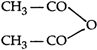question_answer 1) A wave front is an imaginary surface where;
A)
all particles lying on this vibrate in same phase
done
clear
B)
all particles tying on this vibrate in opposite phase
done
clear
C)
some particles in same phase and some in opposite phase
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 2) A long wire is in north-south direction, it is dropped freely towards earth, then no potential difference is induced between its ends. This statement is:
A)
true
done
clear
B)
false
done
clear
C)
meaningless
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 3) Radius of earth is 6400 km. The weight of body at a height of 6400 km from surface of earth will be:
A)
half
done
clear
B)
one fourth
done
clear
C)
one third
done
clear
D)
no change
done
clear
View Answer play_arrow
question_answer 4) The dimension of intensity of magnetic field is:
A)
\[\text{ }\!\![\!\!\text{ }{{\text{M}}^{\text{0}}}{{\text{L}}^{\text{-1}}}{{\text{T}}^{\text{0}}}{{\text{A}}^{\text{1}}}\text{ }\!\!]\!\!\text{ }\]
done
clear
B)
\[\text{ }\!\![\!\!\text{ }{{\text{M}}^{1}}{{\text{L}}^{\text{1}}}{{\text{T}}^{\text{-2}}}{{\text{A}}^{\text{-1}}}\text{ }\!\!]\!\!\text{ }\]
done
clear
C)
\[\text{ }\!\![\!\!\text{ }{{\text{M}}^{1}}{{\text{L}}^{0}}{{\text{T}}^{\text{-2}}}{{\text{A}}^{\text{-1}}}\text{ }\!\!]\!\!\text{ }\]
done
clear
D)
\[\text{ }\!\![\!\!\text{ }{{\text{M}}^{1}}{{\text{L}}^{1}}{{\text{T}}^{\text{-2}}}{{\text{A}}^{\text{1}}}\text{ }\!\!]\!\!\text{ }\]
done
clear
View Answer play_arrow
question_answer 5) On dropping a water drop on surface of oil it will:
A)
mix in oil
done
clear
B)
flow as a film
done
clear
C)
deformed
done
clear
D)
be of spherical shape
done
clear
View Answer play_arrow
question_answer 6) Value of escape velocity from earth surface is:
A)
11.2km/s
done
clear
B)
112m/s
done
clear
C)
22.4km/s
done
clear
D)
11200 km/s
done
clear
View Answer play_arrow
question_answer 7) Which of these relations is wrong?
A)
\[\text{1}\,\text{cal}\,\text{=4}\text{.18}\,\text{J}\]
done
clear
B)
\[\text{1}\,\overset{\text{ }\!\!{}^\circ\!\!\text{ }}{\mathop{\text{A}}}\,\,\text{=}\,\text{1}{{\text{0}}^{\text{18}}}\text{m}\]
done
clear
C)
\[MeV\,\,\text{=}\,\text{1}\text{.6}{{\times }^{\text{-13}}}\text{J}\]
done
clear
D)
\[1N\,\,\text{=}\,\text{1}{{\text{0}}^{\text{-5}}}\] dyne
done
clear
View Answer play_arrow
question_answer 8) The direction of wave front of a wave with the direction of propagation of wave is:
A)
parallel
done
clear
B)
perpendicular
done
clear
C)
opposite
done
clear
D)
at angle\[\]
done
clear
View Answer play_arrow
question_answer 9) Work of electric motor is:
A)
to convert A.C. into D.C.
done
clear
B)
to convert D.C. into A.C.
done
clear
C)
both (a) and (b)
done
clear
D)
to convert A.C. into mechanical energy
done
clear
View Answer play_arrow
question_answer 10) A.C. is measured b:
A)
induction coil
done
clear
B)
moving coil galvanometer
done
clear
C)
tangent galvanometer
done
clear
D)
hot wire ammeter
done
clear
View Answer play_arrow
question_answer 11) Which is true for adiabatic process?
A)
\[\text{d}\,\text{=}\,\text{0}\]
done
clear
B)
\[\text{dU=}\,\text{-}\,\text{dW}\]
done
clear
C)
\[=\]constant
done
clear
D)
S is not constant
done
clear
View Answer play_arrow
question_answer 12) True for real gas is:
A)
do not obey Boyles law
done
clear
B)
obey PV=RT
done
clear
C)
internal energy depends on temperature only
done
clear
D)
obey van der Waals equation
done
clear
View Answer play_arrow
question_answer 13) Lenzs law is bifurcated form of law:
A)
conservation of energy
done
clear
B)
conservation of mass
done
clear
C)
conservation of linear momentum
done
clear
D)
conservation of angular momentum
done
clear
View Answer play_arrow
question_answer 14) Angular momentum is constant in absent of external.
A)
force
done
clear
B)
torque
done
clear
C)
both a and b
done
clear
D)
inertia
done
clear
View Answer play_arrow
question_answer 15) Critical temperature of \[C{{O}_{2}}\] gas is:
A)
\[31.1{}^\circ C\]
done
clear
B)
\[55{}^\circ C\]
done
clear
C)
\[147.1{}^\circ C\]
done
clear
D)
\[273\text{ }K\]
done
clear
View Answer play_arrow
question_answer 16) To form a bigger drop constituting small drops. The surface energy of the bigger drop is... the total surface energy of small drops:
A)
less than
done
clear
B)
more than
done
clear
C)
equal to
done
clear
D)
cant say
done
clear
View Answer play_arrow
question_answer 17) Effect of forward bias on depletion layer is:
A)
decreases
done
clear
B)
increases
done
clear
C)
remains same
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 18) The reason for the appearance of bottom of bucket filled with liquid up :
A)
refraction of light
done
clear
B)
interference
done
clear
C)
diffraction
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 19) On charging a soap bubble its radius:
A)
increases
done
clear
B)
decreases
done
clear
C)
remains same
done
clear
D)
first increases then decreases
done
clear
View Answer play_arrow
question_answer 20) Light moves in straight line because:
A)
it is not absorbed by the atmosphere
done
clear
B)
its velocity is too high
done
clear
C)
effect of diffraction is negligible for light
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 21) Hot wire ameter is used for:
A)
to measure D.C.
done
clear
B)
to measure A.C.
done
clear
C)
neither in A.C. nor in D.C,
done
clear
D)
for both A.C. and D.C.
done
clear
View Answer play_arrow
question_answer 22) Which of the statement is wrong for isothermal state?
A)
Temperature is constant
done
clear
B)
Internal energy is constant
done
clear
C)
No transfer of energy takes place
done
clear
D)
1st two statements are true
done
clear
View Answer play_arrow
question_answer 23) A resistance and r coil are connected in series with an A.C. sourer voltage 20 V. If the potential difference across resistance across resistance is 12 volt. Then what will be potential difference across the coil:
A)
16 volt
done
clear
B)
10 volt
done
clear
C)
8 volt
done
clear
D)
6 volt
done
clear
View Answer play_arrow
question_answer 24) Average power loss in an induction coil is:
A)
\[\frac{1}{2}L{{I}^{2}}\]
done
clear
B)
\[2L{{I}^{2}}\]
done
clear
C)
\[\frac{1}{4}L{{I}^{2}}\]
done
clear
D)
zero
done
clear
View Answer play_arrow
question_answer 25) The minimum wavelength of X-rays produced from an electron accelerated from V volt is:
A)
\[\frac{eV}{hc}\]
done
clear
B)
\[\frac{hc}{eV}\]
done
clear
C)
\[\frac{ch}{eV}\]
done
clear
D)
\[\frac{h}{V}\]
done
clear
View Answer play_arrow
question_answer 26) The electric potential in a conducting hollow sphere will be:
A)
zero
done
clear
B)
proportional to distance from centre
done
clear
C)
inversely proportional to distance of centre
done
clear
D)
inversely proportional to square distance from centre
done
clear
View Answer play_arrow
question_answer 27) Two identical satellite are revolving round the earth at a height of R and 7R from surface of earth. Then which of the following statement is wrong (R is radius of earth)?
A)
The ratio of total energy of both will 4
done
clear
B)
The ratio of K.E. of both will be 4
done
clear
C)
The ratio of P.E. of both will be 4
done
clear
D)
The ratio of total energy of both will 4 and ratio of K.E. and P.E. will be2
done
clear
View Answer play_arrow
question_answer 28) The relation between flowing current \[\text{(i)}\] and deflection \[(\theta )\] obtained in moving coil galvanometer will be:
A)
\[i\propto \tan \,\theta \]
done
clear
B)
\[i\propto \,\theta \]
done
clear
C)
\[i\propto \,{{\theta }^{2}}\]
done
clear
D)
\[i\propto \,\sqrt{\theta }\]
done
clear
View Answer play_arrow
question_answer 29) Bending of light from the edges of object called:
A)
reflection
done
clear
B)
diffraction
done
clear
C)
refraction
done
clear
D)
interference
done
clear
View Answer play_arrow
question_answer 30) Amplification factor of a triode valve depends upon:
A)
temperature of cathode
done
clear
B)
plate voltage
done
clear
C)
grid voltage
done
clear
D)
grid and plate voltage
done
clear
View Answer play_arrow
question_answer 31) The torque acting on a current carrying loop placed in magnetic field does n depend upon:
A)
size of loop
done
clear
B)
area of loop
done
clear
C)
value of current
done
clear
D)
magnetic field
done
clear
View Answer play_arrow
question_answer 32) For a monochromatic light, in minimum deviation, position of prism :
A)
angle of incidence is more than emergence angle
done
clear
B)
incidence angle is \[90{}^\circ \]
done
clear
C)
incidence angle is equal to emergence angle
done
clear
D)
incidence angle emergence angle and prism angle will be same
done
clear
View Answer play_arrow
question_answer 33) Two rod of metal A and B are of same length has their resistance in the ratio 1:2. Which will suffer more loss in weight on inserting in water?
A)
Loss of weight be more for A
done
clear
B)
Loss of weight will be more for B
done
clear
C)
Loss of weight will be equal for both
done
clear
D)
Ratio of weight will be 1 : 2
done
clear
View Answer play_arrow
question_answer 34) A glass slab of thickness 4 cm contains the same number of waves as 5 cm of water column- When both are transmersed by the same monochromatic light. If the refractive index of water is 4/3, what is that of glass:
A)
5/3
done
clear
B)
5/4
done
clear
C)
16/15
done
clear
D)
1/5
done
clear
View Answer play_arrow
question_answer 35) When the torque acting on a rotating body is zero then what will remain constant?
A)
Force
done
clear
B)
Linear momentum
done
clear
C)
Angular momentum
done
clear
D)
All the above
done
clear
View Answer play_arrow
question_answer 36) Work of a convex lens is:
A)
it converges light rays
done
clear
B)
it diverges light rays
done
clear
C)
always form real images
done
clear
D)
always form virtual images
done
clear
View Answer play_arrow
question_answer 37) If a horizontal wire is dropped in east-west direction then:
A)
no e.m.f. will be induced
done
clear
B)
no current will be induced
done
clear
C)
induced current will flow from west to east
done
clear
D)
induced current will flow from east to west
done
clear
View Answer play_arrow
question_answer 38) When a capillary of radius R is dipped into water, water rises upto height H and the mass of this liquid is M. Now if capillary of radius 2R is dipped into water. What will be the mass of the water rises in capillary:
A)
M
done
clear
B)
2M
done
clear
C)
\[\frac{M}{2}\]
done
clear
D)
4M
done
clear
View Answer play_arrow
question_answer 39) If Youngs double slit experiment is performed in water, then fringe width will:
A)
infinite
done
clear
B)
decrease
done
clear
C)
increase
done
clear
D)
remain unchanged
done
clear
View Answer play_arrow
question_answer 40) If the half life time of a radioactive material is 3 hours. What will be its quantity after 9 hours?
