
In electrostatics and current electricity there is a net motion of charged particles in the bulk of the material of the conductor
done
clear
D)
In electrostatics and current electricity there is no net motion of charged particles in die bulk of the material of the conductor
done
clear
View Answer play_arrow
question_answer 45) Binding energy of nucleus is defined as
A)
the energy released, when the nucleus has been separated into its constituent particles
done
clear
B)
the energy added, to separate the nucleus from its constituent particles
done
clear
C)
the energy added to form the nucleus from its constituent particles
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 46) The magnetic field in a region between the poles of an electromagnet is uniform at any time, but its magnitude is increasing at the rate of\[0.02\text{ }T/s\]. A conducting loop is placed in this region, whose plane is perpendicular to the direction of magnetic field. Calculate the emf induced and induced current in the loop. Take cross-section area of loop as \[120c{{m}^{2}}\] and resistance of loop as\[5\Omega \].
A)
\[0.48\,\,V,\,\,0.0096\,\,mA\]
done
clear
B)
\[0.48\,\,mV,\,\,0.0096\,\,mA\]
done
clear
C)
\[0.24\,\,mV,\,\,0.048\,\,mA\]
done
clear
D)
\[0.24\,\,mV,\,\,Zero\]
done
clear
View Answer play_arrow
question_answer 47) A car travels 15 km east at a constant speed of\[20km/h\], then continues east for \[20km\] at a constant speed of\[30km/h\]. What can be said about the magnitude of average velocity?
A)
\[{{v}_{av}}>25km/h\]
done
clear
B)
\[{{v}_{av}}<25km/h\]
done
clear
C)
\[{{v}_{av}}=25km/h\]
done
clear
D)
Information is insufficient
done
clear
View Answer play_arrow
question_answer 48) A body when projected vertically up covers a total distance 5, during its time of flight. If we neglect gravity then how much distance the particle will travel during the same time? Will it fall back?
A)
\[s,\]Yes
done
clear
B)
\[s,\]No
done
clear
C)
\[2s,\]Yes
done
clear
D)
\[2s,\]No
done
clear
View Answer play_arrow
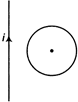
question_answer 49)
If \[i\] is increasing continuously, then find the direction of induced current in the loop.
A)
Anticlockwise
done
clear
B)
Clockwise
done
clear
C)
In any direction
done
clear
D)
No current will be induced
done
clear
View Answer play_arrow
question_answer 50) A vehicle of mass \[m\] is moving on a rough horizontal road with momentum\[p\]. If the coefficient of friction between the tyres and the road be\[\mu \], then the stopping distance is
A)
\[\frac{p}{2\mu mg}\]
done
clear
B)
\[\frac{{{p}^{2}}}{2\mu mg}\]
done
clear
C)
\[\frac{p}{2\mu {{m}^{2}}g}\]
done
clear
D)
\[\frac{{{p}^{2}}}{2\mu {{m}^{2}}g}\]
done
clear
View Answer play_arrow
question_answer 51) The structure of 4-methyl-2-pentene-1-ol is
A)
\[C{{H}_{3}}-C{{H}_{2}}-HC=CH-C{{H}_{2}}OH\]
done
clear
B)
\[C{{H}_{3}}-\overset{\begin{smallmatrix} C{{H}_{3}} \\ | \end{smallmatrix}}{\mathop{CH}}\,-HC=CH-C{{H}_{2}}OH\]
done
clear
C)
\[C{{H}_{3}}-\overset{\begin{smallmatrix} OH \\ | \end{smallmatrix}}{\mathop{CHCH}}\,=\overset{\begin{smallmatrix} C{{H}_{3}} \\ | \end{smallmatrix}}{\mathop{C}}\,-C{{H}_{3}}\]
done
clear
D)
\[C{{H}_{3}}-\overset{\begin{smallmatrix} C{{H}_{3}} \\ | \end{smallmatrix}}{\mathop{C}}\,=CH-C{{H}_{2}}C{{H}_{2}}OH\]
done
clear
View Answer play_arrow
question_answer 52) Soda ash is
A)
\[N{{a}_{2}}C{{O}_{3}}\cdot {{H}_{2}}O\]
done
clear
B)
\[NaOH\]
done
clear
C)
\[NaHC{{O}_{3}}\]
done
clear
D)
\[N{{a}_{2}}C{{O}_{3}}\]
done
clear
View Answer play_arrow
question_answer 53) Tritium is an isotope of
A)
hydrogen
done
clear
B)
titanium
done
clear
C)
tantalum
done
clear
D)
tellurium
done
clear
View Answer play_arrow
question_answer 54) When a nucleus emits an alpha particle, its atomic mass
A)
decreases by 2 units
done
clear
B)
increases by 2 units
done
clear
C)
decreases by 4 units
done
clear
D)
increases by 4 units
done
clear
View Answer play_arrow
question_answer 55) Which of the following is present in tenon?
A)
\[{{F}_{2}}\]
done
clear
B)
\[C{{l}_{2}}\]
done
clear
C)
\[{{I}_{2}}\]
done
clear
D)
\[B{{r}_{2}}\]
done
clear
View Answer play_arrow
question_answer 56) The chemical which is known as the king of chemicals is
A)
sodium hydroxide
done
clear
B)
sulphuric acid
done
clear
C)
calcium carbonate
done
clear
D)
ammonium sulphate
done
clear
View Answer play_arrow
question_answer 57) Carborundum is the name of
A)
\[A{{l}_{2}}{{O}_{3}}\]
done
clear
B)
\[SiC\]
done
clear
C)
\[Si{{O}_{2}}\]
done
clear
D)
\[HP{{O}_{3}}\]
done
clear
View Answer play_arrow
question_answer 58) The \[P-P-P\] bond angle in white phosphorus is
A)
\[{{120}^{o}}\]
done
clear
B)
\[{{109}^{o}},\,\,28\]
done
clear
C)
\[{{90}^{o}}\]
done
clear
D)
\[{{60}^{o}}\]
done
clear
View Answer play_arrow
question_answer 59) The fertilizer named Nitrolim is prepared by the use of
A)
\[CaO+{{N}_{2}}\]
done
clear
B)
\[CaC+{{N}_{2}}\]
done
clear
C)
\[Ca{{C}_{2}}+N\]
done
clear
D)
\[Ca{{C}_{2}}+{{N}_{2}}\]
done
clear
View Answer play_arrow
question_answer 60) Which one is the correct representation of\[NH_{4}^{+}\]ion?