A)
1/9
done
clear
B)
1/27
done
clear
C)
1/6
done
clear
D)
1/8
done
clear
View Answer play_arrow
question_answer 41) Both \[\text{-}\]rays and X-rays are electromagnetic waves, then the main difference between, these is :
A)
X-rays are produced from nucleus while \[\text{-}\]rays are speciality of atom
done
clear
B)
X-rays are speciality of atom and \[\text{-}\]rays are produced from nucleus
done
clear
C)
Frequency of \[\text{-}\]rays is less than X-rays
done
clear
D)
velocity of \[\text{-}\]rays is more than X-rays
done
clear
View Answer play_arrow
question_answer 42) Velocity of light in diamond, glass and water will be in tine following order:
A)
diamond < water < glass
done
clear
B)
water < glass < diamond
done
clear
C)
diamond > water > glass
done
clear
D)
diamond > glass > water
done
clear
View Answer play_arrow
question_answer 43) In Youngs experiment, when the distance between slit and screen is doubled, while separation of slit is halved, then fringe width will be:
A)
4 times
done
clear
B)
1/4
done
clear
C)
doubled
done
clear
D)
unchanged
done
clear
View Answer play_arrow
question_answer 44) The process in which solid converts directly in vapour on heating is known as:
A)
vaporization
done
clear
B)
sublimation
done
clear
C)
boiling
done
clear
D)
condensation
done
clear
View Answer play_arrow
question_answer 45) Two long thin parallel wires are placed at a distance (r) from each other carrying equal current \[I\] in opposite direction. The magnitude of force per unit length which is exerted by each wire on the other is :
A)
\[\frac{{{\mu }_{0}}{{I}^{2}}}{r}\]
done
clear
B)
\[\frac{{{\mu }_{0}}{{I}^{2}}}{2\pi r}\]
done
clear
C)
\[\frac{{{\mu }_{0}}I}{2\pi r}\]
done
clear
D)
\[\frac{{{\mu }_{0}}^{2}I}{2\pi {{r}^{2}}}\]
done
clear
View Answer play_arrow
question_answer 46) At a definite temperature for a given wavelength the ratio of emissive power of all objects and that of perfectly black body at the same temperature is called as :
A)
relative emissivity
done
clear
B)
emissivity
done
clear
C)
absorption coefficient
done
clear
D)
reflection coefficient
done
clear
View Answer play_arrow
question_answer 47) On bombarding neutrons on\[_{\text{92}}{{\text{U}}^{\text{235}}}\] the number of emitted neutrons will be:
A)
1
done
clear
B)
2
done
clear
C)
3
done
clear
D)
4
done
clear
View Answer play_arrow
question_answer 48) Frequency of A.C. in India is:
A)
50 Hz
done
clear
B)
60 Hz
done
clear
C)
220 Hz
done
clear
D)
110 Hz
done
clear
View Answer play_arrow
question_answer 49) If we carry a body of 5 kg in space, then its mass will be:
A)
5 kg
done
clear
B)
10kg
done
clear
C)
2 kg
done
clear
D)
30kg
done
clear
View Answer play_arrow
question_answer 50) Magnetic suspectibility of a diamagnetic substance is:
A)
small and positive
done
clear
B)
small and negative
done
clear
C)
high and negative
done
clear
D)
high and positive
done
clear
View Answer play_arrow
question_answer 51) Refractive index of glass is 3/2 and that of water is 4/3. If speed of light in glass is 2x 108 m/s, then speed of light in water is:
A)
\[2.67\,\times {{10}^{8}}\,m/s\]
done
clear
B)
\[2.67\,\times {{10}^{-2}}\,m/s\]
done
clear
C)
\[2\times {{10}^{8}}\,m/s\]
done
clear
D)
\[2.25\,\times {{10}^{8}}\,m/s\]
done
clear
View Answer play_arrow
question_answer 52) Temperature which is same in centigrade and Fahrenheit scales of temperature is:
A)
\[-40{}^\circ \]
done
clear
B)
\[40{}^\circ \]
done
clear
C)
\[36.6{}^\circ \]
done
clear
D)
\[3.8{}^\circ \]
done
clear
View Answer play_arrow
question_answer 53)
The value of R in the following circuit diagram will be:
A)
5\[\Omega \]
done
clear
B)
6\[\Omega \]
done
clear
C)
8\[\Omega \]
done
clear
D)
24\[\Omega \]
done
clear
View Answer play_arrow
question_answer 54) Focal length of a convergent lens is R. Now if lens is dipped into water of refractive index 1.33, then focal length of lens will be (refractive index of material of lens = 1.5):
A)
R
done
clear
B)
2R
done
clear
C)
4R
done
clear
D)
R/2
done
clear
View Answer play_arrow
question_answer 55) If the refractive index of a material for red, yellow and voilet colours are 1.42, 1.50, 1.62. Then dispersive power of the material will be:
A)
0.4
done
clear
B)
1.33
done
clear
C)
0.2
done
clear
D)
0.1
done
clear
View Answer play_arrow
question_answer 56) A magnet is brought near a (i) rapidly (ii) slowly, the induced e.m.f. will be :
A)
more in case\[\text{I}\]
done
clear
B)
more in case \[\text{II}\]
done
clear
C)
equal in both cases
done
clear
D)
more or less depending upon radius of coil
done
clear
View Answer play_arrow
question_answer 57) A watch shows time as 3 : 25, when this time is seen in a mirror, the time will be.
A)
8: 35
done
clear
B)
9 : 35
done
clear
C)
7 : 35
done
clear
D)
8 : 25
done
clear
View Answer play_arrow
question_answer 58) Two sources of light are called coherent if their:
A)
wavelength is equal
done
clear
B)
amplitudes are equal
done
clear
C)
wavelengths are equal and phase difference is constant
done
clear
D)
amplitude and wavelengths are equal
done
clear
View Answer play_arrow
question_answer 59) When a ray of light enters into water from air, then its wavelength :
A)
decreases
done
clear
B)
increases
done
clear
C)
remains same
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 60) In a A.C. circuit V = 100 sin t volt and \[I=100\,\sin \,\left( 100t+\frac{\pi }{3} \right)\]A, then average power loss will be:
A)
\[{{10}^{4}}W\]
done
clear
B)
100 W
done
clear
C)
2.5 kW
done
clear
D)
10 W
done
clear
View Answer play_arrow
question_answer 61) Frequency range of audible waves is:
A)
20 to 20,000 Hz
done
clear
B)
\[{{10}^{6}}\] to \[{{10}^{8}}\] Hz
done
clear
C)
10-20 Hz
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 62) In a D.C. motor the current in the armature is maximum at the:
A)
time of start
done
clear
B)
time of off
done
clear
C)
position of maximum speed
done
clear
D)
all the times
done
clear
View Answer play_arrow
question_answer 63) Choose the wrong statement for L-C-R series circuit:
A)
potential difference across resistance and the applied e.m.f. are always in same phase
done
clear
B)
the phase difference between e.m.f. and potential difference across inductive coil is \[90{}^\circ \]
done
clear
C)
the phase difference between potential difference across capacitor and potential difference across inductance is \[\text{/2}\]
done
clear
D)
the phase difference between potential difference across capacitor and potential difference across resistance is\[\text{/2}\]
done
clear
View Answer play_arrow
question_answer 64) Wave front means:
A)
all the particles on same wave front are in same phase of vibration
done
clear
B)
all the particles on same wave front are in opposite phase of vibration
done
clear
C)
some particles are in same phase and some arc in opposite phase
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 65) In Youngs experiment, slits are separated by 1 mm and screen is 1 m away from slits. If 10th bright fringe is at a distance of 5 mm from central then wavelengths of light used will be:
A)
\[5000\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
B)
\[6000\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
C)
\[7000\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
D)
\[8000\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
View Answer play_arrow
question_answer 66) A compound lens is formed by combining one convex and one concave lens of same focal length 20 cm. This compound lens is used to see a 5 cm high object situated at a distance of 20 cm. Image will be:
A)
enlarged and reverse
done
clear
B)
small and erect
done
clear
C)
errect and same size of object
done
clear
D)
reverse and same size of object
done
clear
View Answer play_arrow
question_answer 67) Dispersive power of a prism depends upon:
A)
material of prism
done
clear
B)
angle of prism
done
clear
C)
shape of prism
done
clear
D)
incidence angle
done
clear
View Answer play_arrow
question_answer 68) Resistance of a chock coil is:
A)
high
done
clear
B)
low
done
clear
C)
definite
done
clear
D)
infinite
done
clear
View Answer play_arrow
question_answer 69) Fresnels biprism experiment is performed in water in place of air then fringe width will:
A)
increase
done
clear
B)
decrease
done
clear
C)
remains same
done
clear
D)
cant say
done
clear
View Answer play_arrow
question_answer 70) \[\text{-}\]rays can be differentiate from X-rays on the following basis:
A)
on the basis of production
done
clear
B)
on basis of penetrating power
done
clear
C)
on basis of reflection
done
clear
D)
on basis of diffraction
done
clear
View Answer play_arrow
question_answer 71) On a white screen red light from one source and green light from another source is falling. Colour of the screen will be:
A)
black
done
clear
B)
yellow
done
clear
C)
green
done
clear
D)
blue
done
clear
View Answer play_arrow
question_answer 72) Soap film appears coloured because:
A)
dispersion of light
done
clear
B)
interference of light
done
clear
C)
refraction of light
done
clear
D)
total internal refraction
done
clear
View Answer play_arrow
question_answer 73) The ratio of average potential energy and total energy of a body will be :
A)
1 : 2
done
clear
B)
2 : 1
done
clear
C)
3 : 2
done
clear
D)
2 : 3
done
clear
View Answer play_arrow
question_answer 74) Two lenses of power + 2.5 D and - 3.75 D are placed in contact to form compound lens. What will be the focal length of compound lens?
A)
- 20 cm
done
clear
B)
- 40 cm
done
clear
C)
- 60 cm
done
clear
D)
- 80 cm
done
clear
View Answer play_arrow
question_answer 75) If the distance between point light source and screen is doubled then intensity of light will be:
A)
\[\text{4I}\]
done
clear
B)
\[\text{2I}\]
done
clear
C)
\[\text{I/2}\]
done
clear
D)
\[\text{I/4}\]
done
clear
View Answer play_arrow
question_answer 76) Two lenses in contact form an achromatic lens, their focal lengths are in the ratio 2:3 Their dispersive powers must be in the ratio of:
A)
1 : 3
done
clear
B)
2 : 3
done
clear
C)
3 : 2
done
clear
D)
3 : 1
done
clear
View Answer play_arrow
question_answer 77) Thermionic current \[\text{(I)}\]depends on temperature (T) as :
A)
\[{{T}^{2}}\]
done
clear
B)
\[{{T}^{1/2}}\]
done
clear
C)
\[{{T}^{3/2}}\]
done
clear
D)
\[{{T}^{4}}\]
done
clear
View Answer play_arrow
question_answer 78) A pair of isotoncs is :
A)
\[_{\text{6}}{{\text{C}}^{\text{14}}}\,\text{and}{{\,}_{\text{6}}}{{\text{C}}^{\text{12}}}\]
done
clear
B)
\[_{\text{6}}{{\text{C}}^{\text{15}}}\,\text{and}{{\,}_{7}}{{\text{C}}^{\text{14}}}\]
done
clear
C)
\[_{7}{{\text{C}}^{\text{13}}}\,\text{and}{{\,}_{6}}{{\text{C}}^{\text{12}}}\]
done
clear
D)
\[_{7}{{\text{C}}^{\text{13}}}\,\text{and}{{\,}_{6}}{{\text{C}}^{\text{14}}}\]
done
clear
View Answer play_arrow
question_answer 79) Property of converging lens is:
A)
converges light rays on a point
done
clear
B)
diverges light rays
done
clear
C)
no lens is converging
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 80) Direction of current in diode valve is:
A)
cathode to plate
done
clear
B)
plate to cathode
done
clear
C)
current is same for both sides
done
clear
D)
current is zero in diode
done
clear
View Answer play_arrow
question_answer 81) Diode is used as a:
A)
oscillator
done
clear
B)
amplifier
done
clear
C)
rectifier
done
clear
D)
current amplifier
done
clear
View Answer play_arrow
question_answer 82) Process of converting A.C. into D.C. is:
A)
purification
done
clear
B)
amplification
done
clear
C)
rectification
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 83) A mica sheet of thickness t and refractive index \[\mu \] is introduced in the path of one or two in terferring beams in Youngs double slit experiment. The central fringe will be displaced by:
A)
\[\frac{(\mu -1)t}{dD}\]
done
clear
B)
\[0\]
done
clear
C)
\[\infty \]
done
clear
D)
\[\frac{(\mu -1)tD}{d}\]
done
clear
View Answer play_arrow
question_answer 84) Which of the following is anti particle of electron?
A)
\[_{0}{{n}^{1}}\]
done
clear
B)
\[_{1}{{H}^{1}}\]
done
clear
C)
Positron
done
clear
D)
Neutron
done
clear
View Answer play_arrow
question_answer 85) If mass and radius of a hollow sphere are M and R respectively, then its moment of inertia, w.r.t. its diameter is :
A)
\[\frac{2}{5}M{{R}^{2}}\]
done
clear
B)
\[\frac{2}{3}M{{R}^{2}}\]
done
clear
C)
\[\frac{1}{2}M{{R}^{2}}\]
done
clear
D)
\[M{{R}^{2}}\]
done
clear
View Answer play_arrow
question_answer 86) The time of flight and range of a projectile are 10 second and 500 m respectively. The maximum height obtained by it will be:
A)
125 m
done
clear
B)
50 m
done
clear
C)
100 m
done
clear
D)
150 m
done
clear
View Answer play_arrow
question_answer 87) Which of the following is true for triode valve?
A)
\[\,\text{=}\,\text{r}{{ }_{\text{P}}}\text{ }\!\!\times\!\!\text{ }\,{{\text{g}}_{\text{m}}}\]
done
clear
B)
\[\,\text{r}{{ }_{\text{P}}}=\,{{\text{g}}_{\text{m}}}\times \mu \]
done
clear
C)
\[\,\,{{\text{g}}_{\text{m}}}=\mu \times {{r}_{p}}\]
done
clear
D)
\[\,\,{{\text{g}}_{\text{m}}}=\frac{\mu }{{{r}_{p}}}\]
done
clear
View Answer play_arrow
question_answer 88) The heat given to an ideal gas in isothermal conditions is used to:
A)
increase temperature
done
clear
B)
do external work
done
clear
C)
increase temperature and in doing external work
done
clear
D)
increase internal energy
done
clear
View Answer play_arrow
question_answer 89) A current of 20\[\text{A}\] is flowing through a wire for 30 sec. The charge transferred through wire is:
A)
\[2\times {{10}^{-4}}\] coulomb
done
clear
B)
\[4\times {{10}^{-4}}\] coulomb
done
clear
C)
\[6\times {{10}^{-4}}\] coulomb
done
clear
D)
\[8\times {{10}^{-4}}\] coulomb
done
clear
View Answer play_arrow
question_answer 90) lionization energy of hydrogen atom is 13.6 \[\text{eV}\text{.}\,\text{If}\,\text{h=6}\text{.6 }\!\!\times\!\!\text{ 1}{{\text{0}}^{\text{-34}\,}}\text{Joule-sec,}\]then value of R (Rydberg constant) will be:
A)
\[{{10}^{10}}\] per metre
done
clear
B)
\[{{10}^{7}}\] per metre
done
clear
C)
\[{{10}^{4}}\] per metre
done
clear
D)
\[{{10}^{-7}}\] per metre
done
clear
View Answer play_arrow
question_answer 91) Internal energy of an ideal gas depends upon:
A)
temperature, pressure and volume
done
clear
B)
only on temperature and pressure
done
clear
C)
only on pressure and volume
done
clear
D)
only on temperature
done
clear
View Answer play_arrow
question_answer 92) The decreasing order of band gap energy is:
A)
insulator > semiconductor > conductor
done
clear
B)
insulator > conductor > semi conductor
done
clear
C)
conductor < semiconductor < insulator
done
clear
D)
semiconductor < conductor < insulator
done
clear
View Answer play_arrow
question_answer 93) A magnet is brought near a coil (i) rapidly (ii) slowly then induced e.m.f will be:
A)
more in case (i)
done
clear
B)
less in case (i)
done
clear
C)
equal in both cases
done
clear
D)
depends on radius of ring
done
clear
View Answer play_arrow
question_answer 94) if \[h=6.6\times {{10}^{-34}}J-\sec ,\] then energy of quanta of frequency \[{{10}^{15}}Hz\] will be:
A)
\[6.6\,\times {{10}^{-19}}J\]
done
clear
B)
\[6.6\,\times {{10}^{-12}}J\]
done
clear
C)
\[6.6\,\times {{10}^{-14}}J\]
done
clear
D)
\[6.6\,\times {{10}^{-4}}J\]
done
clear
View Answer play_arrow
question_answer 95) Imagine a meteroid revolving around a big star in a circular orbit of radius R with time period T. If the force of attraction between star and meteroid is proportional to\[{{R}^{-5/2}},\] then T2 will be proportional to:
A)
\[{{R}^{3}}\]
done
clear
B)
\[{{R}^{7/2}}\]
done
clear
C)
\[{{R}^{5/2}}\]
done
clear
D)
\[{{R}^{3/2}}\]
done
clear
View Answer play_arrow
question_answer 96) Electric energy is transferred to distant place at high voltage because :
A)
it will stop stealing of wires
done
clear
B)
it stops power decay
done
clear
C)
generator produce energy of high voltage
done
clear
D)
electric energy is transferred quickly on high voltage
done
clear
View Answer play_arrow
question_answer 97) Which of the following reaction can be completed by low pressure:
A)
\[{{N}_{2}}+{{O}_{2}}2NO\]
done
clear
B)
\[{{H}_{2}}+{{I}_{2}}2HI\]
done
clear
C)
\[PC{{l}_{5}}PC{{l}_{3}}+C{{l}_{2}}\]
done
clear
D)
\[{{H}_{2}}+3{{H}_{2}}2N{{H}_{3}}\]
done
clear
View Answer play_arrow
question_answer 98) The oxidation number of As in \[{{H}_{3}}As{{O}_{4}}\]
A)
\[-1\]
done
clear
B)
\[-3\]
done
clear
C)
\[+3\]
done
clear
D)
\[+5\]
done
clear
View Answer play_arrow
question_answer 99) Rectified sprit is:
A)
95.5% pure alcohol + 4.5% water
done
clear
B)
Anhydrous ethyl alcohol
done
clear
C)
Methylated sprit
done
clear
D)
Wood sprit
done
clear
View Answer play_arrow
question_answer 100) Aldehyde and ketone can be differentiated by:
A)
\[NaHS{{O}_{3}}\]
done
clear
B)
Fehling solution
done
clear
C)
\[HgC{{l}_{2}}\]
done
clear
D)
all of these
done
clear
View Answer play_arrow
question_answer 101) Which of the following has not hydrogen bond?
A)
Phenol
done
clear
B)
Liquid ammonia
done
clear
C)
Liquid \[HCl\]
done
clear
D)
Water
done
clear
View Answer play_arrow
question_answer 102) Ethyl amine on reaction with \[HN{{O}_{2}}\]gives:
A)
benzene
done
clear
B)
ethyl alcohol
done
clear
C)
nitrobenzene
done
clear
D)
ammonia
done
clear
View Answer play_arrow
question_answer 103) The eletronic configuration of the element having maximum electronegativity:
A)
\[1{{s}^{2}},2{{s}^{2}}2{{p}^{6}}\]
done
clear
B)
\[1{{s}^{2}},2{{s}^{2}}2{{p}^{5}}\]
done
clear
C)
\[1{{s}^{2}},2{{s}^{2}}\text{ }2{{p}^{6}},3{{s}^{2}}\text{ }3{{p}^{6}}\]
done
clear
D)
\[1{{s}^{2}},2{{s}^{2}}\text{ }2{{p}^{6}},3{{s}^{2}}\text{ }3{{p}^{6}}\text{ }3{{d}^{1}},4{{s}^{2}}\]
done
clear
View Answer play_arrow
question_answer 104) The minimum \[O-O\] bond length is in:
A)
\[{{O}_{2}}^{-2}\]
done
clear
B)
\[{{O}_{2}}^{+}\]
done
clear
C)
\[{{O}_{2}}^{-}\]
done
clear
D)
\[{{O}_{2}}\]
done
clear
View Answer play_arrow
question_answer 105) Aniline on bromination gives:
A)
p-bromo aniline
done
clear
B)
o, p-nitrophenol
done
clear
C)
o-bromo aniline
done
clear
D)
2, 4, 6-tribromo aniline
done
clear
View Answer play_arrow
question_answer 106) \[_{19}{{K}^{40}},{{\,}_{18}}A{{r}^{40}},{{\,}_{20}}C{{a}^{40}}\]are:
A)
isotones
done
clear
B)
isobars
done
clear
C)
isodiapheres
done
clear
D)
isotopes
done
clear
View Answer play_arrow
question_answer 107) Methyl phenyl ether on reaction with \[HN{{O}_{3}}\] and \[{{H}_{2}}S{{O}_{4}}\] results:
A)
done
clear
B)
and
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
question_answer 108) In an element as the distance of an orbit from the nucleus increases the energy of the orbit:
A)
increases
done
clear
B)
decreases
done
clear
C)
does not changes
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 109) Strongest base is:
A)
\[C{{H}_{3}}C{{H}_{2}}N{{H}_{2}}\]
done
clear
B)
\[C{{H}_{3}}N{{H}_{2}}\]
done
clear
C)
\[{{C}_{6}}{{H}_{5}}N{{H}_{2}}\]
done
clear
D)
\[N{{H}_{3}}\]
done
clear
View Answer play_arrow
question_answer 110) Paramagnetic ion is:
A)
\[{{[NiC{{l}_{4}}]}^{-2}}\]
done
clear
B)
\[{{[Ni{{(CN)}_{4}}]}^{-2}}\]
done
clear
C)
both (a) and (b)
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 111) In which of the following molecule is electrovalent?
A)
\[KCl\]
done
clear
B)
\[N{{H}_{4}}Cl\]
done
clear
C)
\[PC{{l}_{5}}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 112) Strongest acid is:
A)
\[ClC{{H}_{2}}COOH\]
done
clear
B)
\[C{{H}_{3}}COOH\]
done
clear
C)
\[C{{l}_{2}}CHCOOH\]
done
clear
D)
\[CC{{l}_{3}}COOH\]
done
clear
View Answer play_arrow
question_answer 113) In which of the following react with ethene and formed propane?
A)
methane
done
clear
B)
propene
done
clear
C)
ethane
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 114) Lithopone used in:
A)
yellow paint
done
clear
B)
fertilization
done
clear
C)
white paint
done
clear
D)
red paint
done
clear
View Answer play_arrow
question_answer 115) Propanol on heating with hydrazine and \[KOH\] gives:
A)
propanal
done
clear
B)
propene
done
clear
C)
propane
done
clear
D)
ethanol
done
clear
View Answer play_arrow
question_answer 116) Phenol on reaction with dilute \[HN{{O}_{3}}\] gives:
A)
o, p-nitrophenol
done
clear
B)
o-nitrophenol
done
clear
C)
p-nitrophenol
done
clear
D)
picric acid
done
clear
View Answer play_arrow
question_answer 117) The compound showing metamerism is:
A)
diethyl ether
done
clear
B)
ethyl chloride
done
clear
C)
1-butyne
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 118) According to Bronsted the acid is:
A)
protophilic
done
clear
B)
proton donor
done
clear
C)
proton acceptor
done
clear
D)
amphoteric
done
clear
View Answer play_arrow
question_answer 119) Out of the following the Avogadro number is:
A)
\[6.023\times {{10}^{23}}\]
done
clear
B)
\[6.023\times {{10}^{22}}\]
done
clear
C)
\[6.032\times {{10}^{22~}}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 120) The correct order of the basicity of amines is (JR= ethyl)
A)
\[N{{H}_{3}}>RN{{H}_{2}}>{{R}_{2}}NH>{{R}_{3}}N\]
done
clear
B)
\[RN{{H}_{2}}>N{{H}_{3}}>{{R}_{2}}NH>{{R}_{3}}N\]
done
clear
C)
\[{{R}_{2}}NH>{{R}_{3}}N>RN{{H}_{2}}>N{{H}_{3}}\]
done
clear
D)
\[{{R}_{2}}NH>RN{{H}_{2}}>{{R}_{3}}N>N{{H}_{3}}\]
done
clear
View Answer play_arrow
question_answer 121) The benzene triozonide obtained from which compound ozonisation?