A)
\[\left( \underset{\begin{smallmatrix} | \\ H \end{smallmatrix}}{\overset{\begin{smallmatrix} H \\ | \end{smallmatrix}}{\mathop{H-N\to H}}}\, \right)\]
done
clear
B)
\[\left( \underset{\begin{smallmatrix} | \\ H \end{smallmatrix}}{\overset{\begin{smallmatrix} H \\ | \end{smallmatrix}}{\mathop{H-N\leftarrow H}}}\, \right)\]
done
clear
C)
\[\left( \underset{\begin{smallmatrix} \uparrow \\ H \end{smallmatrix}}{\overset{\begin{smallmatrix} H \\ \downarrow \end{smallmatrix}}{\mathop{H\to N-H}}}\, \right)\]
done
clear
D)
\[\left( \underset{\begin{smallmatrix} \downarrow \\ H \end{smallmatrix}}{\overset{\begin{smallmatrix} H \\ \uparrow \end{smallmatrix}}{\mathop{H\leftarrow N\to H}}}\, \right)\]
done
clear
View Answer play_arrow
question_answer 61) What is the boiling point of a solution containing \[0.456g\] of camphor (molar mass\[=152\]) dissolved in \[31.4g\] of acetone\[(b.p={{56.30}^{o}}C)\], if the molecular elevation constant per \[100g\] of acetone is\[{{17.2}^{o}}C\]?
A)
\[{{56.46}^{o}}C\]
done
clear
B)
\[{{36.56}^{o}}C\]
done
clear
C)
\[{{56.14}^{o}}C\]
done
clear
D)
\[{{72.52}^{o}}C\]
done
clear
View Answer play_arrow
question_answer 62) What according to Fajans rules leads to increased ionic character?
A)
Large cation and small anion
done
clear
B)
Small cation and small charge on cation
done
clear
C)
Small cation and large charge on cation
done
clear
D)
Large cation and large charge on cation
done
clear
View Answer play_arrow
question_answer 63) The reaction given below is known as Dry ether\[{{C}_{6}}{{H}_{5}}Br+2Na+BrC{{H}_{3}}\xrightarrow{Dry\,\,ether}\]\[{{C}_{6}}{{H}_{5}}C{{H}_{3}}+2NaBr\]
A)
Wurtzs reaction
done
clear
B)
Wurtz Fittigs reaction
done
clear
C)
Friedel-Crafts reaction
done
clear
D)
Wurtz and Crafts reaction
done
clear
View Answer play_arrow
question_answer 64) Compound\[A\]on reduction gives\[B\], which on further reaction with\[CHC{{l}_{3}}\]and alcoholic\[KOH\] gives compound\[C\], which on further hydrolysis gives aniline. The compound\[A\]is
A)
nitrobenzene
done
clear
B)
methylamine
done
clear
C)
nitromethane
done
clear
D)
nitro sobenzene
done
clear
View Answer play_arrow
question_answer 65) DDT is formed from
A)
benzene and chlorobenzene
done
clear
B)
chloral and chlorobenzene
done
clear
C)
chloral and benzene
done
clear
D)
chlorobenzene and chlorine
done
clear
View Answer play_arrow
question_answer 66) Freon-114 is an organic substance used in refrigerators and air conditioners. It is chemically called 1, 2-dichloro tetrafluoroethane. Its correct formula is
A)
\[Cl-\underset{\begin{smallmatrix} | \\ Cl \end{smallmatrix}}{\overset{\begin{smallmatrix} F \\ | \end{smallmatrix}}{\mathop{C}}}\,-\underset{\begin{smallmatrix} | \\ Cl \end{smallmatrix}}{\overset{\begin{smallmatrix} F \\ | \end{smallmatrix}}{\mathop{C}}}\,-H\]
done
clear
B)
\[H-\underset{\begin{smallmatrix} | \\ Cl \end{smallmatrix}}{\overset{\begin{smallmatrix} F \\ | \end{smallmatrix}}{\mathop{C}}}\,-\underset{\begin{smallmatrix} | \\ Cl \end{smallmatrix}}{\overset{\begin{smallmatrix} F \\ | \end{smallmatrix}}{\mathop{C}}}\,-F\]
done
clear
C)
\[F-\underset{\begin{smallmatrix} | \\ F \end{smallmatrix}}{\overset{\begin{smallmatrix} Cl \\ | \end{smallmatrix}}{\mathop{C}}}\,-\underset{\begin{smallmatrix} | \\ F \end{smallmatrix}}{\overset{\begin{smallmatrix} F \\ | \end{smallmatrix}}{\mathop{C}}}\,-F\]
done
clear
D)
\[F-\underset{\begin{smallmatrix} | \\ Cl \end{smallmatrix}}{\overset{\begin{smallmatrix} F \\ | \end{smallmatrix}}{\mathop{C}}}\,-\underset{\begin{smallmatrix} | \\ F \end{smallmatrix}}{\overset{\begin{smallmatrix} Cl \\ | \end{smallmatrix}}{\mathop{C}}}\,-\underset{\begin{smallmatrix} | \\ F \end{smallmatrix}}{\overset{\begin{smallmatrix} F \\ | \end{smallmatrix}}{\mathop{C}}}\,-F\]
done
clear
View Answer play_arrow
question_answer 67) Which disaccharide is present in milk?