A)
Benzene
done
clear
B)
Amine
done
clear
C)
Both (a) and (b)
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 122) Correct order of bond angle is:
A)
\[s{{p}^{3}}>sp<s{{p}^{3}}\]
done
clear
B)
\[sp<s{{p}^{1}}<s{{p}^{3}}\]
done
clear
C)
\[s{{p}^{3}}<s{{p}^{2}}<sp\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 123) The oxidation number of S in Marshal acid:
A)
\[+6\]
done
clear
B)
\[+5\]
done
clear
C)
\[-7\]
done
clear
D)
\[+8\]
done
clear
View Answer play_arrow
question_answer 124) If the pH value of solution is 8 then solution will be:
A)
acidic
done
clear
B)
alkaline
done
clear
C)
neutral
done
clear
D)
amphoteric
done
clear
View Answer play_arrow
question_answer 125) Plaster of Paris is:
A)
\[CuS{{O}_{4}}.\frac{1}{2}{{H}_{2}}O\]
done
clear
B)
\[CaOC{{l}_{2}}\]
done
clear
C)
\[CaS{{O}_{4}}.\,2{{H}_{2}}O\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 126) The non-linear molecule is:
A)
\[{{C}_{2}}{{H}_{2}}\]
done
clear
B)
\[C{{O}_{2}}\]
done
clear
C)
\[HgC{{l}_{2}}\]
done
clear
D)
\[S{{O}_{2}}\]
done
clear
View Answer play_arrow
question_answer 127) Methyl amine on reaction with \[HN{{O}_{2}}\] gives:
A)
\[C{{H}_{2}}-O-C{{H}_{3}}\]
done
clear
B)
\[C{{H}_{3}}-O-N=0\]
done
clear
C)
both (a) and (b)
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 128) Electrophilic in sulphonation is:
A)
\[S{{O}_{3}}\]
done
clear
B)
\[S{{O}_{2}}\]
done
clear
C)
\[{{H}_{2}}O\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 129) Paramagnetic compound is:
A)
\[TiC{{l}_{4}}\]
done
clear
B)
\[CrC{{l}_{3}}\]
done
clear
C)
\[MnC{{l}_{2}}\]
done
clear
D)
\[SeC{{l}_{3}}\]
done
clear
View Answer play_arrow
question_answer 130) Compound having co-ordinate bond is:
A)
\[N{{H}_{3}}\]
done
clear
B)
\[C{{H}_{4}}\]
done
clear
C)
\[C{{O}_{2}}\]
done
clear
D)
\[SO_{3}^{2-}\]
done
clear
View Answer play_arrow
question_answer 131) Two electrons cannot have the same values for all the four quantum number this law is:
A)
(n +1) rule
done
clear
B)
Aufbau rule
done
clear
C)
Hunds rule
done
clear
D)
Paulis exclusion principle
done
clear
View Answer play_arrow
question_answer 132) Modem periodic table is based on:
A)
atomic weight
done
clear
B)
atomic number
done
clear
C)
atomic volume
done
clear
D)
atomic energy
done
clear
View Answer play_arrow
question_answer 133) The acetyl chloride formed when \[PC{{l}_{3}}\]react with:
A)
\[C{{H}_{3}}COOH\]
done
clear
B)
\[C{{H}_{3}}CHO\]
done
clear
C)
\[HCOOH\]
done
clear
D)
\[C{{H}_{3}}OH\]
done
clear
View Answer play_arrow
question_answer 134) Which compound will give iodoform test?
A)
\[C{{H}_{3}}OH\]
done
clear
B)
\[{{C}_{2}}{{H}_{5}}OH\]
done
clear
C)
\[C{{H}_{3}}COOH\]
done
clear
D)
Formalin
done
clear
View Answer play_arrow
question_answer 135) The compound having maximum boiling point is:
A)
ethyl alcohol
done
clear
B)
\[{{({{C}_{2}}{{H}_{5}})}_{3}}N\]
done
clear
C)
\[{{({{C}_{2}}{{H}_{5}})}_{2}}O\]
done
clear
D)
ethyl chloride
done
clear
View Answer play_arrow
question_answer 136) Which of the following molecule is paramagnetic?
A)
\[{{O}_{2}}^{-}\]
done
clear
B)
\[C{{N}^{-}}\]
done
clear
C)
\[CO\]
done
clear
D)
\[N{{O}^{+}}\]
done
clear
View Answer play_arrow
question_answer 137) Lewis base is:
A)
\[S{{O}_{3}}\]
done
clear
B)
\[C{{O}_{2}}\]
done
clear
C)
\[S{{O}_{2}}\]
done
clear
D)
\[ROH\]
done
clear
View Answer play_arrow
question_answer 138) \[C{{H}_{3}}COOH\]form dimer because of:
A)
intramolecular hydrogen bond
done
clear
B)
covalent bond
done
clear
C)
intermolecular hydrogen bond
done
clear
D)
hydrogen bond
done
clear
View Answer play_arrow
question_answer 139) Ion having maximum unpaired electrons?
A)
\[C{{r}^{+2}}\]
done
clear
B)
\[F{{e}^{+2}}\]
done
clear
C)
\[M{{n}^{+4}}\]
done
clear
D)
\[M{{n}^{+2}}\]
done
clear
View Answer play_arrow
question_answer 140) Isopropyl alcohol on oxidation or passing its vapours on hot copper gives:
A)
\[C{{H}_{3}}CHO\]
done
clear
B)
\[C{{H}_{3}}COC{{H}_{3}}\]
done
clear
C)
\[C{{H}_{2}}=C{{H}_{2}}\]
done
clear
D)
\[C{{H}_{3}}-O-C{{H}_{3}}\]
done
clear
View Answer play_arrow
question_answer 141) Isotope have:
A)
different mass number
done
clear
B)
same physical properties
done
clear
C)
same chemical properties
done
clear
D)
same neutron number
done
clear
View Answer play_arrow
question_answer 142) Compound with acidic nature is:
A)
\[Pb(OH)Cl\]
done
clear
B)
\[BaCl\]
done
clear
C)
\[N{{a}_{2}}S{{O}_{4}}\]
done
clear
D)
\[N{{a}_{2}}HP{{O}_{4}}\]
done
clear
View Answer play_arrow
question_answer 143) The gas evolved in fermentation is:
A)
\[C{{H}_{4}}\]
done
clear
B)
\[{{O}_{2}}\]
done
clear
C)
\[C{{O}_{2}}\]
done
clear
D)
\[{{N}_{2}}\]
done
clear
View Answer play_arrow
question_answer 144) Cyclohexene on reaction with bromine yields:
A)
done
clear
B)
done
clear
C)
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 145) The main function of roasting is:
A)
oxidation
done
clear
B)
smelting
done
clear
C)
reduction
done
clear
D)
to remove vapoumess substance
done
clear
View Answer play_arrow
question_answer 146) Minimum \[C-C\] bond length is in:
A)
\[{{C}_{2}}{{H}_{6}}\]
done
clear
B)
\[{{C}_{2}}{{H}_{2}}\]
done
clear
C)
\[{{C}_{6}}{{H}_{6}}\]
done
clear
D)
\[{{C}_{2}}{{H}_{4}}\]
done
clear
View Answer play_arrow
question_answer 147) Which element does not has neutron in its nucleus?
A)
\[Li\]
done
clear
B)
\[H\]
done
clear
C)
\[O\]
done
clear
D)
\[C\]
done
clear
View Answer play_arrow
question_answer 148) The strongest base is:
A)
\[Zn{{(OH)}_{2}}\]
done
clear
B)
\[Al{{(OH)}_{3}}\]
done
clear
C)
\[Ba{{(OH)}_{2}}\]
done
clear
D)
\[Fe{{(OH)}_{3}}\]
done
clear
View Answer play_arrow
question_answer 149) Proton is discovered by:
A)
Rutherford
done
clear
B)
Pauling
done
clear
C)
Thomson
done
clear
D)
Dalton
done
clear
View Answer play_arrow
question_answer 150) When nitro toluene react with \[HN{{O}_{3}}+{{H}_{2}}S{{O}_{4}}\] gives:
A)
picric acid
done
clear
B)
1, 4-dinitro benzene
done
clear
C)
trinitro toluene
done
clear
D)
1, 3-dinitro benzene
done
clear
View Answer play_arrow
question_answer 151) Carbonyl chloride is:
A)
phosgene
done
clear
B)
chlorobenzene
done
clear
C)
both (a) and (b)
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 152) Which molecule have not zero dipole moment?
A)
\[PC{{l}_{5}}\]
done
clear
B)
\[{{C}_{6}}{{H}_{6}}\]
done
clear
C)
\[CC{{l}_{4}}\]
done
clear
D)
\[{{H}_{2}}O\]
done
clear
View Answer play_arrow
question_answer 153) Which compound does not give Cannizzaro reaction?
A)
Acetaldehyde
done
clear
B)
Formaldehyde
done
clear
C)
Benzaldehyde
done
clear
D)
\[CC{{l}_{3}}CHO~\]
done
clear
View Answer play_arrow
question_answer 154) The reaction of ethylene bromide with zinc yields:
A)
alkene
done
clear
B)
alkyne
done
clear
C)
alkane
done
clear
D)
all of these
done
clear
View Answer play_arrow
question_answer 155) Absolute alcohol is represented as:
A)
\[{{C}_{2}}{{H}_{5}}OH+water+petrol\]
done
clear
B)
\[95%\text{ }{{C}_{2}}{{H}_{5}}OH\text{ +}5%\text{ }{{H}_{2}}O\]
done
clear
C)
\[95%\text{ }{{C}_{2}}{{H}_{5}}OH+5%\text{ }C{{H}_{3}}OH\]
done
clear
D)
100% pure alcohol
done
clear
View Answer play_arrow
question_answer 156) The litharz is represented by:
A)
\[PbO\]
done
clear
B)
\[P{{b}_{3}}{{O}_{4}}\]
done
clear
C)
\[PbCr{{O}_{4}}\]
done
clear
D)
\[Pb{{(OH)}_{2}}\]
done
clear
View Answer play_arrow
question_answer 157)
A)
chloretone
done
clear
B)
o-hydroxy benzaldehyde
done
clear
C)
salicyldehyde
done
clear
D)
all of these
done
clear
View Answer play_arrow
question_answer 158) Compound having maximum covalent character is:
A)
\[CaC{{l}_{2}}\]
done
clear
B)
\[KI\]
done
clear
C)
\[BeB{{r}_{2}}\]
done
clear
D)
\[NaF\]
done
clear
View Answer play_arrow
question_answer 159) The appropiate indicator used in the titration of strong acid and weak base is:
A)
phenolphthalene
done
clear
B)
phenol red
done
clear
C)
methyl orange
done
clear
D)
methyl red
done
clear
View Answer play_arrow
question_answer 160) The compound obtained on the hydrolysis of the product formed by acetaldehyde and Grignard reagent is:
A)
done
clear
B)
\[{{(C{{H}_{3}})}_{3}}C-OH\]
done
clear
C)
\[C{{H}_{3}}-OH\]
done
clear
D)
\[C{{H}_{3}}-O-C{{H}_{3}}\]
done
clear
View Answer play_arrow
question_answer 161) a particle is:
A)
\[_{0}{{n}^{1}}\]
done
clear
B)
\[_{2}H{{e}^{4}}\]
done
clear
C)
both (a) and (b)
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 162) It is evident from Rutherfords experiments that:
A)
a-particles move with high velocity
done
clear
B)
most of the part of atom is hollow
done
clear
C)
a-particles are positively charged
done
clear
D)
a-particles are heavier than electron
done
clear
View Answer play_arrow
question_answer 163) Calcium carbide react with \[{{H}_{2}}O\] gives:
A)
ethane
done
clear
B)
\[CH\equiv CH\]
done
clear
C)
\[C{{H}_{2}}=C{{H}_{2}}\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 164) \[Acetylene\xrightarrow{NaN{{H}_{2}}}A\xrightarrow{Iodoform}\] \[B\xrightarrow{{{H}_{2}}O/H{{g}^{2+}}}Z\]is:
A)
\[C{{H}_{3}}COOH\]
done
clear
B)
\[C{{H}_{3}}COC{{H}_{3}}\]
done
clear
C)
\[HCOOH\]
done
clear
D)
\[{{C}_{2}}{{H}_{5}}OH\]
done
clear
View Answer play_arrow
question_answer 165) From the following molecules or ions which acts as Lewis acid and Lewis base:
A)
\[{{H}_{2}}O\]
done
clear
B)
\[S{{O}_{2}}^{-2}\]
done
clear
C)
\[SO_{4}^{-2}\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 166) The hydrogen bond is not present in:
A)
liquid \[N{{H}_{3}}\]
done
clear
B)
phenol
done
clear
C)
liquid \[HCl\]
done
clear
D)
water
done
clear
View Answer play_arrow
question_answer 167) Elements which shows variable valencies are:
A)
s-block element
done
clear
B)
d-block element
done
clear
C)
p-block element
done
clear
D)
radioactive element
done
clear
View Answer play_arrow
question_answer 168) The compound having weakest bond angle is:
A)
\[Be{{F}_{2}}\]
done
clear
B)
\[N{{H}_{3}}\]
done
clear
C)
\[{{H}_{2}}O\]
done
clear
D)
\[C{{H}_{4}}\]
done
clear
View Answer play_arrow
question_answer 169) From the following compounds the soluble compound in water is:
A)
acetone
done
clear
B)
acetaldehyde
done
clear
C)
formaldehyde
done
clear
D)
all of these
done
clear
View Answer play_arrow
question_answer 170) Bond angle in \[s{{p}^{2}}\]hybridisation is:
A)
\[{{109}^{o}}45\]
done
clear
B)
\[{{180}^{o}}\]
done
clear
C)
\[{{90}^{o}}\]
done
clear
D)
\[{{120}^{o}}\]
done
clear
View Answer play_arrow
question_answer 171) Oxidative reagent is:
A)
proton donor
done
clear
B)
electron donor
done
clear
C)
proton accepter
done
clear
D)
electron accepter
done
clear
View Answer play_arrow
question_answer 172) Which is the correct order of bond length from the following?