A)
Sucrose
done
clear
B)
Lactose
done
clear
C)
Maltose
done
clear
D)
Ketose
done
clear
View Answer play_arrow
question_answer 68) Emulsifier is an agent which
A)
accelerates the dispersion
done
clear
B)
stabilises the emulsion
done
clear
C)
homogenises the emulsion
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 69) For a chemical reaction\[xA\to yM\]rate law is\[r=k{{[A]}^{3}}\]. If the concentration of A is doubled, the reaction rate will be
A)
doubled
done
clear
B)
quadrupled
done
clear
C)
increased 4 times
done
clear
D)
increased 8 times
done
clear
View Answer play_arrow
question_answer 70) The formula representing correct relationship between\[{{K}_{c}}\]and\[{{K}_{p}}\]in gaseous equilibrium is
A)
\[\frac{{{K}_{p}}}{RT}={{({{K}_{c}})}^{\Delta {{n}_{g}}}}\]
done
clear
B)
\[\frac{{{K}_{c}}}{RT}={{({{K}_{p}})}^{\Delta {{n}_{g}}}}\]
done
clear
C)
\[{{K}_{p}}={{K}_{c}}{{(RT)}^{\Delta {{n}_{g}}}}\]
done
clear
D)
\[{{K}_{c}}={{K}_{p}}{{(RT)}^{\Delta {{n}_{g}}}}\]
done
clear
View Answer play_arrow
question_answer 71) The bond order of a molecule is given by
A)
the difference between the number of electrons in bonding and anti bonding orbitals
done
clear
B)
total number of electrons in bonding and anti bonding orbitals
done
clear
C)
twice the difference between the number of electrons in bonding and anti bonding orbitals
done
clear
D)
half the difference between number of electrons in bonding and anti bonding orbitals
done
clear
View Answer play_arrow
question_answer 72) Presence ofanitro group in a benzene ring
A)
renders the ring basic
done
clear
B)
deactivates the ring towards nucleophilic substitution
done
clear
C)
deactivates the ring towards electrophilic substitution
done
clear
D)
activates the ring towards electrophilic substitution
done
clear
View Answer play_arrow
question_answer 73) Doping of silicon\[(Si)\]with boron\[(B)\]leads to
A)
\[n-\]type semiconductor
done
clear
B)
\[p-\]type Semiconductor
done
clear
C)
metal
done
clear
D)
insulator
done
clear
View Answer play_arrow
question_answer 74) The standard emf of a cell, involving one electron change is found to be \[0.591V\] at\[{{25}^{o}}C.\] The equilibrium constant of the reaction is\[(F=96500\,\,C\,\,mo{{l}^{-1}})\]
A)
\[1.0\times {{10}^{1}}\]
done
clear
B)
\[1.0\times {{10}^{5}}\]
done
clear
C)
\[1.0\times {{10}^{10}}\]
done
clear
D)
\[1.0\times {{10}^{30}}\]
done
clear
View Answer play_arrow
question_answer 75) Term catalyst was given by
A)
Rutherford
done
clear
B)
Berzilius
done
clear
C)
Wohler
done
clear
D)
Kolbe
done
clear
View Answer play_arrow
question_answer 76) The following acids have been arranged in the order of decreasing acid strength. Identify the correct order \[ClOH(I)\,\,\,BrOH(II)\,\,\,IOH(III)\]
A)
\[I>II>III\]
done
clear
B)
\[II>I>III\]
done
clear
C)
\[III>II>I\]
done
clear
D)
\[I>III>II\]
done
clear
View Answer play_arrow
question_answer 77) \[Xe{{F}_{2}}\]on hydrolysis gives
A)
\[Xe{{O}_{3}}\]
done
clear
B)
\[XeO\]
done
clear
C)
\[Xe\]
done
clear
D)
\[Xe{{O}_{2}}\]
done
clear
View Answer play_arrow
question_answer 78) The properties of\[Zr\]and\[Hf\]are similar because
A)
both belong to \[d-\]block
done
clear
B)
both belong to same group of Periodic Table
done
clear
C)
both have similar radii
done
clear
D)
both have same number of electrons
done
clear
View Answer play_arrow
question_answer 79) The reaction of\[{{K}_{2}}C{{r}_{2}}{{O}_{7}}\]with\[NaCl\]and conc. \[{{H}_{2}}S{{O}_{4}}\]gives
A)
\[Cr{{O}_{2}}C{{l}_{2}}\]
done
clear
B)
\[C{{r}_{2}}{{O}_{3}}\]
done
clear
C)
\[CrC{{l}_{3}}\]
done
clear
D)
\[CrOC{{l}_{2}}\]
done
clear
View Answer play_arrow
question_answer 80) What is the expected structure of the organic product when ethylmagnesium bromide is treated with heavy water\[({{D}_{2}}O)\]?
A)
\[{{C}_{2}}{{H}_{5}}-{{C}_{2}}{{H}_{5}}\]
done
clear
B)
\[{{C}_{2}}{{H}_{5}}OD\]
done
clear
C)
\[{{C}_{2}}{{H}_{6}}\]
done
clear
D)
\[{{C}_{2}}{{H}_{5}}D\]
done
clear
View Answer play_arrow
question_answer 81) The reaction,\[{{(C{{H}_{3}})}_{3}}C-Br\xrightarrow{{{H}_{2}}O}{{(C{{H}_{3}})}_{3}}C-OH\]is
A)
elimination reaction
done
clear
B)
substitution reaction
done
clear
C)
free radical reaction
done
clear
D)
addition reaction
done
clear
View Answer play_arrow
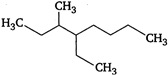
question_answer 82)
Name of the compound given below is
A)
4-ethyl-3-methyloctane
done
clear
B)
3-methyl-4-ethyloctane
done
clear
C)
2, 3-diethylheptane
done
clear
D)
5-ethyl-6-methyloctane
done
clear
View Answer play_arrow
question_answer 83) Which of the following is not true for\[{{[Co{{(F)}_{6}}]}^{3-}}\]?
A)
It is paramagnetic due to the presence of 4 unpaired electrons
done
clear
B)
It has coordination number of 6
done
clear
C)
It is outer orbital complex
done
clear
D)
It involves\[{{d}^{2}}s{{p}^{3}}\]hybridisation
done
clear
View Answer play_arrow
question_answer 84) When one mole of each of the following complex salts is treated with excess of \[AgN{{O}_{3}},\] which of them gives maximum amount of\[AgCl\]?
A)
\[[Co{{(N{{H}_{3}})}_{6}}]C{{l}_{3}}\]
done
clear
B)
\[[Co{{(N{{H}_{3}})}_{5}}Cl]C{{l}_{2}}\]
done
clear
C)
\[[Co{{(N{{H}_{3}})}_{4}}C{{l}_{2}}]Cl\]
done
clear
D)
\[[Co{{(N{{H}_{3}})}_{3}}C{{l}_{3}}]\]
done
clear
View Answer play_arrow
question_answer 85) Reimer-Tiemann reaction is useful for the preparation of
A)
benzaldehyde
done
clear
B)
salicylaldehyde
done
clear
C)
toluene
done
clear
D)
acetophenone
done
clear
View Answer play_arrow
question_answer 86) Which of the following compounds will produce the most stable carbonium ion when subjected to dehydration?