A)
\[C\equiv C<C-C<C\equiv C\]
done
clear
B)
\[C-C<C=C<C\equiv C\]
done
clear
C)
\[C\equiv C<C=C<C-C\]
done
clear
D)
\[C=C<C\equiv C<C-C\]
done
clear
View Answer play_arrow
question_answer 173) The process of fermentation is:
A)
reversible
done
clear
B)
isothermic
done
clear
C)
exothermic
done
clear
D)
endofhermic
done
clear
View Answer play_arrow
question_answer 174) \[C{{H}_{3}}-\underset{OH}{\mathop{\underset{|}{\mathop{C}}\,}}\,H-C{{H}_{2}}-\underset{OH}{\mathop{\underset{|}{\mathop{\overset{C{{H}_{3}}}{\mathop{\overset{|}{\mathop{C}}\,}}\,}}\,}}\,-C{{H}_{3}}\] IUPAC name is:
A)
4-methyl-2, 4-pentane di-ol
done
clear
B)
2-methyl 2, 4-pentane di-ol
done
clear
C)
both above
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 175) The gas evolved in the fermentation process:
A)
\[{{N}_{2}}\]
done
clear
B)
\[{{O}_{2}}\]
done
clear
C)
\[S\]
done
clear
D)
\[C{{O}_{2}}\]
done
clear
View Answer play_arrow
question_answer 176) Green colour obtained in the Bunsens flame test is due to:
A)
\[C{{u}^{2+}}\]
done
clear
B)
\[{{K}^{+}}\]
done
clear
C)
\[N{{a}^{+}}\]
done
clear
D)
\[L{{i}^{+}}\]
done
clear
View Answer play_arrow
question_answer 177)
A)
aniline
done
clear
B)
benzaldehyde
done
clear
C)
nitrobenzene
done
clear
D)
anilium chloride
done
clear
View Answer play_arrow
question_answer 178) The ionization energy of \[{{N}_{2}}\] is greater than \[{{O}_{2}}\] the reason is:
A)
half filled p-orbital
done
clear
B)
penetration effect
done
clear
C)
more nucleus attraction
done
clear
D)
all of the above
done
clear
View Answer play_arrow
question_answer 179) Ionic compounds have not the property:
A)
high boiling point and melting point
done
clear
B)
soluble in polar solvents
done
clear
C)
rotation in direction
done
clear
D)
hard and brittle nature
done
clear
View Answer play_arrow
question_answer 180) The ratio of a bond n bond in benzene is:
A)
\[4\]
done
clear
B)
\[6\]
done
clear
C)
\[2\]
done
clear
D)
\[8\]
done
clear
View Answer play_arrow
question_answer 181) The percentage amount of \[{{N}_{3}}\] in urea is:
A)
\[46.66%\]
done
clear
B)
\[40%\]
done
clear
C)
\[23.23%\]
done
clear
D)
\[50%\]
done
clear
View Answer play_arrow
question_answer 182) At N.T.P. how much \[{{H}_{2}}S{{O}_{4}}\]is required to neutralize \[5g\]of ammonia?
A)
\[98.0g\]
done
clear
B)
\[21.97g\]
done
clear
C)
\[12.25g\]
done
clear
D)
\[10.94g\]
done
clear
View Answer play_arrow
question_answer 183) According to Bronsteds theory the base is:
A)
\[N{{H}_{4}}^{+}\]
done
clear
B)
\[C{{H}_{3}}COOH\]
done
clear
C)
\[C{{l}^{-}}\]
done
clear
D)
\[{{H}_{3}}{{O}^{+}}\]
done
clear
View Answer play_arrow
question_answer 184) Which one is the Lewis acid?
A)
\[{{H}_{2}}O\]
done
clear
B)
\[AlC{{l}_{3}}\]
done
clear
C)
\[RN{{H}_{2}}\]
done
clear
D)
\[N{{H}_{3}}\]
done
clear
View Answer play_arrow
question_answer 185) Effect of catalyst is:
A)
increase the rate of reaction
done
clear
B)
increase the quantity of product
done
clear
C)
both (a) and (b)
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 186) Isomer of chloropropane is:
A)
\[1\]
done
clear
B)
\[2\]
done
clear
C)
\[3\]
done
clear
D)
\[4\]
done
clear
View Answer play_arrow
question_answer 187) Main ore of lead is:
A)
galena
done
clear
B)
mica
done
clear
C)
bauxite
done
clear
D)
rin stone
done
clear
View Answer play_arrow
question_answer 188) \[C{{H}_{3}}COOH+PC{{l}_{3}}\xrightarrow{distillation}A\] product A is :
A)
\[C{{H}_{3}}COO{{C}_{2}}{{H}_{5}}\]
done
clear
B)
done
clear
C)
\[C{{H}_{3}}COOCl\]
done
clear
D)
\[C{{H}_{3}}COCl\]
done
clear
View Answer play_arrow
question_answer 189) Colourless ion is:
A)
\[C{{u}^{+3}}\]
done
clear
B)
\[F{{e}^{+3}}\]
done
clear
C)
\[C{{u}^{+}}\]
done
clear
D)
\[C{{u}^{+2}}\]
done
clear
View Answer play_arrow
question_answer 190) The pH value of \[0.01M\text{ }NaOH\]will be:
A)
\[12\]
done
clear
B)
\[14\]
done
clear
C)
\[13\]
done
clear
D)
\[10\]
done
clear
View Answer play_arrow
question_answer 191) \[A+{{I}_{2}}\xrightarrow{{}}\]iodine solution becomes colourless, A compound is:
A)
chloroform
done
clear
B)
ethelene
done
clear
C)
ethene
done
clear
D)
carbon tetra chloride
done
clear
View Answer play_arrow
question_answer 192) \[C{{H}_{3}}-C{{H}_{2}}-C{{H}_{3}}+HBr\xrightarrow{{}}A\], product A is:
A)
\[C{{H}_{3}}-CHB{{r}_{2}}-C{{H}_{3}}\]
done
clear
B)
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}Br\]
done
clear
C)
\[C{{H}_{3}}-CHBr-C{{H}_{3}}\]
done
clear
D)
\[C{{H}_{2}}Br-C{{H}_{2}}-C{{H}_{2}}Br\]
done
clear
View Answer play_arrow
question_answer 193) Which pair of the atomic numbers is not in the same group?
A)
\[11,19\]
done
clear
B)
\[5,13\]
done
clear
C)
\[4,12\]
done
clear
D)
\[6,16\]
done
clear
View Answer play_arrow
question_answer 194) Lime stone is:
A)
\[CaC{{O}_{3}}\]
done
clear
B)
\[CaO\]
done
clear
C)
both (a) and (b)
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 195) Linear molecule is:
A)
\[C{{O}_{2}}\]
done
clear
B)
\[S{{O}_{2}}\]
done
clear
C)
\[{{H}_{2}}O\]
done
clear
D)
\[C{{H}_{4}}\]
done
clear
View Answer play_arrow
question_answer 196) \[PC{{l}_{5}}\] is used in synthesis of:
A)
\[C{{H}_{3}}COCl\]
done
clear
B)
diethyl ether
done
clear
C)
ethyl alcohol
done
clear
D)
ace
done
clear
View Answer play_arrow
question_answer 197) Dental formula of Rabbit is:
A)
\[\left( \frac{2}{1},\frac{0}{0},\frac{3}{2},\frac{3}{3} \right)\times 2=28\]
done
clear
B)
\[\left( \frac{1}{1},\frac{0}{0},\frac{2}{2},\frac{3}{3} \right)\times 2=24\]
done
clear
C)
\[\left( \frac{2}{2},\frac{0}{0},\frac{3}{3} \right)\times 2=20\]
done
clear
D)
0
done
clear
View Answer play_arrow
question_answer 198) Number of limbs found in class Insecta is:
A)
numerous
done
clear
B)
1 pair
done
clear
C)
3 pairs
done
clear
D)
6 pairs
done
clear
View Answer play_arrow
question_answer 199) Specific character of Rodentia is the:
A)
presence of claw on toes
done
clear
B)
presence of hoof on limbs
done
clear
C)
presence of long root less canines
done
clear
D)
presence of long root less incisors
done
clear
View Answer play_arrow
question_answer 200) In cell cycle replication of DNA occurs in:
A)
interphase
done
clear
B)
\[{{G}_{2}}-\]phase
done
clear
C)
\[{{G}_{1}}-\]phase
done
clear
D)
S-phase
done
clear
View Answer play_arrow
question_answer 201) Sternum of Rabbit is formed of:
A)
fibrous cartilage
done
clear
B)
elastic (flexible) cartilage
done
clear
C)
hyaline cartilage
done
clear
D)
calcified cartilage
done
clear
View Answer play_arrow
question_answer 202) Activity shown by Amoeba if kept in salt solution will be:
A)
chemotaxis
done
clear
B)
simple reaction
done
clear
C)
phototaxis
done
clear
D)
migmotaxis
done
clear
View Answer play_arrow
question_answer 203) Main reason for the higher metabolic rate in insect is :
A)
tubular heart
done
clear
B)
open circulatory system
done
clear
C)
omnivorous food habit
done
clear
D)
direct respiration
done
clear
View Answer play_arrow
question_answer 204) Quantitatively smallest egg is found in:
A)
Blue whale
done
clear
B)
Shark fish
done
clear
C)
Elephant
done
clear
D)
Whale
done
clear
View Answer play_arrow
question_answer 205) Which of the following is the example of structural protein?
A)
Myosin
done
clear
B)
Collagen
done
clear
C)
Keratin
done
clear
D)
All of these
done
clear
View Answer play_arrow
question_answer 206) Rabbit is a:
A)
coward animal
done
clear
B)
burrowing, colonial, nocturnal animal
done
clear
C)
burrowing, colonial, nocturnal and herbivorous animal
done
clear
D)
colonial, nocturnal and herbivorous animal
done
clear
View Answer play_arrow
question_answer 207) Find out the odd one:
A)
Whale
done
clear
B)
Sea horse
done
clear
C)
Chimera
done
clear
D)
Scoliodon
done
clear
View Answer play_arrow
question_answer 208) Pearl oyster is the member of phylum :
A)
Arthropoda
done
clear
B)
Mollusca
done
clear
C)
Echinodermata
done
clear
D)
Annelida
done
clear
View Answer play_arrow
question_answer 209) Main medium for the digestive activity in Amoeba is:
A)
only neutral
done
clear
B)
only alkaline
done
clear
C)
only addic
done
clear
D)
acidic than alkaline
done
clear
View Answer play_arrow
question_answer 210) In general fate of blastopore is :
A)
condyl
done
clear
B)
ear lobe
done
clear
C)
mouth
done
clear
D)
anus
done
clear
View Answer play_arrow
question_answer 211) Number of cranial nerves found in Rabbit:
A)
10 pairs
done
clear
B)
15 pairs
done
clear
C)
12 pairs
done
clear
D)
37 pairs
done
clear
View Answer play_arrow
question_answer 212) Infectious stage of Plasmodium is:
A)
ookinete
done
clear
B)
sporozoite
done
clear
C)
oocyst
done
clear
D)
gametocyts
done
clear
View Answer play_arrow
question_answer 213) Smallest unit of classification is :
A)
family
done
clear
B)
genus
done
clear
C)
species
done
clear
D)
phylum
done
clear
View Answer play_arrow
question_answer 214) Phylum, which has all members of marine habitat is :
A)
Mollusca
done
clear
B)
Echinodermata
done
clear
C)
Annelida
done
clear
D)
Arthropoda
done
clear
View Answer play_arrow
question_answer 215) In Rabbit sweat glands, in dense form are, present on:
A)
eyelids
done
clear
B)
whole skin
done
clear
C)
lips
done
clear
D)
basal part of eyelids
done
clear
View Answer play_arrow
question_answer 216) Jointed legs located on abdominal region is the specific character of class :
A)
Crustacea
done
clear
B)
Arachnida
done
clear
C)
Insecta
done
clear
D)
Diplopoda
done
clear
View Answer play_arrow
question_answer 217) Enzyme present in the saliva of Cockroach is:
A)
pepsin
done
clear
B)
lipase
done
clear
C)
amylase
done
clear
D)
lysozyme
done
clear
View Answer play_arrow
question_answer 218) On catalyzing, hetero-fatty acids gives out:
A)
\[\beta -\]keto acetyl CoA
done
clear
B)
L-methyl metonyl CoA
done
clear
C)
succinyl CoA
done
clear
D)
D methyl metonyl CoA
done
clear
View Answer play_arrow
question_answer 219) In which sub-stage of meiosis synapsis occurs?
A)
Diplotene
done
clear
B)
Leptotcne
done
clear
C)
Pachytene
done
clear
D)
Zygotene
done
clear
View Answer play_arrow
question_answer 220) Water boat Cockroach is referred for :
A)
Blatta germanica
done
clear
B)
Periplanta americana
done
clear
C)
Blatta orientalis
done
clear
D)
all of the above
done
clear
View Answer play_arrow
question_answer 221) Flight muscles in birds are connected with :
A)
corocoid
done
clear
B)
scapula
done
clear
C)
clavicle
done
clear
D)
keel of sternum
done
clear
View Answer play_arrow
question_answer 222) Chemically keratin is:
A)
mucoprotein
done
clear
B)
pheroprotein
done
clear
C)
lipoprotein
done
clear
D)
scleroprotein
done
clear
View Answer play_arrow
question_answer 223) m-RNA is connected with:
A)
mitochondria
done
clear
B)
endoplasmic reticulum
done
clear
C)
ribosome
done
clear
D)
nucleus
done
clear
View Answer play_arrow
question_answer 224) Importance of cleavage is in the:
A)
formation of presumptive infant
done
clear
B)
division of fertilized egg
done
clear
C)
formation of multicellular structure
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 225) Type of cleavage in ova is mainly dependent on:
A)
distribution of yolk
done
clear
B)
origin of spindles in mitosis
done
clear
C)
amount of yolk
done
clear
D)
all of the above
done
clear
View Answer play_arrow
question_answer 226) Nematoblast of Hydra are:
A)
sensory
done
clear
B)
complicated
done
clear
C)
with nematocyst apparatus
done
clear
D)
all of the above
done
clear
View Answer play_arrow
question_answer 227) On the basis of feeding. Hydra is:
A)
holozoic
done
clear
B)
herbivorous
done
clear
C)
carnivorous
done
clear
D)
omnivorous
done
clear
View Answer play_arrow
question_answer 228) Last recipient of electron transport system (ETS) is:
A)
\[NA{{D}^{+}}\]
done
clear
B)
\[NAD{{P}^{+}}\]
done
clear
C)
FAD
done
clear
D)
Ubiquinone (CoQ)
done
clear
View Answer play_arrow
question_answer 229) Pseudocoelom is found in :
A)
Taeitin
done
clear
B)
Ascaris
done
clear
C)
Hydra
done
clear
D)
all of these
done
clear
View Answer play_arrow
question_answer 230) State in which sudden rise in the number of leucocytes (WBC) of mammalian blood occurs is called as:
A)
leucoderma
done
clear
B)
orocucoidia
done
clear
C)
leukemia
done
clear
D)
polycythikia
done
clear
View Answer play_arrow
question_answer 231) Reptile with four chambered heart is:
A)
Snake
done
clear
B)
Crocodile
done
clear
C)
Lizard
done
clear
D)
All of these
done
clear
View Answer play_arrow
question_answer 232) Olfactory lobes of Rabbit are:
A)
solid lobes
done
clear
B)
partially solid lobes
done
clear
C)
hollow lobes
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 233) In Earthworm circular muscles are highly developed in:
A)
gizzard
done
clear
B)
gastric region
done
clear
C)
pharyngeal region
done
clear
D)
oesophagus region
done
clear
View Answer play_arrow
question_answer 234) State in which oxygen is supplied to tissues in insufficient amount, is known as:
A)
pleurasy
done
clear
B)
amphionia
done
clear
C)
hyperoxia
done
clear
D)
hypoxia
done
clear
View Answer play_arrow
question_answer 235) Oxyntic cells are found in:
A)
pyloric part of stomach
done
clear
B)
oesophageal part of stomach
done
clear
C)
fundic part of stomach
done
clear
D)
cardiac part of stomach
done
clear
View Answer play_arrow
question_answer 236) Specific character of amniota is the :
A)
presence of foetal membranes
done
clear
B)
birth of immature foetus
done
clear
C)
presence of cleodoic eggs
done
clear
D)
viviparity
done
clear
View Answer play_arrow
question_answer 237) Difference between birds and mammals is:
A)
modification of forelimb into wings
done
clear
B)
modification of forelimb
done
clear
C)
modification of forelimb into residuals
done
clear
D)
all of above modifications
done
clear
View Answer play_arrow
question_answer 238) Body temperature of Rabbit is:
A)
\[96{}^\circ F\]
done
clear
B)
\[99.8{}^\circ F\]
done
clear
C)
\[100{}^\circ F\]
done
clear
D)
\[103.1{}^\circ F\]
done
clear
View Answer play_arrow
question_answer 239) In Krebs cycle formation of:
A)
34 ATP takes place
done
clear
B)
38 ATP takes place
done
clear
C)
15 ATP from each acetyl CoA takes place
done
clear
D)
12 ATP from each acetyl CoA takes place
done
clear
View Answer play_arrow
question_answer 240) Formation of gonads in Hydra is accelerated by:
A)
cyclic AMP
done
clear
B)
excess of \[C{{O}_{2}}\]
done
clear
C)
lack of \[{{O}_{2}}\]
done
clear
D)
analine
done
clear
View Answer play_arrow
question_answer 241) Which of the structure in Hydra is functionally same as that of pseudopodia in A mocha?
A)
Mouth
done
clear
B)
Tentacles
done
clear
C)
Ectoderm
done
clear
D)
Nematoblast
done
clear
View Answer play_arrow
question_answer 242) Mitosis and meiosis both divisions occurs in:
A)
kidney
done
clear
B)
somatic cells
done
clear
C)
testis
done
clear
D)
germinal cells
done
clear
View Answer play_arrow
question_answer 243) Non-poisonous snake is:
A)
Azgar
done
clear
B)
marine snake
done
clear
C)
Bungarus
done
clear
D)
Viper
done
clear
View Answer play_arrow
question_answer 244) Which of the protozoan is responsible for kala azar disease?
A)
Trypanasoma
done
clear
B)
Ginrdia
done
clear
C)
Monocystis
done
clear
D)
Leishrnania
done
clear
View Answer play_arrow
question_answer 245) Central dogma is:
A)
DNA\[\to \] mRNA\[\to \] protein
done
clear
B)
DNA \[\to \] RNA \[\to \] protein
done
clear
C)
protein\[\to \]RNA\[\to \] DNA
done
clear
D)
RNA \[\to \] DNA \[\to \]protein
done
clear
View Answer play_arrow
question_answer 246) Infectious stage of Ascaris for man is:
A)
adult
done
clear
B)
shelled egg
done
clear
C)
second larval stage
done
clear
D)
first larval stage
done
clear
View Answer play_arrow
question_answer 247) Sound producing organ in birds is:
A)
vocal fibres
done
clear
B)
larynx
done
clear
C)
epiglotis
done
clear
D)
syrinx
done
clear
View Answer play_arrow
question_answer 248) Infective stage of Ascaris is :
A)
embryonated eggs
done
clear
B)
fertilized eggs
done
clear
C)
unfertilized eggs
done
clear
D)
all of above
done
clear
View Answer play_arrow
question_answer 249) Double gradient theory was given by:
A)
Childe
done
clear
B)
Herstedius
done
clear
C)
Rocks
done
clear
D)
Hens Drish
done
clear
View Answer play_arrow
question_answer 250) For which of the following physiological activity sodium ions play important role?
A)
Conduction of nervous impulse
done
clear
B)
Diffusion process
done
clear
C)
Osmosis process
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 251) Specific cells present in phylum Porifera are called :
A)
choanocytes
done
clear
B)
porocytes
done
clear
C)
epidermal cells
done
clear
D)
all of these
done
clear
View Answer play_arrow
question_answer 252) In which cell first reductional division occurs during sperm a to genesis?
A)
Primary spermatocytes
done
clear
B)
Secondary spermatocytes
done
clear
C)
Spermatids
done
clear
D)
Spermatogonia
done
clear
View Answer play_arrow
question_answer 253) Which of the following is an essential amino acid?
A)
Tyrosine
done
clear
B)
Leucine
done
clear
C)
Alanine
done
clear
D)
Glycine
done
clear
View Answer play_arrow
question_answer 254) Rolling of the cells on their axis is called
A)
in vagination
done
clear
B)
divergence
done
clear
C)
convergence
done
clear
D)
splitting
done
clear
View Answer play_arrow
question_answer 255) Which of the following structure present in the mesogloea of Hydra?
A)
Non-fibrous structures
done
clear
B)
Acellular structures
done
clear
C)
Nerve plexus
done
clear
D)
Discoidals
done
clear
View Answer play_arrow
question_answer 256) Function of corpora allata hormone is:
A)
to secrete gonadotropic hormone
done
clear
B)
the oviposition and oogenesis
done
clear
C)
to develop gonads
done
clear
D)
all of the above
done
clear
View Answer play_arrow
question_answer 257) Endoparasite of Earthworm is:
A)
Euglena
done
clear
B)
Monocystis
done
clear
C)
Polystemella
done
clear
D)
Amoeba
done
clear
View Answer play_arrow
question_answer 258) Heparin is:
A)
phosphoprotein
done
clear
B)
glycoprotein
done
clear
C)
lipoprotein
done
clear
D)
chromoprotein
done
clear
View Answer play_arrow
question_answer 259) Example of hair Iess mammal is:
A)
Seal
done
clear
B)
Whale and Dolphin
done
clear
C)
Whale only
done
clear
D)
all of the above
done
clear
View Answer play_arrow
question_answer 260) Metamorphosis is the specific character of:
A)
sub phylum Urochordata
done
clear
B)
class Thaliacea
done
clear
C)
class Larvacea
done
clear
D)
class Ascidiacea
done
clear
View Answer play_arrow
question_answer 261) Limb less amphibian is :
A)
Pipa
done
clear
B)
Siren
done
clear
C)
lcthyophis
done
clear
D)
Proteus
done
clear
View Answer play_arrow
question_answer 262) Place from where sperms penetrates and enters in the ova, is determined by :
A)
microvilli
done
clear
B)
grey present
done
clear
C)
micropyle
done
clear
D)
all of these
done
clear
View Answer play_arrow
question_answer 263) If a fresh water Amoeba is kept in distil water Petridis, work rate of contractile vacuole will:
A)
remain unaffected
done
clear
B)
reduce
done
clear
C)
decline very fast
done
clear
D)
increase very fast
done
clear
View Answer play_arrow
question_answer 264) Totipotent cells in Hydra is referred for:
A)
sensory cells
done
clear
B)
nematoblast
done
clear
C)
germinal cells
done
clear
D)
interstitial cells
done
clear
View Answer play_arrow
question_answer 265) Total volume of air circulated in inspiration and expiration by full effort of Rabbit is;
A)
500 ml
done
clear
B)
1, 100 ml
done
clear
C)
3, 000 ml
done
clear
D)
5, 00 ml
done
clear
View Answer play_arrow
question_answer 266) True coelom appeared in phylum:
A)
Arthropoda
done
clear
B)
Mollusca
done
clear
C)
Porifera
done
clear
D)
Annelida
done
clear
View Answer play_arrow
question_answer 267) Presence of rhabditiform larva is me specific character of:
A)
Earthworm
done
clear
B)
Hydra
done
clear
C)
Ascaris
done
clear
D)
Rabbit
done
clear
View Answer play_arrow
question_answer 268) Largest source of enzyme arginase is:
A)
liver of Reptiles
done
clear
B)
liver of Telcost fishes
done
clear
C)
liver of Elasmobranch fishes
done
clear
D)
liver of Mammals
done
clear
View Answer play_arrow
question_answer 269) Which of the following is viviparous?