A)
\[C{{H}_{3}}-\underset{\begin{smallmatrix} | \\ C{{H}_{3}} \end{smallmatrix}}{\mathop{CH}}\,-C{{H}_{2}}OH\]
done
clear
B)
\[C{{H}_{3}}-\underset{\begin{smallmatrix} | \\ C{{H}_{3}} \end{smallmatrix}}{\overset{\begin{smallmatrix} C{{H}_{3}} \\ | \end{smallmatrix}}{\mathop{C}}}\,-OH\]
done
clear
C)
\[C{{H}_{3}}-\underset{\begin{smallmatrix} | \\ OH \end{smallmatrix}}{\mathop{CH}}\,-C{{H}_{2}}-C{{H}_{3}}\]
done
clear
D)
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}C{{H}_{2}}OH\]
done
clear
View Answer play_arrow
question_answer 87) In\[CC{{l}_{4}}\], the four valencies of carbon are directed towards the comers of a
A)
cube
done
clear
B)
hexagon
done
clear
C)
prism
done
clear
D)
tetrahedron
done
clear
View Answer play_arrow
question_answer 88) Fixation of nitrogen means
A)
manufacture of nitrogen from air
done
clear
B)
nitrogen cycle in nature
done
clear
C)
decomposition of nitrogen
done
clear
D)
conversion of atmospheric nitrogen into nitrogen compounds
done
clear
View Answer play_arrow
question_answer 89) Water is said to be permanently hard, when it contains
A)
chlorides and sulphates of\[Mg\]and\[Ca\]
done
clear
B)
bicarbonates of\[Mg\]and\[Ca\]
done
clear
C)
sulphates of\[Cu\]and\[Hg\]
done
clear
D)
carbonates and bicarbonates of\[Mg\]and\[Sr\]
done
clear
View Answer play_arrow
question_answer 90)
Given below is a statement and a reason I. Statement: Water has a higher boiling point than\[{{H}_{2}}S\]. II. Reason: At room temperature water is a liquid while \[{{H}_{2}}S\]is a gas.
Which of the following are true?
A)
Both I and II are correct however II does not give any proper explanation for statement I.
done
clear
B)
Both I and II are correct, II gives the proper explanation for statement I
done
clear
C)
Both I and II are incorrect
done
clear
D)
I is correct but II is incorrect
done
clear
View Answer play_arrow
question_answer 91) The oxidation number of\[C\]in\[C{{H}_{4}},\,\,C{{H}_{3}}Cl\],\[CHC{{l}_{3}},\,\,C{{H}_{2}}C{{l}_{2}}\]and\[CC{{l}_{4}}\]is respectively
A)
\[-4,\,\,-2,\,\,+2,\,\,0,\,\,+4\]
done
clear
B)
\[+2,\,\,4,\,\,0,\,\,-2,\,\,-4\]
done
clear
C)
\[+4,\,\,+0,\,\,-2,\,\,-2,\,\,+4\]
done
clear
D)
\[0,\,\,2,\,\,-2,\,\,4,\,\,4\]
done
clear
View Answer play_arrow
question_answer 92) \[Z{{n}^{2+}}(aq)+2{{e}^{-}}\xrightarrow{{}}Zn(s)\]. This is
A)
oxidation
done
clear
B)
reduction
done
clear
C)
redox reaction
done
clear
D)
hydrolysis
done
clear
View Answer play_arrow
question_answer 93) The cause of Brownian movement is
A)
heat changes in liquid state
done
clear
B)
convectional currents
done
clear
C)
the impact of molecules of the dispersion medium on the colloidal particles
done
clear
D)
attractive forces between the colloidal particles and molecules of dispersion medium
done
clear
View Answer play_arrow
question_answer 94) At\[700K\], the equilibrium constant\[{{K}_{p}}\]for the reaction\[2S{{O}_{3}}(g)2S{{O}_{2}}(g)+{{O}_{2}}(g)\]is\[1.80\times {{10}^{-3}}k\,\,Pa\]. What is the numerical value of\[{{K}_{c}}\]in moles per litre for this reaction at the same temperature?
A)
\[3.13\times {{10}^{-7}}\]
done
clear
B)
\[3.13\times {{10}^{7}}\]
done
clear
C)
\[3.13\times {{10}^{-10}}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 95) The rate of the chemical reaction doubles for an increase of \[10K\] in absolute temperature from\[298K\]. Calculate\[{{E}_{a}}\]
A)
\[52.988\,\,kJ\]
done
clear
B)
\[42.024\,\,kJ\]
done
clear
C)
\[52.898\,\,kJ\]
done
clear
D)
\[51.898\,\,kJ\]
done
clear
View Answer play_arrow
question_answer 96) Chloroprene is used in making
A)
synthetic rubber
done
clear
B)
plastic
done
clear
C)
petrol
done
clear
D)
All of these
done
clear
View Answer play_arrow
question_answer 97) What is the chemical formula of rust?
A)
\[F{{e}_{2}}{{O}_{3}}\cdot {{H}_{2}}O\]
done
clear
B)
\[F{{e}_{2}}{{O}_{3}}\cdot x{{H}_{2}}O\]
done
clear
C)
\[F{{e}_{2}}{{O}_{3}}\]
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 98) For tetrahedral coordination, the radius ratio \[({{r}^{+}}/{{r}^{-}})\]should be
A)
\[0.155-0.225\]
done
clear
B)
\[0.225-0.414\]
done
clear
C)
\[0.414-0.732\]
done
clear
D)
\[0.732-1\]
done
clear
View Answer play_arrow
question_answer 99) Thermodynamically the most stable form of carbon is
A)
diamond
done
clear
B)
graphite
done
clear
C)
peat
done
clear
D)
coal
done
clear
View Answer play_arrow
question_answer 100) In the following reaction, A and B, respectively are\[A\xrightarrow{HBr}{{C}_{2}}{{H}_{5}}Br\xrightarrow{B}A\]
A)
\[{{C}_{2}}{{H}_{4}},\,\,alc.KOH/\Delta \]
done
clear
B)
\[{{C}_{2}}{{H}_{5}}Cl,\,\,aq.KOH/\Delta \]
done
clear
C)
\[C{{H}_{3}}OH,\,\,aq.KOH/\Delta \]
done
clear
D)
\[{{C}_{2}}{{H}_{2}},\,\,PB{{r}_{3}}\]
done
clear
View Answer play_arrow
question_answer 101) Which of the following is the contractile protein of a muscle?