A)
Scoliodon
done
clear
B)
Star fish
done
clear
C)
Mudpuppy
done
clear
D)
Dipnoi
done
clear
View Answer play_arrow
question_answer 270) Hardest part in animal body is :
A)
bone
done
clear
B)
hair
done
clear
C)
dentine
done
clear
D)
enamel
done
clear
View Answer play_arrow
question_answer 271) Head of Earthworm is located on the :
A)
sub-segment of first segment
done
clear
B)
first segment
done
clear
C)
second segment
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 272) Cell organelle responsible for digestion of internal and external particles is;
A)
auto lysosomo
done
clear
B)
lysosome
done
clear
C)
residual body
done
clear
D)
all of these
done
clear
View Answer play_arrow
question_answer 273) In which direction cristae of Rabbit ear helps in maintaining balance :
A)
circular position of longitudinal axis of semi circular canals
done
clear
B)
transverse position of longitudinal axis of semicircular canals
done
clear
C)
parallel to longitudinal axis of semicircular canals
done
clear
D)
all of the above
done
clear
View Answer play_arrow
question_answer 274) In sperm centriole is located below the:
A)
nucleus
done
clear
B)
acrosome
done
clear
C)
head
done
clear
D)
alt of these
done
clear
View Answer play_arrow
question_answer 275) DNA and RNA has similarity in :
A)
base pairs
done
clear
B)
Replication property
done
clear
C)
Heredity property
done
clear
D)
duplication method
done
clear
View Answer play_arrow
question_answer 276) Sensory centre of hunger is located in :
A)
forebrain
done
clear
B)
mid brain
done
clear
C)
hind brain
done
clear
D)
hypothalamus
done
clear
View Answer play_arrow
question_answer 277) Main difference between the septal and pharyngeal nephridia is of:
A)
nephrostome
done
clear
B)
straight lobes
done
clear
C)
function
done
clear
D)
reservoir
done
clear
View Answer play_arrow
question_answer 278) Cockroach and Earthworm have similarity in:
A)
external and internal segmentation
done
clear
B)
external segmentation
done
clear
C)
internal segmentation
done
clear
D)
pseudo segmentation
done
clear
View Answer play_arrow
question_answer 279) Structure of sperm of Ascaris is :
A)
amoeboid
done
clear
B)
tail less and spindle shaped
done
clear
C)
ovate
done
clear
D)
tail less and amoeboid
done
clear
View Answer play_arrow
question_answer 280) Function of thyroxin hormone is :
A)
to grow
done
clear
B)
to develop
done
clear
C)
self immunization
done
clear
D)
to control metabolism
done
clear
View Answer play_arrow
question_answer 281) Theory of organizer was given by:
A)
Spemann
done
clear
B)
Leuwonhock
done
clear
C)
Ernst Von Baer
done
clear
D)
Wolf
done
clear
View Answer play_arrow
question_answer 282) Centrum of mammalian vertebrae is :
A)
variocoelus
done
clear
B)
procoelus
done
clear
C)
amphicoelus
done
clear
D)
acoelus
done
clear
View Answer play_arrow
question_answer 283) Which cells are responsible for secretion of peritrophic membrane in Cockroach?
A)
Glandular cells of salivary gland
done
clear
B)
Oesophageal part of stomach
done
clear
C)
Cells of internal layer of hindgut
done
clear
D)
Anterior cells of midgut
done
clear
View Answer play_arrow
question_answer 284) Reductional division in gametocytes of Plasmodium occurs in:
A)
nucleus of zygote
done
clear
B)
nucleus of oocyst
done
clear
C)
nucleus of ookinete
done
clear
D)
in all above stages
done
clear
View Answer play_arrow
question_answer 285) Which fish is helpful in biotic control of malaria?
A)
Minnose
done
clear
B)
Gambusia
done
clear
C)
Trout
done
clear
D)
All of these
done
clear
View Answer play_arrow
question_answer 286) Coronary/thebasian valve is located at:
A)
Post canal
done
clear
B)
right pre canal
done
clear
C)
left pre canal
done
clear
D)
all of these
done
clear
View Answer play_arrow
question_answer 287) Deltoid process is the specific character of:
A)
humerus
done
clear
B)
tibia
done
clear
C)
radio-ulna
done
clear
D)
femur
done
clear
View Answer play_arrow
question_answer 288) Gastrutation means:
A)
organogenesis movement
done
clear
B)
disappearance of blastocoel
done
clear
C)
formation of archenteron
done
clear
D)
all of the above
done
clear
View Answer play_arrow
question_answer 289) Important muscles in the respiratory system of Rabbit is:
A)
internal in tercostal muscles
done
clear
B)
external in tercostal muscles
done
clear
C)
diaphragm muscles
done
clear
D)
pectoralis major muscles
done
clear
View Answer play_arrow
question_answer 290) Specific character of Amphibia is:
A)
heterothermic nature
done
clear
B)
presence of movable eyelids
done
clear
C)
being first vertebral animal
done
clear
D)
amphibious nature
done
clear
View Answer play_arrow
question_answer 291) Function of porphyrin pigment in Earthworm is :
A)
coloration
done
clear
B)
excretion
done
clear
C)
secretion
done
clear
D)
to save the skin from the harmful effect of light
done
clear
View Answer play_arrow
question_answer 292) The number of stamens present in Cruciferae family are:
A)
4
done
clear
B)
7
done
clear
C)
2
done
clear
D)
6
done
clear
View Answer play_arrow
question_answer 293) The phase of cell division hi which DNA synthesis take place is:
A)
interphase
done
clear
B)
leptotene
done
clear
C)
diplotene
done
clear
D)
zygotene
done
clear
View Answer play_arrow
question_answer 294) The cell wall of fungus is made up of:
A)
chitin and cellulose
done
clear
B)
protein
done
clear
C)
lipid
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 295) Pesticide 2, 4 D is a:
A)
auxin hormone
done
clear
B)
enzyme
done
clear
C)
vitamin
done
clear
D)
plasmid
done
clear
View Answer play_arrow
question_answer 296) Transpiration occurs maximum when:
A)
high humidity present in atmosphere
done
clear
B)
high water present in soil
done
clear
C)
dry environment with high temperature
done
clear
D)
wind is not blowing
done
clear
View Answer play_arrow
question_answer 297) The unit of classification is:
A)
species
done
clear
B)
genera
done
clear
C)
both (a) and (b)
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 298) Drug quinine is used for:
A)
jaundice
done
clear
B)
malaria
done
clear
C)
dysentry
done
clear
D)
all of the above
done
clear
View Answer play_arrow
question_answer 299) Which of the following receive last electron in electron transport system?
A)
cytochrome a and \[{{a}_{3}}\]
done
clear
B)
oxygen
done
clear
C)
cytochrome-c
done
clear
D)
ubiquinine
done
clear
View Answer play_arrow
question_answer 300) Monadelphous condition is associated with:
A)
stamen
done
clear
B)
epiacalyx
done
clear
C)
corolla
done
clear
D)
gynoecium
done
clear
View Answer play_arrow
question_answer 301) Atropa belladonna is the member of family:
A)
Liliaceae
done
clear
B)
Solanaceae
done
clear
C)
Malvaceae
done
clear
D)
Cucurbitaceae
done
clear
View Answer play_arrow
question_answer 302) In pea yellow colour (\[\text{Y}\]) is dominant over green (\[y\]) and round (\[S\]) seed over wrinkled (\[s\]). If \[YySs\] seeds are formed in F1 by cross of \[\text{YYSS}\] and \[\text{yyss}\]. Then what will be the ratio of F2 if F1 plant is self-pollinated?
A)
9 yellow round: 3 yellow wrinkled : 3 green round : 1 green wrinkled.
done
clear
B)
13 yellow round: 3 green round.
done
clear
C)
9 yellow round: 3 green round.
done
clear
D)
all of the above
done
clear
View Answer play_arrow
question_answer 303) Agrostology is the study of:
A)
grasses
done
clear
B)
plants of hilly area
done
clear
C)
epiphytic plants
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 304) Process of eating and being eaten is a:
A)
food chain
done
clear
B)
biological process
done
clear
C)
producer-consumer relation
done
clear
D)
all of the above
done
clear
View Answer play_arrow
question_answer 305) The genome of bacteria is known as:
A)
nucleolus
done
clear
B)
nucleoid
done
clear
C)
nucleus
done
clear
D)
nucleotides
done
clear
View Answer play_arrow
question_answer 306) \[{{Q}_{10}}\] is a:
A)
temperature quotient
done
clear
B)
respiration quotient
done
clear
C)
measure of perfection
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 307) Hydrogen bond present in water between:
A)
H-O-H
done
clear
B)
H and H
done
clear
C)
O and O
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 308) NADP means:
A)
nicotinamide adenine dinucleotide phosphate
done
clear
B)
nicotinamide dinucleotide
done
clear
C)
nicotinic acid
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 309) The reason for Bhopal gas tragedy was :
A)
carbon dioxide gas
done
clear
B)
oxygen gas
done
clear
C)
potassium thiocyanate
done
clear
D)
methyle isocynate (MIC)
done
clear
View Answer play_arrow
question_answer 310) The RQ of organic acids will be:
A)
one
done
clear
B)
more than one
done
clear
C)
less than one
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 311) The sterile part of moss capsule is known as:
A)
protonema
done
clear
B)
foot
done
clear
C)
columella
done
clear
D)
prothallus
done
clear
View Answer play_arrow
question_answer 312) Pedology is the study of:
A)
disease
done
clear
B)
pollution
done
clear
C)
heredity
done
clear
D)
soil
done
clear
View Answer play_arrow
question_answer 313) Eugenics is related with:
A)
heredity
done
clear
B)
cytology
done
clear
C)
improvement in human beings
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 314) The sugar in ATP is:
A)
deoxy ribose
done
clear
B)
ribose
done
clear
C)
glucose
done
clear
D)
fructose
done
clear
View Answer play_arrow
question_answer 315) ATP contains:
A)
adenine, ribose and three phosphate groups
done
clear
B)
adenine and phosphate
done
clear
C)
only ribose sugar
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 316) The vascular cryptogames are:
A)
bryophytes
done
clear
B)
pteridophytes
done
clear
C)
monocots
done
clear
D)
angiospenns
done
clear
View Answer play_arrow
question_answer 317) Match the following : (A) (B) (A) Glycolysis (a) Mitochondria (B) Krebs cycle (b) \[{{F}_{1}}\] particle (C) Electron transport (c) cytoplasm system
A)
A (b), B (a), C (c)
done
clear
B)
A (c), B (a), C (b)
done
clear
C)
A (a), B (b) C (c)
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 318) Which of the following is growth inhibitor ?
A)
Abscissic acid
done
clear
B)
Gibberellins
done
clear
C)
Auxins
done
clear
D)
Ethylen
done
clear
View Answer play_arrow
question_answer 319) Oxalis shows:
A)
nastic movement
done
clear
B)
tropic movement
done
clear
C)
turgor movement
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 320) Calvin cycle is found in:
A)
chloroplast
done
clear
B)
mitochondria
done
clear
C)
cytoplasm
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 321) Who crystallize the plant virus?
A)
W. M. Stanley
done
clear
B)
Mendel
done
clear
C)
A. Jenner
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 322) The three scientists who rediscovering the Mendels law were?
A)
Hugo de Vries, Carl Correns and Tschermak
done
clear
B)
de Vries, Correns, T. H. Morgan
done
clear
C)
both (a) and (b)
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 323) Casparian strip is found in:
A)
vascular bundle
done
clear
B)
between stem and root
done
clear
C)
radial and inner wall of endodermis
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 324) Which of the following plant is used in the experiment of photosynthes is?
A)
Hydrilla
done
clear
B)
Mango
done
clear
C)
Rose
done
clear
D)
Neem
done
clear
View Answer play_arrow
question_answer 325) A membranous cell orgnnelle is:
A)
lysosome
done
clear
B)
ribosome
done
clear
C)
mitochondria
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 326) The main constituent of bacterial cell wall is:
A)
peptidoglycan
done
clear
B)
cellulose
done
clear
C)
chitin
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 327) Ulothrix belongs to class:
A)
Rodophyceae
done
clear
B)
Chlorophyceae
done
clear
C)
blue-green algae
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 328) The percentage of \[C{{O}_{2}}\] in atmosphere is:
A)
30%
done
clear
B)
0.3%
done
clear
C)
0.03%
done
clear
D)
3%
done
clear
View Answer play_arrow
question_answer 329) Which of the following is an auxin?
A)
Indole acetic acid
done
clear
B)
Malic acid
done
clear
C)
Pyruvic acid
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 330) Which part is edible in litchi?
A)
Pericarp
done
clear
B)
Embryo
done
clear
C)
Aril
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 331) Which of the following is not the characteristic of gyninosperm?
A)
Triple fusion
done
clear
B)
Absence of vessels
done
clear
C)
Both (a) and (b)
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 332) Which of the following is not represent an aquatic plant?