A)
Tubulin
done
clear
B)
Myosin
done
clear
C)
Tropo myosin
done
clear
D)
All of these
done
clear
View Answer play_arrow
question_answer 102) The formation of multi valents at meiosis in diploid organism is due to
A)
mono so my
done
clear
B)
inversion
done
clear
C)
deletion
done
clear
D)
reciprocal translocation
done
clear
View Answer play_arrow
question_answer 103) In mammals, histamine is secreted by
A)
fibroblasts
done
clear
B)
histocytes
done
clear
C)
lymphocytes
done
clear
D)
mast cells
done
clear
View Answer play_arrow
question_answer 104) Which of the microorganisms is used for production of citric acid in industries?
A)
Lactobacillus vulgaris
done
clear
B)
Pencillium citrinum
done
clear
C)
Aspergillus niger
done
clear
D)
Rhizopus nigricans
done
clear
View Answer play_arrow
question_answer 105) Yeast (Saccharomyces cerevisiae) is used for the industrial production of
A)
butanal
done
clear
B)
citric acid
done
clear
C)
tetracycline
done
clear
D)
ethanol
done
clear
View Answer play_arrow
question_answer 106) The layer of cells that secrete enamel of tooth is
A)
dentoblast
done
clear
B)
ameloblast
done
clear
C)
osteoblast
done
clear
D)
odontoblast
done
clear
View Answer play_arrow
question_answer 107) Which important greenhouse gas, other than carbon dioxide, is being produced from the agricultural fields?
A)
Arsine
done
clear
B)
Sulphur dioxide
done
clear
C)
Ammonia
done
clear
D)
Nitrous oxide
done
clear
View Answer play_arrow
question_answer 108) The role of double fertilisation in angiosperms is to produce
A)
endosperm
done
clear
B)
integuments
done
clear
C)
cotyledons
done
clear
D)
endocarp
done
clear
View Answer play_arrow
question_answer 109) The exchange of gases in the alveoli of the lungs takes place by
A)
osmosis
done
clear
B)
simple diffusion
done
clear
C)
passive transport
done
clear
D)
active transport
done
clear
View Answer play_arrow
question_answer 110) A plant hormone used for inducing morphogenesis in plant tissue culture is
A)
abscisic acid
done
clear
B)
gibberellins
done
clear
C)
cytokinins
done
clear
D)
ethylene
done
clear
View Answer play_arrow
question_answer 111) Which of the following is non-symbiotic biofertilizer?
A)
VAM
done
clear
B)
Azotobacter
done
clear
C)
Anabaena
done
clear
D)
Rhizobium
done
clear
View Answer play_arrow
question_answer 112) Carbon monoxide is a pollutant because it
A)
reacts with oxygen
done
clear
B)
inhibits glycolysis
done
clear
C)
reacts with haemoglobin
done
clear
D)
makes nervous system inactive
done
clear
View Answer play_arrow
question_answer 113) If there was no carbon dioxide in the earths atmosphere, the temperature of earths surface would be
A)
same as present
done
clear
B)
less than the present
done
clear
C)
higher than the present
done
clear
D)
dependent on the amount of oxygen in the atmosphere
done
clear
View Answer play_arrow
question_answer 114) Total number of bones in the hindlimb of a man is
A)
14
done
clear
B)
21
done
clear
C)
24
done
clear
D)
30
done
clear
View Answer play_arrow
question_answer 115) When a single gene influences more than one trait, it is called
A)
pleiotropy
done
clear
B)
epistasis
done
clear
C)
pseudo dominance
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 116) Calcitonin is a thyroid hormone which
A)
elevates potassium level in blood
done
clear
B)
lowers calcium level in blood
done
clear
C)
elevates calcium level in blood
done
clear
D)
has no effect on calcium
done
clear
View Answer play_arrow
question_answer 117) A condition of failure of kidney to form urine is called
A)
deamination
done
clear
B)
entropy
done
clear
C)
anuria
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 118) Ulothrix can be described as a
A)
filamentous alga with flagellated reproductive stages
done
clear
B)
non-motile colonial alga lacking zoospores
done
clear
C)
filamentous alga lacking flagellated reproductive stages
done
clear
D)
membranous alga producing zoospores
done
clear
View Answer play_arrow
question_answer 119) The long bones are hollow and connected by air passage. They are the characteristics of
A)
Aves
done
clear
B)
mammals
done
clear
C)
Reptilia
done
clear
D)
land vertebrates
done
clear
View Answer play_arrow
question_answer 120) Botulism caused by Clostridium botulinum affects the
A)
spleen
done
clear
B)
intestine
done
clear
C)
lymph glands
done
clear
D)
neuromuscular junction
done
clear
View Answer play_arrow
question_answer 121) Cellulose, the most important constituent of plant cell wall is made up of
A)
branched chain of glucose molecules linked by`\[\alpha ,\,\,1,\,\,6-\]glycosidic bond at the site of branching
done
clear
B)
unbranched chain of glucose molecules linked by \[\alpha ,\,\,1,\,\,4-\]glycosidic bond
done
clear
C)
branched chain of glucose molecules linked by \[\beta ,\,\,1,\,\,4-\]glycosidic bond in straight chain and \[\alpha ,\,\,1,\,\,6-\]glycosidic bond at the site of branching
done
clear
D)
unbranched chain of glucose molecules linked by \[\beta ,\,\,1,\,\,4-\]glycosidic bond
done
clear
View Answer play_arrow
question_answer 122) How many genome types are present in a typical green plants cell?
A)
Two
done
clear
B)
Three
done
clear
C)
More than five
done
clear
D)
More than ten
done
clear
View Answer play_arrow
question_answer 123) Bryophytes are dependent on water because
A)
archegonium has to remain filled with water for fertilization
done
clear
B)
water is essential for fertilisation for their homos porous nature
done
clear
C)
water is essential for their vegetative propagation
done
clear
D)
the sperms can easily reach upto egg in the archegonium
done
clear
View Answer play_arrow
question_answer 124) The main role of bacteria in the carbon cycle involves
A)
photosynthesis
done
clear
B)
assimilation of nitrogenous compounds
done
clear
C)
chemosynthesis
done
clear
D)
digestion or breakdown of organic compounds
done
clear
View Answer play_arrow
question_answer 125) Farmers have reported over 50% higher yields of rice by using which of the following biofertiliser?
A)
Mycorrhiza
done
clear
B)
Azolla pinnata
done
clear
C)
Cyanobacteria
done
clear
D)
Legume-Rhizobium symbiosis
done
clear
View Answer play_arrow
question_answer 126) Species restricted to a given area are called
A)
sibling
done
clear
B)
endemic
done
clear
C)
sympatric
done
clear
D)
allopatric
done
clear
View Answer play_arrow
question_answer 127) Solenocytes are the main excretory structures in
A)
annelids
done
clear
B)
molluscs
done
clear
C)
echinodermates
done
clear
D)
Platyhelminthes
done
clear
View Answer play_arrow
question_answer 128) Puccinia forms
A)
uredia and pycnia on barberry leaves
done
clear
B)
uredia and aecia on wheat leaves
done
clear
C)
uredia and telia on wheat leaves
done
clear
D)
uredia and aecia on barberry leaves
done
clear
View Answer play_arrow
question_answer 129) In a terrestial ecosystem such as forest, maximum energy is in which trophic level?
A)
\[{{T}_{1}}\]
done
clear
B)
\[{{T}_{2}}\]
done
clear
C)
\[{{T}_{3}}\]
done
clear
D)
\[{{T}_{4}}\]
done
clear
View Answer play_arrow
question_answer 130) The DNA of E. coli is
A)
single-stranded and linear
done
clear
B)
single-stranded and circular
done
clear
C)
double-stranded and linear
done
clear
D)
double-stranded and circular
done
clear
View Answer play_arrow
question_answer 131) In the five kingdom system of classification, which single kingdom out of the following can include blue-green algae, nitrogen-fixing bacteria and methangonic archaebacteria?
A)
Monera
done
clear
B)
Fungi
done
clear
C)
Plantae
done
clear
D)
Protista
done
clear
View Answer play_arrow
question_answer 132) A woman with two genes (one on each X-chromosome) for haemophilia and one gene for colour blindness on the X-chromosomes marries a normal man. How will the progeny be?
A)
All sons and daughters haemophilic and colourblind
done
clear
B)
Haemophilic and colourblind daughters
done
clear
C)
50% haemophilic colourblind sons and 50% haemophilic sons
done
clear
D)
50% haemophilic daughters and 50% colour blind daughters
done
clear
View Answer play_arrow
question_answer 133) The response of different organisms to the environmental rhythms of light and darkness is called
A)
phototaxis
done
clear
B)
phototropism
done
clear
C)
vernalisation
done
clear
D)
photo periodism
done
clear
View Answer play_arrow
question_answer 134) The functional unit of contractile system in striated muscle is
A)
cross bridge
done
clear
B)
myofibril
done
clear
C)
sarcomere
done
clear
D)
\[Z-\]band
done
clear
View Answer play_arrow
question_answer 135) The contraction of gall bladder is due to
A)
gastrin
done
clear
B)
secretin
done
clear
C)
cholecystokinin
done
clear
D)
enter gastrone
done
clear
View Answer play_arrow
question_answer 136) Typhoid fever is caused by
A)
Giardia
done
clear
B)
Salmonella
done
clear
C)
Shigella
done
clear
D)
Escherichia
done
clear
View Answer play_arrow
question_answer 137) Microtubule is involved in the
A)
cell division
done
clear
B)
DNA recognition
done
clear
C)
muscle contraction
done
clear
D)
membrane architecture
done
clear
View Answer play_arrow
question_answer 138) The diversity in the type of beaks of finches adapted to different feeding habits on the Galapagos islands, as observed by Darwin, provides evidence for
A)
origin of species by natural selection
done
clear
B)
intraspecific variations
done
clear
C)
intraspecific competition
done
clear
D)
interspecific competition
done
clear
View Answer play_arrow
question_answer 139) The most important component of the oral contraceptive pills is
A)
progesterone
done
clear
B)
growth hormone
done
clear
C)
thyroxine
done
clear
D)
luteinising hormone
done
clear
View Answer play_arrow
question_answer 140) Genes that are involved in turning on or off the transcription of a set structural genes are called
A)
polymorphic genes
done
clear
B)
operator genes
done
clear
C)
redundant genes
done
clear
D)
regulatory genes
done
clear
View Answer play_arrow
question_answer 141) Which one of the following statements about cytochrome\[{{P}_{450}}\]is wrong?
A)
It has an important role in metabolism
done
clear
B)
It contains iron
done
clear
C)
It is a coloured cell
done
clear
D)
It is an enzyme involved in oxidation reactions
done
clear
View Answer play_arrow
question_answer 142) Lactose is composed of
A)
glucose and fructose
done
clear
B)
glucose and glucose
done
clear
C)
glucose and galactose
done
clear
D)
fructose and galactose
done
clear
View Answer play_arrow
question_answer 143) Human immunodeficiency virus \[(HIV)\] has a protein coat and a genetic material which is
A)
single-stranded DNA
done
clear
B)
single-stranded RNA
done
clear
C)
double-stranded RNA
done
clear
D)
double-stranded DNA
done
clear
View Answer play_arrow
question_answer 144) The book Genera Plantarum was written by
A)
Hutchinson
done
clear
B)
Bessey
done
clear
C)
Engler and Pranti
done
clear
D)
Bentham and Hooker
done
clear
View Answer play_arrow
question_answer 145) In which of the following would you place the plants having vascular tissue lacking seeds?
A)
Gymnosperms
done
clear
B)
Algae
done
clear
C)
Pteridophytes
done
clear
D)
Bryophytes
done
clear
View Answer play_arrow
question_answer 146) Sympathetic nervous system induces
A)
heartbeat
done
clear
B)
secretion of digestive juice
done
clear
C)
Secretion of saliva
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 147) The middle piece of the sperm contains
A)
proteins
done
clear
B)
centriole
done
clear
C)
nucleus
done
clear
D)
mitochondria
done
clear
View Answer play_arrow
question_answer 148) The respiratory centres which control inspiration and expiration are located in
A)
spinal cord
done
clear
B)
diencephalon
done
clear
C)
cerebellum
done
clear
D)
medulla oblongata
done
clear
View Answer play_arrow
question_answer 149) The proteins are synthesised at
A)
ribosomes
done
clear
B)
mitochondria
done
clear
C)
centrosomes
done
clear
D)
Golgi bodies
done
clear
View Answer play_arrow
question_answer 150) The term aquaculture means
A)
aspergillosis
done
clear
B)
inland fisheries
done
clear
C)
marine fisheries
done
clear
D)
Both (b) and (c)
done
clear
View Answer play_arrow
question_answer 151) Which part of the world has high density of organisms?
A)
Deciduous forests
done
clear
B)
Grasslands
done
clear
C)
Savannas
done
clear
D)
Tropical rainforests
done
clear
View Answer play_arrow
question_answer 152) Tablets to prevent contraception contain
A)
progesterone
done
clear
B)
FSH
done
clear
C)
LH
done
clear
D)
Both (b) and (c)
done
clear
View Answer play_arrow
question_answer 153) A system of classification in which a large number of traits are considered is
A)
phylogenetic system
done
clear
B)
artificial system
done
clear
C)
natural system
done
clear
D)
synthetic system
done
clear
View Answer play_arrow
question_answer 154) Hybridisation between \[Tt\times tt\] gives rise to the progeny of ratio
A)
\[1:1\]
done
clear
B)
\[1:2:1\]
done
clear
C)
\[1:2\]
done
clear
D)
\[4:1\]
done
clear
View Answer play_arrow
question_answer 155) Active immunity means
A)
resistance developed after disease
done
clear
B)
increasing quantity of blood
done
clear
C)
resistance developed before disease
done
clear
D)
increasing rate of heart-beat
done
clear
View Answer play_arrow
question_answer 156) The canal system is a characteristic feature of
A)
echinoderms
done
clear
B)
helminthes
done
clear
C)
coelenterates
done
clear
D)
sponges
done
clear
View Answer play_arrow
question_answer 157) Hybridoma cells are
A)
product of spore formation in bacteria
done
clear
B)
hybrid cells resulting from myeloma cells
done
clear
C)
nervous cells of frog
done
clear
D)
only cells having oncogenes
done
clear
View Answer play_arrow
question_answer 158) In DNA when AGCT occurs, their association is as per which of the following pair?
A)
\[AC-GT\]
done
clear
B)
\[AG-CT\]
done
clear
C)
\[AT-GC\]
done
clear
D)
All of these
done
clear
View Answer play_arrow
question_answer 159) After ovulation, Graafian follicle regresses into
A)
corpus luteum
done
clear
B)
corpus callosum
done
clear
C)
corpus albicans
done
clear
D)
corpus artesia
done
clear
View Answer play_arrow
question_answer 160) Which cranial nerve has the highest number of branches?
A)
Facial nerve
done
clear
B)
Trigeminal
done
clear
C)
Vagus nerve
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 161) Carbon dioxide acceptor in \[{{C}_{3}}-\]plants is
A)
\[PGA\]
done
clear
B)
\[RuDP\]
done
clear
C)
\[PEP\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 162) The problem due to Rh~ factor arises when the blood of two (\[R{{h}^{+}}\]and\[R{{h}^{-}}\]) mix-up
A)
during pregnancy
done
clear
B)
through transfusion
done
clear
C)
in a test tube
done
clear
D)
Both (a) and (b)
done
clear
View Answer play_arrow
question_answer 163) The Pneumococcus experiment proves that
A)
DNA is the genetic material
done
clear
B)
RNA sometime controls the production of DNA and proteins
done
clear
C)
bacteria undergo binary fission
done
clear
D)
bacteria do not reproduce sexually
done
clear
View Answer play_arrow
question_answer 164) Haemophilic man marries a normal woman. Their offsprings will be
A)
all boys haemophilic
done
clear
B)
all normal
done
clear
C)
all girls haemophilic
done
clear
D)
all haemophilic
done
clear
View Answer play_arrow
question_answer 165) DNA is mainly found in
A)
nucleus only
done
clear
B)
cytoplasm only
done
clear
C)
nucleus and cytoplasm
done
clear
D)
nucleolus
done
clear
View Answer play_arrow
question_answer 166) Which of the following is mismatched?
A)
Vitamin - K - Beri-beri
done
clear
B)
Vitamin - D - Rickets
done
clear
C)
Vitamin - C - Scurvy
done
clear
D)
Vitamin - A - Xerophthalmia
done
clear
View Answer play_arrow
question_answer 167) The maximum biomagnification would be in which of the following case of aquatic ecosystem?
A)
Fishes
done
clear
B)
Phytoplanktons
done
clear
C)
Birds
done
clear
D)
Zooplanktons
done
clear
View Answer play_arrow
question_answer 168) Some of the enzymes, which are associated in converting fats into carbohydrates are present in
A)
microsomes
done
clear
B)
liposomes
done
clear
C)
Golgi bodies
done
clear
D)
glyoxysomes
done
clear
View Answer play_arrow
question_answer 169) The antherozoids of Funaria are
A)
mono ciliated
done
clear
B)
biciliated
done
clear
C)
multiciliated
done
clear
D)
aciliated
done
clear
View Answer play_arrow
question_answer 170) Haemoglobin is a type of
A)
carbohydrate
done
clear
B)
vitamin
done
clear
C)
skin pigment
done
clear
D)
respiratory pigment
done
clear
View Answer play_arrow
question_answer 171) Dichotomous branching is found in
A)
Marchantia
done
clear
B)
fern
done
clear
C)
liverworts
done
clear
D)
Funaria
done
clear
View Answer play_arrow
question_answer 172) Photosynthetic bacteria have pigments in
A)
chromoplasts
done
clear
B)
leucoplasts
done
clear
C)
chloroplasts
done
clear
D)
chromatophore
done
clear
View Answer play_arrow
question_answer 173) Which is (he principal cation in the plasma of the blood?
A)
Magnesium
done
clear
B)
Sodium
done
clear
C)
Potassium
done
clear
D)
Calcium
done
clear
View Answer play_arrow
question_answer 174) Glucose is carried from digestive tract to liver by
A)
pulmonary vein
done
clear
B)
hepatic portal vein
done
clear
C)
hepatic artery
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 175) Edible part of litchi is
A)
pericarp
done
clear
B)
fleshy aril
done
clear
C)
mesocarp
done
clear
D)
endosperm
done
clear
View Answer play_arrow
question_answer 176) The joint between atlas and axis is called
A)
pivot joint
done
clear
B)
hinge joint
done
clear
C)
saddle joint
done
clear
D)
angular joint
done
clear
View Answer play_arrow
question_answer 177) Cholecystokinin and duocrinin are secreted by
A)
adrenal cortex
done
clear
B)
thyroid gland
done
clear
C)
pancreas
done
clear
D)
intestine
done
clear
View Answer play_arrow
question_answer 178) Net gain of ATP molecules during aerobic respiration is
A)
36 molecules
done
clear
B)
60 molecules
done
clear
C)
38 molecules
done
clear
D)
48 molecules
done
clear
View Answer play_arrow
question_answer 179) The type of placentation in which ovary is syncarpous, unilocular and ovules on sutures is called
A)
marginal placentation
done
clear
B)
parietal placentation
done
clear
C)
axile placentation
done
clear
D)
superficial placentation
done
clear
View Answer play_arrow
question_answer 180) The blood group with antibody\[-a\] and antibody\[-b\]is
A)
B
done
clear
B)
A
done
clear
C)
O
done
clear
D)
AB
done
clear
View Answer play_arrow
question_answer 181) The endosperm of gymnosperms is
A)
polyploid
done
clear
B)
haploid
done
clear
C)
triploid
done
clear
D)
diploid
done
clear
View Answer play_arrow
question_answer 182) Rate of heartbeat is determined by
A)
Purkinje fibres
done
clear
B)
papillary muscles
done
clear
C)
SA-node
done
clear
D)
AV-node
done
clear
View Answer play_arrow
question_answer 183) Columella is a specialised structure found in the sporangium of
A)
Spirogyra
done
clear
B)
Rhizopus
done
clear
C)
Ulothrix
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 184) Which of the following is related to genetic engineering?
A)
Mutation
done
clear
B)
Plasmid
done
clear
C)
Plastid
done
clear
D)
Heterosis
done
clear
View Answer play_arrow
question_answer 185) Greenhouse effect refers to
A)
production of cereals
done
clear
B)
cooling of earth
done
clear
C)
trapping of UV-rays
done
clear
D)
warming of earth
done
clear
View Answer play_arrow
question_answer 186) Which of the following are homologous organs?
A)
Nails of human being and claws in animals
done
clear
B)
Wings of bird and wings of insect
done
clear
C)
Wings of bird and hand of human
done
clear
D)
Wings of bat and cockroach
done
clear
View Answer play_arrow
question_answer 187) The junction between the axon of one neuron and the dendrite of the next is called
A)
junction point
done
clear
B)
a synapse
done
clear
C)
a joint
done
clear
D)
constant bridge
done
clear
View Answer play_arrow
question_answer 188) To which of the following family do folic acid and pantothenic acid belong?
A)
Vitamin\[-C\]
done
clear
B)
Vitamin\[-K\]
done
clear
C)
Vitamin\[-A\]
done
clear
D)
Vitamin\[-B\]complex
done
clear
View Answer play_arrow
question_answer 189) The thickening of walls of arteries are called
A)
arthritis
done
clear
B)
atherosclerosis
done
clear
C)
anaeurysm
done
clear
D)
Both (a) and (c)
done
clear
View Answer play_arrow
question_answer 190) Due to which of the following organism yield of rice has been increased?
A)
Anabaena
done
clear
B)
Bacillus polymyxa
done
clear
C)
Bacillus papulae
done
clear
D)
Sesbania
done
clear
View Answer play_arrow
question_answer 191) The rate of photosynthesis is higher in
A)
red light
done
clear
B)
blue light
done
clear
C)
green light
done
clear
D)
very high light
done
clear
View Answer play_arrow
question_answer 192) The closing and opening of the leaves of Mimosa pudica is due to
A)
chemonastic movement
done
clear
B)
ther monastic movement
done
clear
C)
seismonastic movement
done
clear
D)
hydrotropic movement
done
clear
View Answer play_arrow
question_answer 193) Energy transfer from one trophic level to other in a food chain is
A)
20%
done
clear
B)
10%
done
clear
C)
2%
done
clear
D)
1%
done
clear
View Answer play_arrow
question_answer 194) The antibodies are
A)
germs
done
clear
B)
carbohydrates
done
clear
C)
proteins
done
clear
D)
lipids
done
clear
View Answer play_arrow
question_answer 195) Temperature changes in the environment affect most of the animals which are
A)
homoiothermic
done
clear
B)
aquatic
done
clear
C)
poikilothermic
done
clear
D)
desert living
done
clear
View Answer play_arrow
question_answer 196) The term humulin is used for
A)
human insulin
done
clear
B)
powerful antibiotic
done
clear
C)
isoenzyme
done
clear
D)
hydrolytic enzyme
done
clear
View Answer play_arrow
question_answer 197) The gonadotropic hormones are produced in
A)
interstitial cells of testes
done
clear
B)
adrenal cortex
done
clear
C)
adenohypophysis of pituitary
done
clear
D)
posterior part of thyroid
done
clear
View Answer play_arrow
question_answer 198) The first transgenic crop was
A)
cotton
done
clear
B)
pea
done
clear
C)
tobacco
done
clear
D)
flax
done
clear
View Answer play_arrow
question_answer 199) The process of replication in plasmid DNA, other than initiation, is controlled by
A)
mitochondrial gene
done
clear
B)
bacterial gene
done
clear
C)
plasmid gene
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 200) Which of the following regarded as a unit of nervous tissue?
A)
Myelin sheath
done
clear
B)
Axons
done
clear
C)
Dendrites
done
clear
D)
Neurons
done
clear
View Answer play_arrow