A)
Eichhornia
done
clear
B)
Wolffia
done
clear
C)
Nepenthes
done
clear
D)
Vallisnaria
done
clear
View Answer play_arrow
question_answer 333) Dominant phase in bryophytes is:
A)
gametophytic phase
done
clear
B)
sporophytic phase
done
clear
C)
both (a) and (b)
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 334) Synapsis is found in:
A)
leptotene phase
done
clear
B)
zygotene phase
done
clear
C)
pachytene phase
done
clear
D)
diplotene phase
done
clear
View Answer play_arrow
question_answer 335) Edible part of Cauliflower is:
A)
in florescence
done
clear
B)
flower
done
clear
C)
leaf
done
clear
D)
stem
done
clear
View Answer play_arrow
question_answer 336) The algae has seen green due to:
A)
bacterio-chlorophyll
done
clear
B)
chlorophyll
done
clear
C)
carotene
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 337) Cyclosis movement is also known as:
A)
autonomous movements
done
clear
B)
photonastic movements
done
clear
C)
movement of curvature
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 338) Pyoo is a type of:
A)
carotene
done
clear
B)
chlorophyll
done
clear
C)
phycobillin
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 339) Sunken stomata is the characteristic of:
A)
halophytes
done
clear
B)
xerophytes
done
clear
C)
mesophytes
done
clear
D)
hydrophytes
done
clear
View Answer play_arrow
question_answer 340) Lichens are the good example of:
A)
symbiotic relationship
done
clear
B)
parasitism
done
clear
C)
paredator and prey relationship
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 341) Svedbergs units ribosomes in prokaryotic and eukaryotic cell are :
A)
70S and 80S
done
clear
B)
50S and 30S
done
clear
C)
60S and 100S
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 342) The nucleic acid is absent in:
A)
nucleus
done
clear
B)
mitochondria
done
clear
C)
chloroplast
done
clear
D)
peroxysomes
done
clear
View Answer play_arrow
question_answer 343) Two different concentration solutions are separated by a semipermeable membrane which statement is correct?
A)
Solute will move towards solutions of higher concentration
done
clear
B)
Solvent will move towards solution of higher concentration
done
clear
C)
Both solute and solution will move towards higher concentration
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 344) Cork cambium is also known as:
A)
phelloderm
done
clear
B)
phellam
done
clear
C)
phallogen
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 345) Which of the following is not a peteridophyte ?
A)
Azolla
done
clear
B)
Lilium
done
clear
C)
Selaginella
done
clear
D)
Lycopodium
done
clear
View Answer play_arrow
question_answer 346) The pigment which is found in all algae:
A)
chlorophyll-a
done
clear
B)
chlorophyll-b
done
clear
C)
carotenoid
done
clear
D)
xanthophylls
done
clear
View Answer play_arrow
question_answer 347) Who discovered the glyoxysome?
A)
Jenssen and Jenssen
done
clear
B)
Tolbert
done
clear
C)
Beevers
done
clear
D)
Purkinje
done
clear
View Answer play_arrow
question_answer 348) In which plant stomata opens in night and closes in day?
A)
Succulent plants
done
clear
B)
Hydrophytes
done
clear
C)
Mesophytes
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 349) In tree ecosystem, pyramid of numbers is:
A)
upright
done
clear
B)
inverted
done
clear
C)
horizontal
done
clear
D)
all of the above
done
clear
View Answer play_arrow
question_answer 350) Which is present in cell wall of gram (+) and gram (-) bacteria?
A)
Murien
done
clear
B)
Cellulose
done
clear
C)
Lipoprotein
done
clear
D)
Only protein
done
clear
View Answer play_arrow
question_answer 351) Floral formula \[Br\oplus \,\underset{+}{\overset{\uparrow }{\mathop{\text{o}}}}\,{{K}_{(5)}}{{\overset\frown{{{C}_{(5)}}A}}_{(5)}}\,{{G}_{\underline{(2)}}}\] is concerned with:
A)
Malvaceae
done
clear
B)
Solanaceae
done
clear
C)
Papilionaceae
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 352) Which is diploid in Riccia?
A)
Spore mother cell
done
clear
B)
Spore
done
clear
C)
Gamete
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 353) Special structures between cell wall and plasma membrane of fungus are :
A)
plasmodesmata
done
clear
B)
lomasomes
done
clear
C)
plasmid
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 354) Pigments which found in all algae are?
A)
Chlorophyll-a and \[\beta \]-carofrene
done
clear
B)
Chlorophyll-a and \[\alpha \]-carotene
done
clear
C)
Chlorophyll-b and \[\beta \]-carotene
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 355) World population day is celebrated on:
A)
1-March
done
clear
B)
1-July
done
clear
C)
1-May
done
clear
D)
1-June
done
clear
View Answer play_arrow
question_answer 356) The alkaloids obtained from opium are:
A)
morphine, codeine, thebaine, narceine
done
clear
B)
thebaine, caffeine, nicoteine
done
clear
C)
cytokinin, gibberelline, pyrethrum
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 357) Ratio of recessive to dominant characters will be in F2 of mono hybrid cross:
A)
9 : 3 : 3 : 1
done
clear
B)
3 : 1
done
clear
C)
1 : 2 : 1
done
clear
D)
1 : 1
done
clear
View Answer play_arrow
question_answer 358) What will be the ratio of AaBb if AABB is crossed with aabb:
A)
8/16
done
clear
B)
7/16
done
clear
C)
4/16
done
clear
D)
3/16
done
clear
View Answer play_arrow
question_answer 359) Albugo is a:
A)
obligate parasite
done
clear
B)
facultative parasite
done
clear
C)
symbiotic
done
clear
D)
facultative saprophyte
done
clear
View Answer play_arrow
question_answer 360) In pea yellow colour (\[\text{Y}\]) is dominant over green (\[y\]) and round (\[\text{S}\]) seed is dominant over wrinkled (\[s\]). If \[\text{YySs}\] forms in F1 by cross of YYSS and \[yyss\] what will be the ratio of F2 when F1 plant is self-pollinated?
A)
9 yellow round : 3 yellow wrinkled:3 green round : 1 green wrinkled
done
clear
B)
9 yellow round : 7 yellow green
done
clear
C)
15 yellow round : 1 wrinkled green
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 361) Who discovered the syngamy in angiosperm?
A)
Nawaschin
done
clear
B)
Strassburger
done
clear
C)
Coulter and Chamberlin
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 362) Characters of pea like, pod colour, seed colour of flower etc. in Mendels experiment are known as:
A)
phenotype
done
clear
B)
genotype
done
clear
C)
all of the above
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 363) If temperature is increased \[10{}^\circ C\] the rate of dark reaction of photosynthesis will become:
A)
double
done
clear
B)
triple
done
clear
C)
four times
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 364) The floral formula of Solanaceae family is:
A)
\[{{K}_{(5)\,}}{{\overset\frown{{{C}_{(5)}}\,A}}_{(5)}}\,{{G}_{(\underline{2})}}\]
done
clear
B)
\[{{P}_{3\,}}{{K}_{5}}\,{{C}_{5}}{{A}_{3}}\,{{G}_{(\underline{2})}}\]
done
clear
C)
\[{{P}_{3\,}}{{K}_{3}}\,{{A}_{3}}\,{{G}_{(3)}}\]
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 365) Peroxysome is concerned with:
A)
photo respiration
done
clear
B)
transpiration
done
clear
C)
fungus
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 366) What will happen if \[{{O}_{2}}\] cone increased in environment?
A)
Photosynthesis will increase in \[{{C}_{3}}\] plants
done
clear
B)
Photosynthesis will decrease in \[{{C}_{3}}\] plants
done
clear
C)
Photosynthesis will increase in \[{{C}_{4}}\] plants
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 367) In bicollateral vascular bundle the tissues found from out to inner side will be :
A)
outer phloem, outer cambium, xylem, inner cambium and inner phloem
done
clear
B)
outer phloem, inner phloem, outer cambium, inner cambium, xylem
done
clear
C)
all of the above
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 368) Which of the following is wrong statement?
A)
Water is not necessary for fertilization of Albugo
done
clear
B)
Water required in fertilization of Albugo
done
clear
C)
Heterogamous sexual reproduction is found
done
clear
D)
A sexual reproduction takes place by conidia
done
clear
View Answer play_arrow
question_answer 369) The false statement for bryophyta is:
A)
they grow in shaddy places
done
clear
B)
vascular bundles are absent
done
clear
C)
gametophyte is not free
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 370) Dark reaction of photosynthesis takes place in:
A)
both light and dark
done
clear
B)
only in light
done
clear
C)
only in dark
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 371) Biosphere is consist of:
A)
decomposers
done
clear
B)
heterotroph
done
clear
C)
autotroph and producers
done
clear
D)
all of the above
done
clear
View Answer play_arrow
question_answer 372) Alkaloids obtained from Rauwolfia serpentine, are:
A)
serpentine, reserpine
done
clear
B)
atropine
done
clear
C)
cocaine
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 373) Sporophyte of Riccia is a spherical structure which is known as?
A)
Sporangium
done
clear
B)
Sporogonium
done
clear
C)
Capsule
done
clear
D)
Sporotheca
done
clear
View Answer play_arrow
question_answer 374) Bicollateral bundles means:
A)
they have phloem both on outer and inner side of xylem
done
clear
B)
they have xylem both on outer and inner side of phloem
done
clear
C)
both (a) and (b)
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 375) Mass flow hypothesis was given by:
A)
Munch
done
clear
B)
Mendel
done
clear
C)
Schleiden
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 376) The term ecosystem was coined by:
A)
Haeckel
done
clear
B)
Tansley
done
clear
C)
R. Mishra
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 377) Most of the morphine is used in the manufacturing of:
A)
morphine hydrochloride and morphine sulphate
done
clear
B)
opium tincture
done
clear
C)
opium powder
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 378) The trophic level of lion in forest is:
A)
\[{{T}_{3}}\]
done
clear
B)
\[{{T}_{4}}\]
done
clear
C)
\[{{T}_{2}}\]
done
clear
D)
\[{{T}_{1}}\]
done
clear
View Answer play_arrow
question_answer 379) Which ecosystem is most stable?
A)
Mountain
done
clear
B)
Desert
done
clear
C)
Sea
done
clear
D)
Forest
done
clear
View Answer play_arrow
question_answer 380) Codon which initiate polypeptide chain is:
A)
AUG and GUG
done
clear
B)
UAA
done
clear
C)
UGA
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 381) Barr body in women is consists of:
A)
inactive paternal \[X\]chromosome
done
clear
B)
inactive maternal \[X\] chromosome
done
clear
C)
both a and b
done
clear
D)
Inactive maternal or paternal\[X\]chromosome
done
clear
View Answer play_arrow
question_answer 382) Histone protein of nucleosome are:
A)
\[{{H}_{2}}A\] and \[{{H}_{2}}B\]
done
clear
B)
\[{{H}_{3}}\]
done
clear
C)
\[{{H}_{4}}\]
done
clear
D)
all of the above
done
clear
View Answer play_arrow
question_answer 383) Gossypium sp. (cotton) concerned with family:
A)
Malvaceae
done
clear
B)
Solanaceae
done
clear
C)
Crucifcrae
done
clear
D)
Cumpositae
done
clear
View Answer play_arrow
question_answer 384) Ostwald and Tippo has divided plant kingdom in to:
A)
two sub kingdoms
done
clear
B)
three sub kingdoms
done
clear
C)
four sub kingdoms
done
clear
D)
five sub kingdoms
done
clear
View Answer play_arrow
question_answer 385) Plants which best grow on acidic soil is called :
A)
halophyte
done
clear
B)
mesophyte
done
clear
C)
oxalophyte
done
clear
D)
xerophytes
done
clear
View Answer play_arrow
question_answer 386) The gases harmful or historical monuments are:
A)
\[N{{O}_{2}}\] and \[S{{P}_{2}}\]
done
clear
B)
\[{{O}_{2}}\] and \[{{N}_{2}}\]
done
clear
C)
C and \[{{N}_{2}}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 387) Water logging is found in:
A)
cpmcrete
done
clear
B)
clay soil
done
clear
C)
sandy soil
done
clear
D)
alluvial soil
done
clear
View Answer play_arrow
question_answer 388) Mesosomes arc analogous to:
A)
Chloroplast
done
clear
B)
mitochondria
done
clear
C)
lysosome
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 389) Who discovered the virus?
A)
Ivanowsky
done
clear
B)
W. M. Stanley
done
clear
C)
K. C. Mehta
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 390) Botanical name of Tea is:
A)
Camellia sinensis
done
clear
B)
Coffea Arabica
done
clear
C)
Theobroma cacao
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 391) Cystolith is made up of:
A)
calcium oxalate
done
clear
B)
calcium carbonate
done
clear
C)
magnesium carbonate
done
clear
D)
none of the above
done
clear
View Answer play_arrow
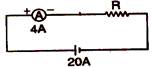
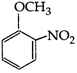
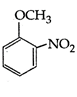 and
and 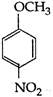
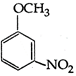
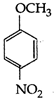
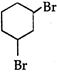
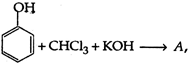 , product A is:
, product A is:
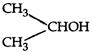
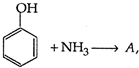 product A is:
product A is: