question_answer 1) Which nature of light is exposed by its diffraction and interference?
A)
Nature of light is electromagnetic
done
clear
B)
Wave nature of light
done
clear
C)
Quantum nature of light
done
clear
D)
Longitudinal nature of light
done
clear
View Answer play_arrow
question_answer 2) What is unit of magnetic permeability?
A)
\[\frac{\text{weber}}{\text{amper-metre}}\]
done
clear
B)
\[\frac{\text{weber}}{\text{metre}}\]
done
clear
C)
\[\frac{\text{tesla}}{\text{ampere}}\]
done
clear
D)
\[\frac{\text{tesla}}{\text{metre}}\]
done
clear
View Answer play_arrow
question_answer 3) Lowest wavelength of X-rays in continuous spectrum is:
A)
\[\frac{eV}{ch}\]
done
clear
B)
\[\frac{ch}{eV}\]
done
clear
C)
\[\frac{c}{heV}\]
done
clear
D)
\[\frac{hev}{c}\]
done
clear
View Answer play_arrow
question_answer 4) Which is not the unit of work out of these?
A)
watt-sec
done
clear
B)
N-m
done
clear
C)
kg-m/s
done
clear
D)
Joule
done
clear
View Answer play_arrow
question_answer 5) The moment of inertia and mass of hollow sphere and solid sphere is equal then what is ratio between their radius?
A)
1 : 2
done
clear
B)
\[\sqrt{3}:\sqrt{5}\]
done
clear
C)
\[\sqrt{5}:\sqrt{3}\]
done
clear
D)
5 : 4
done
clear
View Answer play_arrow
question_answer 6) In depletion layer which are present:
A)
only electron
done
clear
B)
only holes
done
clear
C)
electron and holes
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 7) Due to which oil rises up with the help of piece of cloth in lamp:
A)
viscosity of oil
done
clear
B)
capillary method
done
clear
C)
surface tension
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 8) In \[{{H}_{2}}\] molecule (atom) its total energy is proportional to:
A)
\[{{n}^{2}}\]
done
clear
B)
n
done
clear
C)
\[\frac{1}{n}\]
done
clear
D)
\[\frac{1}{{{n}^{2}}}\]
done
clear
View Answer play_arrow
question_answer 9) What will be the effect on potential barrier on decreasing forward voltage in p-n function diode:
A)
increase
done
clear
B)
decrease
done
clear
C)
no effect
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 10) Which is the best device to measure e.m.f. of a cell:
A)
Voltmeter
done
clear
B)
Voltmeter
done
clear
C)
Ammeter
done
clear
D)
Potentiometer
done
clear
View Answer play_arrow
question_answer 11) Which value cannot be measured by potentiometer?
A)
Resistance
done
clear
B)
Capacity
done
clear
C)
Electric current
done
clear
D)
Potential
done
clear
View Answer play_arrow
question_answer 12) If Fresnels biprism experiment is held in water in spite of air, then what will be the effect on fringe width :
A)
decrease
done
clear
B)
increase
done
clear
C)
no effect
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 13) Moment of inertia is minimum in cube when:
A)
about tangent
done
clear
B)
about axis
done
clear
C)
about centre of mass and passes through the axis
done
clear
D)
alt of these
done
clear
View Answer play_arrow
question_answer 14) What is the condition for lowest power defect in A.C. circuit?
A)
High resistane high inductance
done
clear
B)
High resistance low inductance
done
clear
C)
Low resistance high inductance
done
clear
D)
Low resistance and low inductance
done
clear
View Answer play_arrow
question_answer 15) X-rays are produced in laboratory by:
A)
radiation
done
clear
B)
decomposition of the atom
done
clear
C)
bombardment of high energy electron on heavy metal
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 16) Find out the true relation:
A)
\[{{e}_{\lambda }}{{a}_{\lambda }}={{E}_{\lambda }}\]
done
clear
B)
\[{{e}_{\lambda }}{{E}_{\lambda }}={{a}_{\lambda }}\]
done
clear
C)
\[{{e}_{\lambda }}={{a}_{\lambda }}{{E}_{\lambda }}\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 17) Kinetic energy of an ideal gas depends on its:
A)
temperature
done
clear
B)
pressure
done
clear
C)
volume
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 18) What should be in A.C. circuit to increase power factor?
A)
Capacitor
done
clear
B)
Chock coil
done
clear
C)
Low resistance with induction
done
clear
D)
All of these
done
clear
View Answer play_arrow
question_answer 19) What are the dimensions of surface tension:
A)
\[\text{ }\!\![\!\!\text{ }{{\text{M}}^{\text{1}}}{{\text{L}}^{\text{1}}}{{\text{T}}^{\text{-2}}}\text{ }\!\!]\!\!\text{ }\]
done
clear
B)
\[\text{ }\!\![\!\!\text{ }{{\text{M}}^{\text{1}}}{{\text{L}}^{0}}{{\text{T}}^{\text{-2}}}\text{ }\!\!]\!\!\text{ }\]
done
clear
C)
\[\text{ }\!\![\!\!\text{ }{{\text{M}}^{\text{-1}}}{{\text{L}}^{2}}{{\text{T}}^{\text{-1}}}\text{ }\!\!]\!\!\text{ }\]
done
clear
D)
\[\text{ }\!\![\!\!\text{ }{{\text{M}}^{\text{-1}}}{{\text{L}}^{2}}{{\text{T}}^{\text{-2}}}\text{ }\!\!]\!\!\text{ }\]
done
clear
View Answer play_arrow
question_answer 20) A particle moves along a circle with a uniform speed\[\upsilon .\] After it has made an angle of \[60{}^\circ \], then its speed will be:
A)
\[\upsilon \]
done
clear
B)
\[\frac{\upsilon }{\sqrt{2}}\]
done
clear
C)
\[\frac{\upsilon }{\sqrt{3}}\]
done
clear
D)
zero
done
clear
View Answer play_arrow
question_answer 21) The moment of inertia of a plate of mass m, length \[l\]and breadth b :
A)
\[m\left( \frac{{{l}^{2}}+{{b}^{2}}}{12} \right)\]
done
clear
B)
\[m\left( \frac{{{l}^{2}}}{4}+\frac{{{b}^{2}}}{12} \right)\]
done
clear
C)
\[m\left( \frac{{{l}^{2}}+{{b}^{2}}}{6} \right)\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 22) The displacement equation of a particle is \[x=3\,\sin \,2t\,+4\,\cos \,2t.\] The amplitude and maximum velocity will be respectively:
A)
5, 10
done
clear
B)
3, 2
done
clear
C)
4, 2
done
clear
D)
3, 4
done
clear
View Answer play_arrow
question_answer 23) One sphere collides with another sphere of same mass at rest in elastically. If the value of coefficients of restification is 1/2. Tine ratio of there speed after collision will be:
A)
1 : 2
done
clear
B)
2 : 1
done
clear
C)
1 : 3
done
clear
D)
3 : 1
done
clear
View Answer play_arrow
question_answer 24) If the kinetic energy of a body increase by 0.1% then the percentage increase in its momentum will be:
A)
0.05%
done
clear
B)
0.1%,
done
clear
C)
1.0%
done
clear
D)
10%
done
clear
View Answer play_arrow
question_answer 25) Out of these on which g does not depend:
A)
density
done
clear
B)
mass
done
clear
C)
shape
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 26) Angular velocity of a wheel is 2 rad/sec calculate the number of rotation of wheel in 5 seconds:
A)
5/\[\pi \]
done
clear
B)
1/\[\pi \]
done
clear
C)
10\[\pi \]
done
clear
D)
20\[\pi \]
done
clear
View Answer play_arrow
question_answer 27) How to adjust three capacitor to get high energy on same potential?
A)
Two parallel one in series
done
clear
B)
Three are in series
done
clear
C)
Three are in parallel
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 28) In the vaccum diode or valve oxide coated emitter are used because:
A)
the work function is more
done
clear
B)
they emits more electron at for temperature
done
clear
C)
work function is high
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 29) A disc whose radius R is revolving around its geometrical axis if we increase radius to 2R and constant speed then the centripetal force will be:
A)
half
done
clear
B)
double
done
clear
C)
equal
done
clear
D)
one fourth
done
clear
View Answer play_arrow
question_answer 30) A man standing on a revolving platform spreading his hands suddenly he stretches his hand. Then:
A)
angular velocity increase
done
clear
B)
angular velocity decrease
done
clear
C)
angular momentum increase
done
clear
D)
angular moment is zero
done
clear
View Answer play_arrow
question_answer 31) Calculate the number of \[\alpha \] and \[\] particles in radioactive decay\[_{92}{{X}^{234}}{{\xrightarrow{{}}}_{82}}{{Y}^{222}}\]:
A)
3, 3
done
clear
B)
3, 1
done
clear
C)
5, 3
done
clear
D)
3, 5
done
clear
View Answer play_arrow
question_answer 32) Which is not the dimensions of frequency?
A)
\[\frac{1}{\sqrt{LC}}\]
done
clear
B)
\[\frac{1}{RC}\]
done
clear
C)
\[\frac{C}{L}\]
done
clear
D)
\[\frac{R}{L}\]
done
clear
View Answer play_arrow
question_answer 33) Two equal and opposite charge are displaced x metre apart, then the potential depends upon:
A)
only on \[q\]
done
clear
B)
on \[x\]
done
clear
C)
on \[qx\]
done
clear
D)
on \[q/x\]
done
clear
View Answer play_arrow
question_answer 34) What is necessary condition to obtain interference pattern?
A)
Equal frequency
done
clear
B)
Equal amplitude
done
clear
C)
Constant phase difference
done
clear
D)
Equal amplitude and frequency
done
clear
View Answer play_arrow
question_answer 35) By one wire diffraction pattern is obtained, if we increase the diameter of wire then fringe width:
A)
increase
done
clear
B)
decrease
done
clear
C)
no change
done
clear
D)
all of these
done
clear
View Answer play_arrow
question_answer 36) Farad is not equal to:
A)
CV
done
clear
B)
\[\frac{{{\text{Q}}^{\text{2}}}}{\text{J}}\]
done
clear
C)
\[\frac{\text{J}}{{{\text{V}}^{\text{2}}}}\]
done
clear
D)
\[\text{QV}\]
done
clear
View Answer play_arrow
question_answer 37) Wave front is a imaginary surface, where:
A)
phase difference are equal
done
clear
B)
everywhere phase is equal
done
clear
C)
phase difference is different
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 38) In an experiment with a potentiometer it was found that no deflection is caused in the ameter on joining the two terminals with cells with 125 cm length of the potentiometer wire. If resistance of 2\[\Omega \]is connected to the terminal of cell, then balancing length is 100 cm, calculate the internal resistance of cell:
A)
\[0.5\,\Omega \]
done
clear
B)
\[1.5\,\Omega \]
done
clear
C)
\[1.25\,\Omega \]
done
clear
D)
\[\frac{4}{5}\,\Omega \]
done
clear
View Answer play_arrow
question_answer 39) Kinetic energy of emitted electrons in photoelectric effect does not depends on :
A)
wave length
done
clear
B)
frequency
done
clear
C)
intensity
done
clear
D)
work function
done
clear
View Answer play_arrow
question_answer 40) Neutrino hypothesis was given by:
A)
Sommerfeild
done
clear
B)
Pauli
done
clear
C)
Fermi
done
clear
D)
Chadwick
done
clear
View Answer play_arrow
question_answer 41) On charging a soap bouble its size:
A)
decreases
done
clear
B)
increases
done
clear
C)
remains same
done
clear
D)
cannot be said
done
clear
View Answer play_arrow
question_answer 42) Most accurate value is:
A)
\[{{C}_{P}}=3,C{{ }_{\upsilon }}=1\]
done
clear
B)
\[{{C}_{P}}=5,C{{ }_{\upsilon }}=3\]
done
clear
C)
\[{{C}_{P}}=4,C{{ }_{\upsilon }}=5\]
done
clear
D)
\[{{C}_{P}}=6,C{{ }_{\upsilon }}=2\]
done
clear
View Answer play_arrow
question_answer 43) In which type of wave energy is not transferred?
A)
Heat waves
done
clear
B)
Target waves
done
clear
C)
Stationary waves
done
clear
D)
Unstationary waves
done
clear
View Answer play_arrow
question_answer 44) If the absorb heat of system is \[\delta Q,\] work done by the system is \[\delta W,\] and change in internal energy is \[dU,\] then which quantity does not depend on path:
A)
\[dU\]
done
clear
B)
\[\delta W\]
done
clear
C)
\[\delta Q\]
done
clear
D)
\[\delta Q\,\text{and}\,\delta W\]
done
clear
View Answer play_arrow
question_answer 45) By which law, direction of induced e.m.f. can be found :
A)
Amperes law
done
clear
B)
Flemings law
done
clear
C)
Faradays law
done
clear
D)
Lenzs law
done
clear
View Answer play_arrow
question_answer 46) Half-life of a radioactive element is 30 days, then the remaining amount after 90 days:
A)
1/3
done
clear
B)
1/4
done
clear
C)
1/8
done
clear
D)
1/16
done
clear
View Answer play_arrow
question_answer 47) Which is not a possible electronic configuration?
A)
\[\text{1} {{\text{s}}^{\text{2}}}\text{,}\,\text{2}{{\text{s}}^{\text{2}}},\,\text{2}{{\text{p}}^{\text{5}}}\]
done
clear
B)
\[\text{1} {{\text{s}}^{\text{2}}}\text{,}\,{{\text{2}}_{\text{s}}}^{\text{2}}\text{,}\,\text{2}{{\text{p}}^{\text{6}}}\text{,}\,\text{3s}\]
done
clear
C)
\[\text{1} {{\text{s}}^{\text{2}}}\text{,}\,1{{\text{p}}^{\text{6}}}\text{,}\,2\text{s}\]
done
clear
D)
\[1{{s}^{2}},\,2{{s}^{2}},\,2{{p}^{6}},\,3{{s}^{2}}\]
done
clear
View Answer play_arrow
question_answer 48) What will be the relation between wavelength of photon and electron of same energy?
A)
\[{{}_{\text{Ph}}}\text{}\,{{}_{\text{e}}}\]
done
clear
B)
\[{{}_{\text{Ph}}}-\,{{}_{\text{e}}}\]
done
clear
C)
\[{{}_{e}}>\,{{}_{Ph}}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 49) A black body radiated high intensity radiation of 13\[\text{M}\]when temperature is \[200{}^\circ K\] if the temperature is increased upto \[1000\overset{\text{o}}{\mathop{\text{A}}}\,\]. Then calculate high intensity radiation:
A)
70 \[\text{M}\]
done
clear
B)
70 m
done
clear
C)
2.8 \[\text{M}\]
done
clear
D)
2.8 mm
done
clear
View Answer play_arrow
question_answer 50) When there will be a phase difference of \[180{}^\circ \] between incident and reflected rays:
A)
air to glass
done
clear
B)
glass to water
done
clear
C)
glass to air
done
clear
D)
water to air
done
clear
View Answer play_arrow
question_answer 51) The unit of dipole moment is:
A)
coulomb-metre
done
clear
B)
coulomb/metre
done
clear
C)
coulomb-metre2
done
clear
D)
metre/coulomb
done
clear
View Answer play_arrow
question_answer 52) Highest frequency waves are:
A)
microwave
done
clear
B)
infrared
done
clear
C)
ultrovoilet
done
clear
D)
X-rays
done
clear
View Answer play_arrow
question_answer 53) Frequency of an open organ pipe is\[f\]then it is dipped in water up to half of its length then new frequency will be:
A)
\[f\]
done
clear
B)
\[f\]/2
done
clear
C)
2\[f\]
done
clear
D)
\[\frac{3}{4}f\]
done
clear
View Answer play_arrow
question_answer 54) Radius of half period zone is proportional to:
A)
\[\sqrt{n}\]
done
clear
B)
\[\frac{1}{\sqrt{n}}\]
done
clear
C)
\[{{n}^{2}}\]
done
clear
D)
\[1/n\]
done
clear
View Answer play_arrow
question_answer 55) According to law of conservation of energy:
A)
energy can be destroyed cant be created
done
clear
B)
energy can neither be created nor be destroyed
done
clear
C)
energy can either be created or destroyed
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 56) What is the emissive power of sun when its temperature is doubled:
A)
2 times
done
clear
B)
4 times
done
clear
C)
8 times
done
clear
D)
16 times
done
clear
View Answer play_arrow
question_answer 57) In a diode on increasing plate voltage plate current is not increasing, then how to increase the plate current?
A)
Increase in filament current
done
clear
B)
Decrease in distance between cathode and plate
done
clear
C)
Increase the temperature of anode
done
clear
D)
Increase the temperature of cathode
done
clear
View Answer play_arrow
question_answer 58) In progressive wave transfer of energy per second on:
A)
maximum at antinode
done
clear
B)
maximum on antinode
done
clear
C)
maximum at node and but zero on antinode
done
clear
D)
maximum at antinode and zero on node
done
clear
View Answer play_arrow
question_answer 59) Phase difference between \[V\]and\[I\] in purely inductive A.C. circuit:
A)
\[0{}^\circ \]
done
clear
B)
\[180{}^\circ \]
done
clear
C)
\[90{}^\circ \]
done
clear
D)
\[360{}^\circ \]
done
clear
View Answer play_arrow
question_answer 60) Ratio between magnetic intensity and magnetic field intensity is:
A)
permeability
done
clear
B)
magnetic flux
done
clear
C)
magnetic nature
done
clear
D)
magnetic permeability
done
clear
View Answer play_arrow
question_answer 61) Behaviour of zone plate coincides with :
A)
concave lens
done
clear
B)
convex lens
done
clear
C)
concave mirror
done
clear
D)
convex mirror
done
clear
View Answer play_arrow
question_answer 62) A particle is executing S.H.M. its maximum velocity is\[{{\upsilon }_{0,}}\] then what will be its velocity at a distance half of the amplitude from mean position?
A)
\[\frac{{{\upsilon }_{0}}}{2}\]
done
clear
B)
\[{{\upsilon }_{0}}\]
done
clear
C)
\[{{\upsilon }_{0}}\sqrt{\frac{3}{2}}\]
done
clear
D)
\[{{\upsilon }_{0}}\sqrt{\frac{3}{2}}\]
done
clear
View Answer play_arrow
question_answer 63) Real gas behaves as ideal gas on:
A)
high temperature
done
clear
B)
low pressure
done
clear
C)
high temperature and low pressure
done
clear
D)
low temperature and high pressure
done
clear
View Answer play_arrow
question_answer 64) In a diode valve, mutual conductance is 25 mA /V and anode resistance is 20\[k\Omega \] is used as an amplifier. Whose amplification is 100, then calculate load resistance:
A)
\[1\,k\Omega \]
done
clear
B)
\[5\,k\Omega \]
done
clear
C)
\[10\,k\Omega \]
done
clear
D)
\[20\,k\Omega \]
done
clear
View Answer play_arrow
question_answer 65) White light is used in Youngs double slits experiment, distance between two slits is \[B(>>b),\]dispersion of interference film is small wavelength of one fringe then calculate wavelength of fringe :
A)
\[\frac{{{b}^{2}}}{B}\]
done
clear
B)
\[\frac{2{{b}^{2}}}{B}\]
done
clear
C)
\[\frac{{{b}^{2}}}{B}\frac{{{b}^{2}}}{3B}\]
done
clear
D)
all of these
done
clear
View Answer play_arrow
question_answer 66) Positive rays was discovered by:
A)
Thomson
done
clear
B)
Goldstein
done
clear
C)
W. Crookes
done
clear
D)
Rutherford
done
clear
View Answer play_arrow
question_answer 67) In Thomson mass spectrograph, electric and magnetic field are applied:
A)
perpendicularly together
done
clear
B)
perpendicularly
done
clear
C)
parallel
done
clear
D)
parallel together
done
clear
View Answer play_arrow
question_answer 68) Discovery of positive rays is helpful in discovery of:
A)
proton
done
clear
B)
isotopes
done
clear
C)
electrons
done
clear
D)
\[\text{-}\]particles
done
clear
View Answer play_arrow
question_answer 69) What is the effect on Fresnels biprism experiment when the use of white light is made:
A)
fringe are affected
done
clear
B)
diffraction pattern is spread ore
done
clear
C)
central fringe is white and all are coloured
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 70) Truth of chock coil is:
A)
high resistance, low inductance
done
clear
B)
high resistance, high inductance
done
clear
C)
low resistance, low inductance
done
clear
D)
low resistance, high inductance
done
clear
View Answer play_arrow
question_answer 71) Main frequency is 100 Hz and second two frequency are 300 Hz and 500 Hz, then the pipe is:
A)
open pipe
done
clear
B)
closed pipe
done
clear
C)
one end is open and second end is closed
done
clear
D)
all of these
done
clear
View Answer play_arrow
question_answer 72) The moment of inertia of a rod of 1 m length with respect to its mid point is:
A)
\[\frac{M}{12}\]
done
clear
B)
\[\frac{M}{3}\]
done
clear
C)
\[\frac{M}{2}\]
done
clear
D)
\[M\]
done
clear
View Answer play_arrow
question_answer 73) The frequency of oscillation of an object of mass in suspended by means of a spring of force constant k is given by\[f=\,(C{{m}^{x}}{{k}^{y}}),\] the value of x and y are:
A)
\[\frac{1}{2}\frac{1}{2}\]
done
clear
B)
\[-\frac{1}{2}\frac{1}{2}\]
done
clear
C)
\[-\frac{1}{2}\frac{1}{3}\]
done
clear
D)
\[\frac{1}{2}-\frac{1}{2}\]
done
clear
View Answer play_arrow
question_answer 74)
As shown in the figure half the space between plates of a capacitor is filled with an insulator material of dielectric constant\[K,\]if initial capacity was \[C\] then the new capacity is:
A)
\[\frac{C}{2}(K+1)\]
done
clear
B)
\[\frac{C}{2(K+1)}\]
done
clear
C)
\[\frac{(K+1)}{2C}\]
done
clear
D)
\[C\,(K+1)\]
done
clear
View Answer play_arrow
question_answer 75) Which of the following pair has identical dimensions?
A)
Momentum and force
done
clear
B)
Surface tension and surface energy
done
clear
C)
Pressure and Youngs coefficient
done
clear
D)
Momentum of force and angular momentum
done
clear
View Answer play_arrow
question_answer 76) The angular speed of a minute hand of a clock:
A)
\[\frac{}{\text{1600}}\text{rad/s}\]
done
clear
B)
\[\frac{}{\text{3000}}\text{rad/s}\]
done
clear
C)
\[\frac{}{\text{30}}\text{rad/s}\]
done
clear
D)
\[\frac{}{1800}\text{rad/s}\]
done
clear
View Answer play_arrow
question_answer 77) The ratio of mass of two circular ring is 1 : 2 and ratio of radius is 2 : 1 then what will be the, ratio of their momentums of inertia ?
A)
1 : 4
done
clear
B)
2 : 1
done
clear
C)
4 : 1
done
clear
D)
\[\sqrt{2}:1\]
done
clear
View Answer play_arrow
question_answer 78)
Force constant is k and elastic force is\[f,\] then what will be the displacement?
A)
\[\sqrt{3}\]
done
clear
B)
\[\frac{1}{\sqrt{3}}\]
done
clear
C)
\[\frac{1}{2}\]
done
clear
D)
\[\frac{\sqrt{3}}{2}\]
done
clear
View Answer play_arrow
question_answer 79) If the value of \[g\] on a planet is \[8\,m/{{s}^{2}}\] and its radius is 1000 km, then find the value of escape velocity on that planet:
A)
1.41 km/s
done
clear
B)
2.82 km/s
done
clear
C)
4km/s
done
clear
D)
8 km/s
done
clear
View Answer play_arrow
question_answer 80) Surface tension of a soap is \[2\times {{10}^{-2}}N/m,\] then calculate the work done to make a bubble of radius 1 cm:
A)
\[\text{4}\text{ }\!\!\times\!\!\text{ 1}{{\text{0}}^{\text{-6}}}\text{J}\]
done
clear
B)
\[\text{8}\text{ }\!\!\times\!\!\text{ 1}{{\text{0}}^{\text{-6}}}\text{J}\]
done
clear
C)
\[\text{12}\text{ }\!\!\times\!\!\text{ 1}{{\text{0}}^{\text{-6}}}\text{J}\]
done
clear
D)
\[\text{16}\text{ }\!\!\times\!\!\text{ 1}{{\text{0}}^{\text{-6}}}\text{J}\]
done
clear
View Answer play_arrow
question_answer 81) What should be the minimum velocity of body, at which it should be thrown to make it satellite of earth:
A)
\[11\times {{10}^{3}}\,m/s\]
done
clear
B)
\[8\times {{10}^{3}}\,m/s\]
done
clear
C)
\[6.4\,\times {{10}^{3}}\,m/s\]
done
clear
D)
\[4\times {{10}^{3}}\,m/s\]
done
clear
View Answer play_arrow
question_answer 82) In bin bridge spectrograph the electric field is \[{{10}^{4}}\] volt/m and magnetic field is 1 T, then the velocity of undeflected positive ions is:
A)
\[{{10}^{7}}\,m/s\]
done
clear
B)
\[{{10}^{4}}\,m/s\]
done
clear
C)
\[{{10}^{5}}\,m/s\]
done
clear
D)
\[{{10}^{2}}\,m/s\]
done
clear
View Answer play_arrow
question_answer 83) The equation of a wave is \[y={{10}^{-4}}\,\sin \,(60t+2x),\]where\[x\] and\[y\] are in metre,\[t\] in sec, then which statement is true?
A)
Wavelength is \[\pi \] meter
done
clear
B)
Frequency is \[\left( \frac{30}{\pi } \right)\]Hz
done
clear
C)
Velocity is 30 m/s
done
clear
D)
All of these
done
clear
View Answer play_arrow
question_answer 84) The number of lines of force from 1 coulomb charge is:
A)
0
done
clear
B)
1
done
clear
C)
\[\frac{1}{{{\varepsilon }_{0}}}\]
done
clear
D)
\[\infty \]
done
clear
View Answer play_arrow
question_answer 85) Which of the following is the one of iron?
A)
Galena
done
clear
B)
Bauxite
done
clear
C)
Hematite
done
clear
D)
Black zac
done
clear
View Answer play_arrow
question_answer 86) The oxidation number of iron in \[F{{e}_{0.94}}O\]is:
A)
\[+2\]
done
clear
B)
\[+2\] and \[+3\]
done
clear
C)
\[+3\]
done
clear
D)
\[+1\]
done
clear
View Answer play_arrow
question_answer 87) Which of the molecule or iron is coplanar?
A)
\[N{{O}_{3}}^{-}\]
done
clear
B)
\[C{{O}_{3}}^{-2}\]
done
clear
C)
\[S{{O}_{3}}^{-2}\]
done
clear
D)
\[S{{O}_{3}}\]
done
clear
View Answer play_arrow
question_answer 88) \[{{C}_{2}}{{H}_{5}}-O-{{C}_{2}}{{H}_{5}}\]on reaction with chlorine in the presence of light gives:
A)
\[C{{H}_{3}}COCl\]
done
clear
B)
\[{{({{C}_{2}}C{{l}_{5}})}_{2}}O\]
done
clear
C)
\[C{{H}_{3}}-Cl\]
done
clear
D)
all of these
done
clear
View Answer play_arrow
question_answer 89) The main ore o obtain zinc is:
A)
zinc blend
done
clear
B)
black zinc
done
clear
C)
both (a) and (b)
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 90) Which one does not show \[s{{p}^{2}}\] hybridisation?
A)
\[C{{O}_{3}}^{-2}\]
done
clear
B)
\[C{{O}_{2}}\]
done
clear
C)
\[N{{O}_{3}}\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 91) Strongest acid is:
A)
formic acid
done
clear
B)
acetiacid
done
clear
C)
proponic acid
done
clear
D)
\[C{{l}_{2}}C{{H}_{2}}COOH\]
done
clear
View Answer play_arrow
question_answer 92) Schiffs reagent gives the pink colour with:
A)
\[{{C}_{2}}{{H}_{5}}CHO\]
done
clear
B)
Propanone
done
clear
C)
\[{{C}_{2}}{{H}_{5}}OH\]
done
clear
D)
Propane -2-ol
done
clear
View Answer play_arrow
question_answer 93) Less soluble compound is:
A)
\[Be{{(OH)}_{2}}\]
done
clear
B)
\[Ba{{(OH)}_{2}}\]
done
clear
C)
\[Ca{{(OH)}_{2}}\]
done
clear
D)
\[Mg{{(OH)}_{2}}\]
done
clear
View Answer play_arrow
question_answer 94) Which of the following react with acetylene and formed dichloroacetaldehyde?
A)
\[HOCl\]
done
clear
B)
\[HCl\]
done
clear
C)
Both (a) and (b)
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 95) Which isomerism which is present in self-aldehyde?
A)
Chain isomerism
done
clear
B)
Position isomerism
done
clear
C)
Functional isomerism
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 96) Which of the following react with acetic acid for obtain acetic anhydride?
A)
\[{{P}_{2}}{{O}_{5}}\]
done
clear
B)
\[PC{{l}_{5}}\]
done
clear
C)
\[{{P}_{2}}{{O}_{3}}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 97) Ketone \[+X\xrightarrow{{}}oxime\]. The name of X is:
A)
\[N{{H}_{2}}-N{{H}_{2}}\]
done
clear
B)
\[N{{H}_{3}}\]
done
clear
C)
\[N{{H}_{2}}-OH\]
done
clear
D)
\[N{{H}_{2}}-NH-CO-N{{H}_{2}}\]
done
clear
View Answer play_arrow
question_answer 98) Which electronic configuration follows Hunds rule:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
question_answer 99) The correct order of strength of Lewis bases is:
A)
\[{{F}^{-}}>O{{H}^{-}}>C{{H}_{3}}^{-}>N{{H}_{2}}^{-}\]
done
clear
B)
\[NH_{2}^{-}>CH_{3}^{-}>O{{H}^{-}}>{{F}^{-}}\]
done
clear
C)
\[O{{H}^{-}}>C{{H}_{3}}^{-}>N{{H}_{2}}^{-}>{{F}^{-}}\]
done
clear
D)
\[C{{H}_{3}}^{-}>NH_{2}^{-}>O{{H}^{-}}>{{F}^{-}}\]
done
clear
View Answer play_arrow
question_answer 100) Which has the least solubility product?
A)
\[Be{{(OH)}_{2}}\]
done
clear
B)
\[Ba{{(OH)}_{2}}\]
done
clear
C)
\[Mg{{(OH)}_{2}}\]
done
clear
D)
\[Ca{{(OH)}_{2}}\]
done
clear
View Answer play_arrow
question_answer 101) Industrial alcohol is:
A)
\[100%\text{ }{{C}_{2}}{{H}_{5}}OH\]
done
clear
B)
\[93%\]to \[95%\text{ }C{{H}_{3}}OH\]
done
clear
C)
\[95%\]absoult alcohol \[+5%\,C{{H}_{3}}OH\]
done
clear
D)
\[90%\]alcohol
done
clear
View Answer play_arrow
question_answer 102) In which of the following is not acidic salt?
A)
\[NaHC{{O}_{3}}~~\]
done
clear
B)
\[Na{{H}_{2}}P{{O}_{2}}\]
done
clear
C)
\[KHS{{O}_{4}}\]
done
clear
D)
\[Na{{H}_{2}}P{{O}_{4}}\]
done
clear
View Answer play_arrow
question_answer 103) Which of the following ion will have maximum hydration energy?
A)
\[S{{r}^{+2}}\]
done
clear
B)
\[B{{a}^{+2}}\]
done
clear
C)
\[C{{a}^{+2}}\]
done
clear
D)
\[M{{g}^{+2}}\]
done
clear
View Answer play_arrow
question_answer 104) From the following mixtures which is electrolyzed in the metallurgy of aluminium:
A)
alumina \[NaOH\]
done
clear
B)
alumina \[+\] silicon
done
clear
C)
alumina 4-cryolite
done
clear
D)
aluminium chloride \[+CaC{{l}_{2}}\]
done
clear
View Answer play_arrow
question_answer 105) The solubility product of chalk is \[9.3\times {{10}^{-8}}\]. What will be solubility of this?
A)
\[3.04\times {{10}^{-4}}mole/litre\]
done
clear
B)
\[5\times {{10}^{-6}}\text{ }mole/litre\]
done
clear
C)
\[3\times {{10}^{-5}}mole/litre\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 106) Which of the following added to \[C{{u}_{2}}S\] for blaster formation?
A)
\[CuC{{O}_{3}}\]
done
clear
B)
\[F{{e}_{2}}{{O}_{3}}\]
done
clear
C)
\[CuO\]
done
clear
D)
\[C{{u}_{2}}O\]
done
clear
View Answer play_arrow
question_answer 107) Most suitable indicator in the titration of acetic acid and sodium hydroxide is:
A)
methyl orange
done
clear
B)
phenolphthalein
done
clear
C)
phenol red
done
clear
D)
all of the above
done
clear
View Answer play_arrow
question_answer 108) Which compound does not have ionic bond?
A)
Magnesium chloride
done
clear
B)
Zinc sulphide
done
clear
C)
Calcium fluoride
done
clear
D)
Graphite
done
clear
View Answer play_arrow
question_answer 109) The solubility product of \[BaS{{O}_{4}}\]is \[25\times {{10}^{-12}}\]. What will be the solubility of this:
A)
\[5\times {{10}^{-6}}mole/litre\]
done
clear
B)
\[25\times {{10}^{-6}}mole/litre\]
done
clear
C)
\[5\times {{10}^{-12}}mole/litre\]
done
clear
D)
\[25\times {{10}^{-12}}mole/litre\]
done
clear
View Answer play_arrow
question_answer 110) Argentite ore is reacted with which compound to obtain it in solution?
A)
\[Fe{{(CN)}_{6}}^{-4}\]
done
clear
B)
\[NaCN\]
done
clear
C)
\[{{C}_{6}}{{H}_{5}}-CN\]
done
clear
D)
\[C{{H}_{3}}CN\]
done
clear
View Answer play_arrow
question_answer 111) The ion which has not bond order \[2.5\] is:
A)
\[{{O}_{2}}^{-}\]
done
clear
B)
\[{{N}_{2}}^{+}\]
done
clear
C)
\[{{O}_{2}}^{+}\]
done
clear
D)
\[{{N}_{2}}^{-}\]
done
clear
View Answer play_arrow
question_answer 112) Which of the following pairs does not show same properties?
A)
\[B,Si\]
done
clear
B)
\[Na,Ca\]
done
clear
C)
\[Li,Mg\]
done
clear
D)
\[Be,Al\]
done
clear
View Answer play_arrow
question_answer 113) The position isomer of butyne is:
A)
\[3\]
done
clear
B)
\[4\]
done
clear
C)
\[2\]
done
clear
D)
\[1\]
done
clear
View Answer play_arrow
question_answer 114) \[HCHO+N{{H}_{3}}\xrightarrow{{}}X,\], product X is:
A)
amino form
done
clear
B)
hexamethylene tetramine
done
clear
C)
urotropin
done
clear
D)
all of these
done
clear
View Answer play_arrow
question_answer 115) \[Glucose\xrightarrow[(yeast)]{X\,enzyme}alcohol\] enzyme is:
A)
invertase
done
clear
B)
zymase
done
clear
C)
maltase
done
clear
D)
diatase
done
clear
View Answer play_arrow
question_answer 116) The ore of aluminium is:
A)
siderite
done
clear
B)
stephanite
done
clear
C)
malachite
done
clear
D)
cryolite
done
clear
View Answer play_arrow
question_answer 117) Which of the following is not used as primary standard in the redox reaction:
A)
Mohr salt
done
clear
B)
\[KMn{{O}_{4}}\]
done
clear
C)
Oxalic acid
done
clear
D)
\[{{K}_{2}}C{{r}_{2}}{{O}_{7}}\]
done
clear
View Answer play_arrow
question_answer 118) The compound which is not used in redox titration:
A)
\[{{(COOH)}_{2}}\]
done
clear
B)
\[KMn{{O}_{4}}\]
done
clear
C)
\[{{K}_{2}}C{{r}_{2}}{{O}_{7}}\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 119) The most stable carbonium ion is:
A)
\[C{{H}_{3}}-\overset{+}{\mathop{C}}\,H-{{C}_{2}}{{H}_{5}}\]
done
clear
B)
\[C{{H}_{3}}-\overset{+}{\mathop{C}}\,{{H}_{2}}\]
done
clear
C)
\[\overset{+}{\mathop{C}}\,{{H}_{3}}\]
done
clear
D)
\[C{{H}_{3}}-CH-C{{H}_{3}}\]
done
clear
View Answer play_arrow
question_answer 120) Which of the following reduction in acidic medium gives aniline?
A)
Nitrosobenzene
done
clear
B)
Hydrazobenzene
done
clear
C)
Nitrobenzene
done
clear
D)
Azobenzene
done
clear
View Answer play_arrow
question_answer 121) The compound/ion which is used in the nitration of benzene:
A)
nitrite ion
done
clear
B)
nitronium ion
done
clear
C)
sulphuric acid
done
clear
D)
nitric acid
done
clear
View Answer play_arrow
question_answer 122) An electron is transferred towards nuclei from infinite then the potential energy:
A)
increases
done
clear
B)
ramains constant
done
clear
C)
decreases
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 123) In this reaction: \[3B{{r}_{2}}+6C{{O}_{3}}^{2-}+3{{H}_{2}}O\xrightarrow{{}}5B{{r}^{-}}\]\[+Br{{O}_{3}}+6HC{{O}_{3}}^{-}\]
A)
oxidation of bromine and reduction of carbonate
done
clear
B)
reduction of bromine and oxidation of carbonate
done
clear
C)
no oxidation and no reduction of bromine
done
clear
D)
oxidation and reduction of bromine
done
clear
View Answer play_arrow
question_answer 124) Which alkene gives the same product by the Markownikoffs and anti Markownikoffs method?
A)
\[\beta \]-butylene
done
clear
B)
Propylene
done
clear
C)
\[\alpha \]-amylene
done
clear
D)
\[\alpha \]-butylene
done
clear
View Answer play_arrow
question_answer 125) The number of double bond in B.H.C. is:
A)
\[1\]
done
clear
B)
\[2\]
done
clear
C)
\[3\]
done
clear
D)
\[zero\]
done
clear
View Answer play_arrow
question_answer 126) In following reaction product A is: \[{{C}_{6}}{{H}_{5}}N{{H}_{2}}+C{{H}_{3}}COCl\to A\]
A)
\[{{C}_{6}}{{H}_{6}}NOCl\]
done
clear
B)
\[{{C}_{6}}{{H}_{5}}N{{H}_{2}}COC{{H}_{3}}\]
done
clear
C)
\[{{C}_{6}}{{H}_{5}}NHCOC{{H}_{3}}\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 127) In which of the following has the minimum oxidation number of \[Mn\]:
A)
\[KMn{{O}_{4}}\]
done
clear
B)
\[{{K}_{2}}Mn{{O}_{4}}\]
done
clear
C)
\[Mn{{O}_{2}}\]
done
clear
D)
\[Mn{{O}_{3}}\]
done
clear
View Answer play_arrow
question_answer 128) Which of the following has its pH near about 7 in its aqueous solution of concentration\[1M\]:
A)
sodium acetate
done
clear
B)
ammonium chloride
done
clear
C)
\[N{{a}_{2}}S{{O}_{4}}\]
done
clear
D)
\[FeC{{l}_{3}}\]
done
clear
View Answer play_arrow
question_answer 129) From overlapping of s-s orbital gives:
A)
\[\sigma \] bond
done
clear
B)
H-bond
done
clear
C)
\[\pi \]bond
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 130) The compound which have minimum bond angle:
A)
\[{{H}_{2}}O\]
done
clear
B)
\[{{H}_{2}}S\]
done
clear
C)
\[C{{H}_{4}}\]
done
clear
D)
\[N{{H}_{3}}\]
done
clear
View Answer play_arrow
question_answer 131) Which element will form nitride?
A)
\[Rb\]
done
clear
B)
\[Na\]
done
clear
C)
\[Li\]
done
clear
D)
\[K\]
done
clear
View Answer play_arrow
question_answer 132) The conjugate base of \[NH_{4}^{\oplus }\] is:
A)
\[N{{H}_{4}}\]
done
clear
B)
\[N{{H}_{2}}\]
done
clear
C)
\[N{{H}_{3}}\]
done
clear
D)
\[\overset{+}{\mathop{N}}\,{{H}_{3}}\]
done
clear
View Answer play_arrow
question_answer 133) The reaction between benzene and \[C{{H}_{3}}COCl\]gives:
A)
benzoyl chloride
done
clear
B)
toluene
done
clear
C)
acetophenone
done
clear
D)
benzophenone
done
clear
View Answer play_arrow
question_answer 134) \[C{{H}_{3}}-CHO+HCN\xrightarrow{{}}A\] \[\xrightarrow{hydrolysis}B,\] compound B is:
A)
malonic acid
done
clear
B)
glycolic acid
done
clear
C)
maleic acid
done
clear
D)
lactic acid
done
clear
View Answer play_arrow
question_answer 135) The reaction between formaldehyde and sodium hydroxide gives:
A)
\[{{C}_{2}}{{H}_{5}}OH+HCOONa\]
done
clear
B)
\[{{C}_{2}}{{H}_{5}}OH+C{{H}_{3}}COONa\]
done
clear
C)
\[C{{H}_{3}}OH+HCOONa\]
done
clear
D)
\[C{{H}_{3}}\text{OH}+C{{H}_{3}}COONa\]
done
clear
View Answer play_arrow
question_answer 136) \[A+HN{{O}_{2}}\xrightarrow{{}}\]alcohol, compound A is:
A)
primary amine
done
clear
B)
tertiary amine
done
clear
C)
secondary amine
done
clear
D)
none the these
done
clear
View Answer play_arrow
question_answer 137) The groups present in picric acid are:
A)
\[-COOH\]and \[-N{{O}_{2}}\]
done
clear
B)
\[-OH\]
done
clear
C)
\[-OH\] and \[-N{{O}_{2}}\]
done
clear
D)
\[-N{{O}_{2}}\]
done
clear
View Answer play_arrow
question_answer 138) Number of a and n bond in 1-butenc-3-yne:
A)
\[5\sigma \] and \[5\pi \]
done
clear
B)
\[7\sigma \] and \[3\pi \]
done
clear
C)
\[8\sigma \]and \[2\pi \]
done
clear
D)
\[6\sigma \]and \[4\pi \]
done
clear
View Answer play_arrow
question_answer 139) The product obtained by the reaction of ether with Bayers reagent is:
A)
\[C{{H}_{3}}OH\]
done
clear
B)
\[C{{H}_{3}}C{{H}_{2}}OH\]
done
clear
C)
\[\underset{\begin{smallmatrix} | \\ OH \end{smallmatrix}}{\mathop{C}}\,{{H}_{2}}-\underset{\begin{smallmatrix} | \\ OH \end{smallmatrix}}{\mathop{C}}\,{{H}_{2}}\]
done
clear
D)
\[C{{H}_{3}}-\underset{\begin{smallmatrix} \,|| \\ O \end{smallmatrix}}{\mathop{C}}\,-C{{H}_{3}}\]
done
clear
View Answer play_arrow
question_answer 140) P.V.C. is formed by the polymerization of:
A)
ethene
done
clear
B)
1-chloropropene
done
clear
C)
propene
done
clear
D)
1-chloro ethane
done
clear
View Answer play_arrow
question_answer 141) What will be the position number of isomers in \[{{C}_{2}}{{H}_{4}}C{{l}_{2}}\]?
A)
\[3\]
done
clear
B)
\[2\]
done
clear
C)
\[1\]
done
clear
D)
zero
done
clear
View Answer play_arrow
question_answer 142) Which one of the following compounds does not give \[{{C}_{2}}{{H}_{5}}Cl\]by the reaction of \[{{C}_{2}}{{H}_{5}}OH\]?
A)
\[C{{l}_{2}}\]
done
clear
B)
\[PC{{l}_{5}}\]
done
clear
C)
\[SOC{{l}_{2}}\]
done
clear
D)
\[ZnC{{l}_{2}}+HCl\]
done
clear
View Answer play_arrow
question_answer 143) Molecular formula of bleaching powder is:
A)
\[CaOC{{l}_{2}}\]
done
clear
B)
\[CaC{{O}_{3}}\]
done
clear
C)
\[Ca{{(OH)}_{2}}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 144) The atomic number of \[Fe\] is 26. The number of proton in \[Fe\] is:
A)
\[46\]
done
clear
B)
\[26\]
done
clear
C)
\[20\]
done
clear
D)
\[6\]
done
clear
View Answer play_arrow
question_answer 145) Which one have smallest size?
A)
\[M{{g}^{+2}}\]
done
clear
B)
\[B{{e}^{+2}}\]
done
clear
C)
\[N{{a}^{+}}\]
done
clear
D)
\[L{{i}^{+}}\]
done
clear
View Answer play_arrow
question_answer 146) Principal quantum number \[n=1\] die number of l, w, s quantum arc:
A)
\[n=l,l=0,m=0,s=+\frac{1}{2},-\frac{1}{2}\]
done
clear
B)
\[n=1,l=1,m=1,s=+\frac{1}{2}\]
done
clear
C)
\[n=1,l=2,m=0,s=+\frac{1}{2}\]
done
clear
D)
\[n=1,l=0,m=1,s=-\frac{1}{2}\]
done
clear
View Answer play_arrow
question_answer 147) Which ion does not have colour?
A)
\[Cu\,(I)\]
done
clear
B)
\[Cr\,(III)\]
done
clear
C)
\[Co(II)\]
done
clear
D)
\[Cu(II)\]
done
clear
View Answer play_arrow
question_answer 148) Which of the following ions does not have its electronic configuration \[[Ar]3{{d}^{1}}\]?
A)
\[N{{i}^{+2}}\]
done
clear
B)
\[N{{i}^{+4}}\]
done
clear
C)
\[C{{o}^{+3}}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 149) Acetone forms oxime by the reaction with:
A)
ammonia
done
clear
B)
\[{{(C{{H}_{3}})}_{2}}C=NH\]
done
clear
C)
\[{{(C{{H}_{3}})}_{3}}NH\]
done
clear
D)
\[N{{H}_{2}}-OH\]
done
clear
View Answer play_arrow
question_answer 150) \[CH\equiv CH+HBr\xrightarrow{{}}X,\], product X is:
A)
ethylene bromide
done
clear
B)
vinyl bromide
done
clear
C)
bromo ethane
done
clear
D)
ethyledene bromide
done
clear
View Answer play_arrow
question_answer 151) \[C{{H}_{3}}CHO+Sodium\text{ }cupritartarate\xrightarrow{{}}\] red precipitate. In which red precipitate is:
A)
\[C{{u}_{2}}O\]
done
clear
B)
\[N{{a}_{2}}C{{O}_{3}}\]
done
clear
C)
\[N{{a}_{2}}O\]
done
clear
D)
\[C{{O}_{2}}\]
done
clear
View Answer play_arrow
question_answer 152) In the following pairs which pair does not show the proper similarity?
A)
\[Na-Ca\]
done
clear
B)
\[Li-Mg\]
done
clear
C)
\[Be-Al~\]
done
clear
D)
\[B-Si\]
done
clear
View Answer play_arrow
question_answer 153) What will be the ratio of the radius of second and third orbits of hydrogen?
A)
\[9:4\]
done
clear
B)
\[2:3\]
done
clear
C)
\[3:2\]
done
clear
D)
\[4:9\]
done
clear
View Answer play_arrow
question_answer 154) For triple the pH of a solution the concentration of hydrogen ion should be:
A)
\[1/100\text{ }times\]
done
clear
B)
half
done
clear
C)
double
done
clear
D)
\[1/1000\text{ }times\]
done
clear
View Answer play_arrow
question_answer 155) The blue colour of the liquid ammonia solution of metal is due to:
A)
ammoniated electron
done
clear
B)
ammoniated anion
done
clear
C)
ammoniated cation
done
clear
D)
both (a) and (b)
done
clear
View Answer play_arrow
question_answer 156) Which compound affect the pH of \[N{{H}_{4}}OH\]solution?
A)
\[N{{H}_{4}}Cl~\]
done
clear
B)
\[CHC{{l}_{3}}\]
done
clear
C)
\[C{{H}_{3}}Cl\]
done
clear
D)
\[CH{{ }_{3}}OH\]
done
clear
View Answer play_arrow
question_answer 157) \[{{C}_{2}}{{H}_{5}}OH+X\to {{C}_{2}}{{H}_{5}}-O-{{C}_{2}}{{H}_{5}}\], X is:
A)
with \[Na\] at \[413K\]
done
clear
B)
with cone. \[{{H}_{2}}S{{O}_{4}}\] at \[413K\]
done
clear
C)
with cone. \[HCl\] at \[413K\]
done
clear
D)
with \[B{{r}_{2}}\]at \[413K\]
done
clear
View Answer play_arrow
question_answer 158) The gram number of atom in 46 gram sodium is:
A)
\[4\]
done
clear
B)
\[5\]
done
clear
C)
\[2\]
done
clear
D)
\[3\]
done
clear
View Answer play_arrow
question_answer 159) Fehling solution is:
A)
\[CuS{{O}_{4}}+lime\]
done
clear
B)
\[CuS{{O}_{4}}+NaOH(aq.)\]
done
clear
C)
\[CuS{{O}_{4}}+N{{a}_{2}}C{{O}_{3}}\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 160) The compound which has zero dipole moment:
A)
\[C{{O}_{2}}\]
done
clear
B)
\[S{{O}_{2}}\]
done
clear
C)
\[{{H}_{2}}O\]
done
clear
D)
\[CO\]
done
clear
View Answer play_arrow
question_answer 161) Alkali metals in comparison to alkaline earth metal have:
A)
low ionic radii
done
clear
B)
low reactivity
done
clear
C)
high melting
done
clear
D)
low ionization potential
done
clear
View Answer play_arrow
question_answer 162) Atomic size is the order of:
A)
\[{{10}^{\,-12}}m\]
done
clear
B)
\[{{10}^{-6}}m\]
done
clear
C)
\[{{10}^{-9}}m\]
done
clear
D)
\[{{10}^{-5}}m\]
done
clear
View Answer play_arrow
question_answer 163) An acidic solution whose concentration is equal to \[0.1M\]and the degree of ionization is 1%. Then the dissociation constant of acid will be:
A)
\[1.01\times {{10}^{5}}\]
done
clear
B)
\[1.01\times {{10}^{-5}}\]
done
clear
C)
\[1.01\times {{10}^{3}}\]
done
clear
D)
\[1.01\times {{10}^{-3}}\]
done
clear
View Answer play_arrow
question_answer 164) \[CHC{{l}_{3}}\xrightarrow[light]{Air\,and}X,\]product X is:
A)
\[HOCl\]
done
clear
B)
\[COC{{l}_{2}}\]
done
clear
C)
\[C{{l}_{2}}\]
done
clear
D)
\[HCOCl\]
done
clear
View Answer play_arrow
question_answer 165) Tollen reagent can be reduced in \[Ag\] by:
A)
ketone
done
clear
B)
acetic acid
done
clear
C)
ether
done
clear
D)
aldehyde
done
clear
View Answer play_arrow
question_answer 166) The number of s block element is:
A)
greater than p block element
done
clear
B)
greater than d block element
done
clear
C)
lesser than p and d block element
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 167) \[{{C}_{2}}{{H}_{5}}Br+C{{H}_{3}}CHO\xrightarrow{{}}X\xrightarrow{H.OH}\]Y, product Y is :
A)
propanol
done
clear
B)
2-butanol
done
clear
C)
both (a) and (c)
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 168) Which of the following equilibrium is uneffected by the change in pressure?
A)
\[2{{O}_{3(g)}}3{{O}_{2(g)}}\]
done
clear
B)
\[2N{{O}_{2(g)}}{{N}_{2}}{{O}_{4}}\]
done
clear
C)
\[2H{{I}_{(g)}}{{H}_{2(g)}}+{{I}_{2(g)}}\]
done
clear
D)
\[PC{{l}_{5(g)}}PC{{l}_{3(g)}}+C{{l}_{2(g)}}\]
done
clear
View Answer play_arrow
question_answer 169) The compound with smallest \[C-H\] bond will be:
A)
\[{{C}_{2}}{{H}_{6}}\]
done
clear
B)
\[{{C}_{2}}{{H}_{2}}\]
done
clear
C)
\[{{C}_{2}}{{H}_{4}}\]
done
clear
D)
\[{{C}_{2}}{{H}_{2}}B{{r}_{2}}\]
done
clear
View Answer play_arrow
question_answer 170) Phenol + potassium per disulphate\[\xrightarrow{KOH}X,\], product X is:
A)
quinol
done
clear
B)
phenoquinon
done
clear
C)
both (a) and (b)
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 171) Which of the following solutions gives red colour with HPh?
A)
\[5M\,NaOH\]
done
clear
B)
\[0.1M\text{ }NaC{{O}_{3}}\]
done
clear
C)
\[0.1M\text{ }N{{H}_{4}}OH\]
done
clear
D)
\[0.1M\text{ }NaCl\]
done
clear
View Answer play_arrow
question_answer 172) Which one of the following reaction will not affected by increases in pressure?
A)
\[2{{H}_{2}}+{{O}_{2}}\xrightarrow{{}}2{{H}_{2}}O\]
done
clear
B)
\[{{N}_{2}}\text{+}3{{H}_{2}}\xrightarrow{{}}2N{{H}_{3}}\]
done
clear
C)
\[2Cu+S\xrightarrow{{}}C{{u}_{2}}S\]
done
clear
D)
\[{{H}_{2}}+{{I}_{2}}\xrightarrow{{}}2HI\]
done
clear
View Answer play_arrow
question_answer 173) In which of the following is simple salt?
A)
\[NaOH\]
done
clear
B)
\[NaCl\]
done
clear
C)
\[N{{a}_{2}}C{{O}_{3}}\]
done
clear
D)
\[NaCN\]
done
clear
View Answer play_arrow
question_answer 174) \[X+{{I}_{2}}+NaOH\xrightarrow{{}}\]not iodo form, X is:
A)
\[{{C}_{2}}{{H}_{5}}OH\]
done
clear
B)
\[C{{H}_{3}}OH\]
done
clear
C)
\[CH3CHO\]
done
clear
D)
\[C{{H}_{3}}COC{{H}_{3}}\]
done
clear
View Answer play_arrow
question_answer 175) Which one of the following does not has its magnetic moments value equal to \[5.9\] Bohr magneton:
A)
\[C{{r}^{+}}\]
done
clear
B)
\[M{{n}^{+2}}\]
done
clear
C)
\[F{{e}^{+3}}\]
done
clear
D)
\[C{{O}^{+3}}\]
done
clear
View Answer play_arrow
question_answer 176) The value of \[{{K}_{c}}\] for this reaction is: \[A+B2C+D\]:
A)
\[K=\frac{{{[C]}^{2}}[D]}{[A][B]}\]
done
clear
B)
\[{{K}_{c}}=\frac{[A][B]}{[C][D]}\]
done
clear
C)
both (a) and (b)
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 177) The rule of full filling the orbit obey:
A)
Aufbaus
done
clear
B)
Hund
done
clear
C)
Pauli
done
clear
D)
Heisenberg
done
clear
View Answer play_arrow
question_answer 178) The bond having least polar nature is:
A)
\[H-O\]
done
clear
B)
\[H-F\]
done
clear
C)
\[H-Cl\]
done
clear
D)
\[H-S\]
done
clear
View Answer play_arrow
question_answer 179)
A)
\[B{{r}_{2}}\] (Br water)
done
clear
B)
\[~NaBr\]
done
clear
C)
\[HBr\]
done
clear
D)
\[KBr\]
done
clear
View Answer play_arrow
question_answer 180) Which chemical is released by vagus nerve to stow down the heart beat?
A)
Adrenalin
done
clear
B)
Acetyichotine
done
clear
C)
Nor epinephrine
done
clear
D)
Epinephrine
done
clear
View Answer play_arrow
question_answer 181) Pacinian corpuscles are receptor of:
A)
Deep pressure
done
clear
B)
Pain
done
clear
C)
Temperature
done
clear
D)
Hearing
done
clear
View Answer play_arrow
question_answer 182) Two homologous chromosome are connected at:
A)
telomere
done
clear
B)
chromomere
done
clear
C)
centromere
done
clear
D)
sineptonemal mixture
done
clear
View Answer play_arrow
question_answer 183) Which of the following plant lipid forms essential part of a good diet?
A)
Linolic acid
done
clear
B)
Palmittic acid
done
clear
C)
Oleic acid
done
clear
D)
Stearic acid
done
clear
View Answer play_arrow
question_answer 184) Endomitosis is related with:
A)
mitosis of chromosomes in the presence of nuclear membrane
done
clear
B)
mitosis without nucleus
done
clear
C)
mitosis in uterine wall
done
clear
D)
frequent mitosis
done
clear
View Answer play_arrow
question_answer 185) Which activity occurs first during the development of an egg?
A)
Induction
done
clear
B)
Differential growth
done
clear
C)
Polarity
done
clear
D)
Gastrulatron
done
clear
View Answer play_arrow
question_answer 186) Waste material in Amoeba is taken out by:
A)
ptasmalemma
done
clear
B)
cytoplasm
done
clear
C)
nephron
done
clear
D)
vacuole
done
clear
View Answer play_arrow
question_answer 187) Red and white pulp tissue are the parts of:
A)
spleen
done
clear
B)
muscle fibre
done
clear
C)
lymph layer
done
clear
D)
lymph nodules
done
clear
View Answer play_arrow
question_answer 188) Pick out the pair of essential (indespensibic) amino acid:
A)
leucine and glycine
done
clear
B)
valin and histidine
done
clear
C)
tryptophtin and glutamic acid
done
clear
D)
lycine and phenyl alanine
done
clear
View Answer play_arrow
question_answer 189) Right auricle of the mammalian heart release blood through :
A)
tricuspid valve
done
clear
B)
vena cava
done
clear
C)
pulmonary aorta
done
clear
D)
mitral valve
done
clear
View Answer play_arrow
question_answer 190) Adrenal medulla is derived from:
A)
ectoderm
done
clear
B)
mosoderm
done
clear
C)
endoderm
done
clear
D)
both a and c
done
clear
View Answer play_arrow
question_answer 191) Millipede (Julus) and Centipede (Scolopendra) are included in:
A)
Arachnida
done
clear
B)
Myriapoda
done
clear
C)
Scaphopoda
done
clear
D)
Palecypoda
done
clear
View Answer play_arrow
question_answer 192) Which of the following pair include oldest and primitive vertebraites?
A)
Amphioxus and Herdmania
done
clear
B)
Doliolum and Scoliodon
done
clear
C)
Balanoglossus and Salpa
done
clear
D)
Petromyzon and Myxine
done
clear
View Answer play_arrow
question_answer 193) Mitotic prophase is characterized by:
A)
condensation of chromosome
done
clear
B)
disappearance of nucleolus
done
clear
C)
formation of meta phasic plate
done
clear
D)
division of centromere
done
clear
View Answer play_arrow
question_answer 194) Purkinje fibres are present in:
A)
mammalian heart
done
clear
B)
voluntary muscles
done
clear
C)
cerebral cortex
done
clear
D)
ampullae of semi-circular canal of internal ear
done
clear
View Answer play_arrow
question_answer 195) The sound reception can be interpreted by:
A)
olfactory lobe
done
clear
B)
cerebral hemisphere
done
clear
C)
cerebellum
done
clear
D)
medulla oblongata
done
clear
View Answer play_arrow
question_answer 196) Ciliated epithelium lines the :
A)
tracheal and fallopian tube
done
clear
B)
bile duct and ureter epithelium
done
clear
C)
trachea and oesophagus
done
clear
D)
pharyngeal and stomach mucosae
done
clear
View Answer play_arrow
question_answer 197) Sperm penetration bring changes in ova, is:
A)
formation of first polar body
done
clear
B)
I-meiosis
done
clear
C)
II-meiosis
done
clear
D)
formation of pronuclei
done
clear
View Answer play_arrow
question_answer 198) Which of the foetal membrane is directly connected with blood?
A)
Allantoic
done
clear
B)
Amnion
done
clear
C)
Chorion
done
clear
D)
Yolk sac
done
clear
View Answer play_arrow
question_answer 199) Chief cells of the gastric glands secreting zymogen, have well developed :
A)
smooth ER
done
clear
B)
rough ER
done
clear
C)
mitochondria
done
clear
D)
lysosome
done
clear
View Answer play_arrow
question_answer 200) Removal of thymus gland in the early life of an experimental mammal will cause :
A)
lack of lymphocytes
done
clear
B)
lack of antibodies
done
clear
C)
lack of lymph nodes and lymph vessels
done
clear
D)
all of the above
done
clear
View Answer play_arrow
question_answer 201) How many embryonic segments fuse to form head of adult Cockroach?
A)
4
done
clear
B)
6
done
clear
C)
8
done
clear
D)
10
done
clear
View Answer play_arrow
question_answer 202) In which of the following eggs meroblastic cleavage occurs?
A)
isolecithal
done
clear
B)
microlecithal
done
clear
C)
centrolecithal
done
clear
D)
telolecithal
done
clear
View Answer play_arrow
question_answer 203) Craniostylic jaw suspensor is found in:
A)
Shark
done
clear
B)
Limbless amphibians
done
clear
C)
Flightless birds
done
clear
D)
Rabbit
done
clear
View Answer play_arrow
question_answer 204) Which two extra cranial nerves are present in amniotes?
A)
Laryngoal and hypoglossal
done
clear
B)
Spinal accessory and hypoglossal
done
clear
C)
Hypoglossal and facial
done
clear
D)
Trigeminal and glossopharyngeal
done
clear
View Answer play_arrow
question_answer 205) Function of spermatheca in Earthworm is:
A)
sperm maturation
done
clear
B)
collection of sperms of same animal
done
clear
C)
collection of sperms of other animal
done
clear
D)
fertilization
done
clear
View Answer play_arrow
question_answer 206) Name the mammalian tissue which has no power of regeneration after damage/injury:
A)
peripheral nervous tissue
done
clear
B)
skin epidermis
done
clear
C)
tendon
done
clear
D)
smooth muscles
done
clear
View Answer play_arrow
question_answer 207) \[{{N}_{2}}\] waste in the form of uric acid is excreted by:
A)
Amoeba
done
clear
B)
Dog fish
done
clear
C)
Rabbit
done
clear
D)
Crow
done
clear
View Answer play_arrow
question_answer 208) \[C{{O}_{2}}\] is mainly transported by:
A)
respiratory pigment
done
clear
B)
dissolution of gases
done
clear
C)
\[{{O}_{2}}\] taken by tissues
done
clear
D)
bicarbonates
done
clear
View Answer play_arrow
question_answer 209) Notochord is present in the larva of:
A)
Herdmania
done
clear
B)
Cephalodiscus
done
clear
C)
Balanoglossus
done
clear
D)
Amphioxus
done
clear
View Answer play_arrow
question_answer 210) In telophase of mitosis :
A)
chromosomes arrange in the middle of cell
done
clear
B)
chromosome fibres become clear
done
clear
C)
chromosomes condense at opposite pole
done
clear
D)
disappearence of nuclear membrane
done
clear
View Answer play_arrow
question_answer 211) The most primitive type of mammalian placenta is :
A)
epithelio - chorial
done
clear
B)
syndesmo - chorial
done
clear
C)
endothelio - chorial
done
clear
D)
haemo - chorial
done
clear
View Answer play_arrow
question_answer 212) What will the RNA code, if the DNA codes ATG?
A)
ATG
done
clear
B)
AUG
done
clear
C)
UAC
done
clear
D)
TAC
done
clear
View Answer play_arrow
question_answer 213) Correct member of the phyla is :
A)
Arthropoda - Arachnida - Lobstar
done
clear
B)
Arthropoda - Myriapoda - Scorpion
done
clear
C)
Mollusca - Cephalopoda - Octopus
done
clear
D)
Mollusca - Gastropoda - Squid
done
clear
View Answer play_arrow
question_answer 214) Phylum mollusca can be differentiated from other invertebrates by having:
A)
bilateral symmetry and external skeleton
done
clear
B)
mantle and gill
done
clear
C)
shell and non segmented body
done
clear
D)
mantle and non segmented body
done
clear
View Answer play_arrow
question_answer 215) Micro and macro nucleus are found in:
A)
Entamoeba and Plasmodium
done
clear
B)
Euglena and Volvox
done
clear
C)
Paramecium and Vorticella
done
clear
D)
Entamoeba and Vorticella
done
clear
View Answer play_arrow
question_answer 216) Biceps are attached with:
A)
radius
done
clear
B)
ulna
done
clear
C)
femur
done
clear
D)
scapula
done
clear
View Answer play_arrow
question_answer 217) Symptom of paroxysm in malaria (infection) are dipicted in stage:
A)
sporozoite
done
clear
B)
gametocyte
done
clear
C)
pre erythrocytic cycle
done
clear
D)
erythrocytic cycle
done
clear
View Answer play_arrow
question_answer 218) Cell membrane spents energy in:
A)
movement of solute from lower to higher concentration
done
clear
B)
diffusion of gases
done
clear
C)
movement of solute from high to lower concentration
done
clear
D)
movement of solvent
done
clear
View Answer play_arrow
question_answer 219) DNA gets doubled in :
A)
\[{{G}_{1}}\] phase
done
clear
B)
S phase
done
clear
C)
\[{{G}_{2}}\] phase
done
clear
D)
M phase
done
clear
View Answer play_arrow
question_answer 220) Which of the following order is correct for layers from outer to inner in Ascariss body wall?
A)
Cuticle, epidermis, longitudinal and circular muscle
done
clear
B)
Cuticle, epidermis, longitudinal muscles
done
clear
C)
Epidermis, cuticle, longitudinal and circular muscle
done
clear
D)
Epidermis, longitudinal, cuticle and circular muscles
done
clear
View Answer play_arrow
question_answer 221) Uicondylic skull with 10 pairs of cranial nerves is present in:
A)
Pisces
done
clear
B)
Amphibia
done
clear
C)
Reptilia
done
clear
D)
Mammalia
done
clear
View Answer play_arrow
question_answer 222) In Frog embryo, spreading of the layer of presumptive ectodermal cells on the underlying mass of cells is called:
A)
epiboly
done
clear
B)
invagination
done
clear
C)
ingcssion
done
clear
D)
delamination
done
clear
View Answer play_arrow
question_answer 223) Which of the following harmful substance found in blood are changed into harmless by Ornithine cycle?
A)
\[C{{O}_{2}}\] and \[N{{H}_{2}}-CO-N{{H}_{2}}\]
done
clear
B)
\[C{{O}_{2}}\] and \[N{{H}_{2}}\]
done
clear
C)
\[N{{H}_{3}}\] and \[N{{H}_{2}}-CO-N{{H}_{2}}\]
done
clear
D)
\[N{{H}_{3}}\] and uric acid
done
clear
View Answer play_arrow
question_answer 224) What will happen if corpora allata is disccted out from the nymph of an insect nymph will?
A)
Become adult
done
clear
B)
Immediately metamorphosis into second nymph and will remain in that stage
done
clear
C)
Die after some time
done
clear
D)
Remain nymph for long life
done
clear
View Answer play_arrow
question_answer 225) Exposed surface of the human body is covered by the tissue:
A)
glandular epithelium
done
clear
B)
straited squamous epithelium
done
clear
C)
ciliated epithelium
done
clear
D)
epidermis
done
clear
View Answer play_arrow
question_answer 226) In which of the following group, alimentary canal is absent?
A)
Taenia, Schistosoma
done
clear
B)
Ascaris, Fusciola
done
clear
C)
Taenia, Echinococcus
done
clear
D)
Trichuris, Fasciola
done
clear
View Answer play_arrow
question_answer 227) How many types of gamete will form by Aa Bb Cc cell?
A)
2
done
clear
B)
4
done
clear
C)
8
done
clear
D)
16
done
clear
View Answer play_arrow
question_answer 228) Sharpeys fibres are invaded in:
A)
collagen
done
clear
B)
muscle
done
clear
C)
bone
done
clear
D)
skin
done
clear
View Answer play_arrow
question_answer 229) Too short eye ball and too flat lens produce:
A)
near sightedness
done
clear
B)
far sightness
done
clear
C)
astigmatism
done
clear
D)
presshyopia
done
clear
View Answer play_arrow
question_answer 230) The reabsorption of ions by tubules of kidney is enhanced by :
A)
cortisol
done
clear
B)
aldosterone
done
clear
C)
calciferol
done
clear
D)
erythropeitin
done
clear
View Answer play_arrow
question_answer 231) Flat membranous bag (cisternae) is characterized in :
A)
mitochondria
done
clear
B)
golgi bodies
done
clear
C)
peroxisome
done
clear
D)
endoplasmic reticulum
done
clear
View Answer play_arrow
question_answer 232) How many high phosphate bonds are consumed during glucose formation from pyruvate?
A)
4
done
clear
B)
8
done
clear
C)
16
done
clear
D)
32
done
clear
View Answer play_arrow
question_answer 233) Collection and disperssion of embryonic cell is called :
A)
differentiation
done
clear
B)
induction
done
clear
C)
morphogenesis
done
clear
D)
blastomy
done
clear
View Answer play_arrow
question_answer 234) Intron is:
A)
the part of DNA which codes for protein synthesis
done
clear
B)
a part of DNA which do not join in m-RNA
done
clear
C)
part which helps to Join pieces of DNA
done
clear
D)
the part of DNA which do not code for protein synthesis
done
clear
View Answer play_arrow
question_answer 235) Find out the correct pair of phylum example :
A)
Echinodermata - Cuttle fish
done
clear
B)
Arthropoda - Silver fish
done
clear
C)
Pisces - Jelly fish
done
clear
D)
Mollusca - Star fish
done
clear
View Answer play_arrow
question_answer 236) Blood pressure and activity of non-straited muscles arc dependent on the hormone secretion of:
A)
anterior lobe of pituitary gland
done
clear
B)
middle lobe of pituitary gland
done
clear
C)
posterior lobe of pituitary gland
done
clear
D)
adrenal cortex
done
clear
View Answer play_arrow
question_answer 237) Which of the following pair is monosaccharide?
A)
Glucose and sucrose
done
clear
B)
Ribose and maltose
done
clear
C)
Fructose and glucose
done
clear
D)
Ribose and sucrose
done
clear
View Answer play_arrow
question_answer 238) Cell body (soma) of motor nerves is situated in:
A)
dorsal root of spinal cord
done
clear
B)
dorsal horn of spinal cord
done
clear
C)
ventral and lateral horn of spinal cord
done
clear
D)
ventral root of spinal nerve
done
clear
View Answer play_arrow
question_answer 239) Micro villi of intestine serve to:
A)
release of digestive enzyme
done
clear
B)
increase membrane area for absorption
done
clear
C)
protect cells from invading micro-organism
done
clear
D)
establish inter-cellular contact
done
clear
View Answer play_arrow
question_answer 240) Which of the following is incorrect regarding insect?
A)
Cephalization and jointed appandages
done
clear
B)
Chitinous exoskeleton and wings
done
clear
C)
Cephalization and complete metamorphosis
done
clear
D)
Well developed sensory organs and haemocoel
done
clear
View Answer play_arrow
question_answer 241) Animals bearing numerous (multiple) setae, arc included in class:
A)
Hirudinea
done
clear
B)
Oligochaeta
done
clear
C)
Polychaeta
done
clear
D)
Onchyophora
done
clear
View Answer play_arrow
question_answer 242) Which of the following has haploid chromosome?
A)
Oogonia
done
clear
B)
Primary oocyte
done
clear
C)
Secondary oocyte
done
clear
D)
Primary spermatocyte
done
clear
View Answer play_arrow
question_answer 243) Organ of corti includes:
A)
scala vestibular
done
clear
B)
scala media
done
clear
C)
semicircular canal
done
clear
D)
sensory hair
done
clear
View Answer play_arrow
question_answer 244) Cortex medulla and membrane labyrinth of vertebrates is formed of:
A)
ectoderm
done
clear
B)
endoderm
done
clear
C)
mesoderm
done
clear
D)
both b and c
done
clear
View Answer play_arrow
question_answer 245) Chief cell of gastric gland are:
A)
simple tubular
done
clear
B)
coiled tubular
done
clear
C)
branched tubular
done
clear
D)
compound tubular
done
clear
View Answer play_arrow
question_answer 246) In male mammals primary germ cell is formed by:
A)
cortex of testis
done
clear
B)
medulla of testis
done
clear
C)
germinal epithelium
done
clear
D)
apical part of embryo
done
clear
View Answer play_arrow
question_answer 247) Before cellular respiration, amino acid undergo the process of:
A)
decarboxylation
done
clear
B)
hydrolysis
done
clear
C)
deamination
done
clear
D)
phosphorylation
done
clear
View Answer play_arrow
question_answer 248) Antennae of Cockroach function as:
A)
tactile and sensory receptor
done
clear
B)
gustatory receptor
done
clear
C)
auditory receptor
done
clear
D)
olfactory receptor
done
clear
View Answer play_arrow
question_answer 249) Male Cockroach can be identified by:
A)
anal style
done
clear
B)
anal cerci
done
clear
C)
antennae
done
clear
D)
wings
done
clear
View Answer play_arrow
question_answer 250) Characteristic of autotomy (leaving an external body organ by own) is found in :
A)
Lizards
done
clear
B)
Snakes
done
clear
C)
Frogs
done
clear
D)
Cockroaches
done
clear
View Answer play_arrow
question_answer 251) In which form iron is present in haemoglobin?
A)
Ionic
done
clear
B)
Unionic
done
clear
C)
\[F{{e}^{2+}}\]
done
clear
D)
\[F{{e}^{+3}}\]
done
clear
View Answer play_arrow
question_answer 252) Nephrostome is a component of:
A)
septal nephridia
done
clear
B)
integumentary nephridia
done
clear
C)
pharyngeal and septal nephridia
done
clear
D)
pharyngeal and integumentary nephridia
done
clear
View Answer play_arrow
question_answer 253) Diabetes insipedus is caused by the deficiency of hormone:
A)
insulin
done
clear
B)
vasopressin
done
clear
C)
glucagon
done
clear
D)
oxytocin
done
clear
View Answer play_arrow
question_answer 254) Gastric glands of mammals are:
A)
simple tubular
done
clear
B)
simple coiled tubular
done
clear
C)
branched tubular
done
clear
D)
compound tubular
done
clear
View Answer play_arrow
question_answer 255) Bones of pelvic girdle forms a cavity in which head of the femur is fitted are:
A)
ilium only
done
clear
B)
ilium and is chium
done
clear
C)
ilium, ischium and pubis
done
clear
D)
ischium and pubis
done
clear
View Answer play_arrow
question_answer 256) Lactose is compose of:
A)
glucose + fructose
done
clear
B)
glucose + galactose
done
clear
C)
galactose + mannose
done
clear
D)
fructose + galactose
done
clear
View Answer play_arrow
question_answer 257) Genus Aedes of mosquito is a vector of:
A)
filaria
done
clear
B)
plague
done
clear
C)
dengue fever
done
clear
D)
elephantiasis
done
clear
View Answer play_arrow
question_answer 258) Thyroidectomised tadpole will metamorphosis by the administration of:
A)
TSH
done
clear
B)
thyroxine
done
clear
C)
pituitary extract
done
clear
D)
iodine
done
clear
View Answer play_arrow
question_answer 259) Which part of the human eye adjusts the focal length of lens?
A)
Cornea
done
clear
B)
Aquous humour
done
clear
C)
Conjunctive
done
clear
D)
Ciliary body
done
clear
View Answer play_arrow
question_answer 260) Function of t-RNA is to:
A)
copy the genetic code from DNA in nucleus
done
clear
B)
collection of RNA in ribosome
done
clear
C)
carry amino acids to ribosomes
done
clear
D)
transfer of gentic code from nucleus to cytoplasm
done
clear
View Answer play_arrow
question_answer 261) Which of the following is a connecting link between two phylum?
A)
Archneopteryx -Aves and Mammalia
done
clear
B)
Amphioxus-Echinodermata and Chordata
done
clear
C)
Ornithorhynchus -Aves and Reptilia
done
clear
D)
Peripatus -Annelida and Arthropoda
done
clear
View Answer play_arrow
question_answer 262) A parasite in absence of host, become saprophytic. It is termed as :
A)
obligate parasite
done
clear
B)
facultative parasite
done
clear
C)
obligate saprophyte
done
clear
D)
facultative saprophyte
done
clear
View Answer play_arrow
question_answer 263) Which of the following groups cause relapse malaria?
A)
Plasmodium falciparum and Plasmodium vivax
done
clear
B)
Plasmodium ovale and Plasmodium vivax
done
clear
C)
Plasmodium falciparum and Plasmodium ovale
done
clear
D)
Only Plasmodium falciparum
done
clear
View Answer play_arrow
question_answer 264) Chloragogen cells are present in:
A)
Taenia
done
clear
B)
Earthworm
done
clear
C)
Hydra
done
clear
D)
Sycon
done
clear
View Answer play_arrow
question_answer 265) Smooth endoplasmic reticulum is mainly found in cell which is actively engaged in:
A)
lipid metabolism
done
clear
B)
catabolic activity
done
clear
C)
protein
done
clear
D)
secreting activity
done
clear
View Answer play_arrow
question_answer 266) Nucleolus is a:
A)
round structure present near the nucleus in cytoplasm
done
clear
B)
round structure composed of r-RNA situated in the nucleus
done
clear
C)
rod-shaped structure present near the nucleus in cytoplasm
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 267) Features of acraniate vertebrates are:
A)
median and paired fins, 7 gill slits and acoelous vertebral column
done
clear
B)
paired fin, 5 gill slits, coelous vertebral column
done
clear
C)
median fin, 7 gill slits, acoelous vertebral column
done
clear
D)
median fin, 5 gill slits, and coelous vertebral column
done
clear
View Answer play_arrow
question_answer 268) Lower jaw of Rabbit articulates with :
A)
jugal
done
clear
B)
alisphenoid
done
clear
C)
squamosal
done
clear
D)
parietal
done
clear
View Answer play_arrow
question_answer 269) Which of the following is not an odd digitate pentadacythlus?
A)
Donkey
done
clear
B)
Came
done
clear
C)
Zebra
done
clear
D)
Rhinoceros
done
clear
View Answer play_arrow
question_answer 270) Function of ccdysone hormone in insect is:
A)
the growth and development of larva
done
clear
B)
the maturation into adult and laying eggs
done
clear
C)
to carry moulting in larval stage to form pupa
done
clear
D)
the secretion of cuticle in adult
done
clear
View Answer play_arrow
question_answer 271) Which of the spiracles in Cockroach are larger keep open always?
A)
First and second pair
done
clear
B)
First and third pair
done
clear
C)
First and tenth pair
done
clear
D)
Second and third pair
done
clear
View Answer play_arrow
question_answer 272) Number of juvenile stages form during development of fertilized egg into adult Ascaris are:
A)
1
done
clear
B)
2
done
clear
C)
3
done
clear
D)
4
done
clear
View Answer play_arrow
question_answer 273) Type of sexual reproduction in Protists, bearing diploid chromosome, is:
A)
zygotic meiosis
done
clear
B)
binary fission
done
clear
C)
cyst formation
done
clear
D)
gametangial meiosis
done
clear
View Answer play_arrow
question_answer 274) Glomerular area of adrenal cortex is responsible for:
A)
water and electrolyte balance
done
clear
B)
carbohydrate metabolism
done
clear
C)
steroid and hormone secretion
done
clear
D)
blood pressure
done
clear
View Answer play_arrow
question_answer 275) The sign of osmotic potential is:
A)
(-)
done
clear
B)
(+)
done
clear
C)
(x)
done
clear
D)
(÷)
done
clear
View Answer play_arrow
question_answer 276) Mendel was not imagine about which of the following?
A)
Linkage
done
clear
B)
Incomplete dominance
done
clear
C)
Dominance
done
clear
D)
Segregation of alleles
done
clear
View Answer play_arrow
question_answer 277) Which of the following gas is used for ripening of fruits?
A)
Nitrogen
done
clear
B)
Oxygen
done
clear
C)
Ethylene
done
clear
D)
Acetylene
done
clear
View Answer play_arrow
question_answer 278) Hold fast cell of Ulothrix shows as a:
A)
companion cell
done
clear
B)
dead cell
done
clear
C)
division of labour
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 279) The autosomes are:
A)
somatic chromosomes
done
clear
B)
sexual chromosomes
done
clear
C)
asexual chromosome
done
clear
D)
both (a) and (b)
done
clear
View Answer play_arrow
question_answer 280) Sun hemp is obtained from:
A)
Crotalaria jumcea
done
clear
B)
Linum usitatiseinum
done
clear
C)
Cochorus capsularis
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 281) Palenology is the study of:
A)
morphology of pollens
done
clear
B)
germination of pollens
done
clear
C)
development of pollens
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 282) The reaction of indole acetic acid (IAA) was tested by:
A)
avena curvature test
done
clear
B)
soybean callous test
done
clear
C)
\[\alpha \]-amylase test
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 283) The main characteristic of bryophyta is:
A)
spore formation
done
clear
B)
absence of roots
done
clear
C)
absence of vascular bundle
done
clear
D)
dependence of sporophyte on gametophyte
done
clear
View Answer play_arrow
question_answer 284) Crossing over takes place between:
A)
unequal chromatids
done
clear
B)
equal chromatids
done
clear
C)
any two chromatids
done
clear
D)
homologous chromosomes
done
clear
View Answer play_arrow
question_answer 285) The enzyme involved in fixation of \[C{{O}_{2}}\] in \[{{C}_{4}}\] plants is:
A)
PEP carboxylase
done
clear
B)
Ru DP carboxylase
done
clear
C)
RuDP oxydase
done
clear
D)
both (a) and (b)
done
clear
View Answer play_arrow
question_answer 286) Cell organelle covered by single unit membrane is:
A)
lysosome
done
clear
B)
nucleus
done
clear
C)
chloroplast
done
clear
D)
mitochondria
done
clear
View Answer play_arrow
question_answer 287) Vegetative reproduction in Cycas occurs by:
A)
bulbils
done
clear
B)
sporophylls
done
clear
C)
fission
done
clear
D)
scale leaves
done
clear
View Answer play_arrow
question_answer 288) Suspensor is a part of:
A)
developing embryo
done
clear
B)
mature embryo
done
clear
C)
germinating embryo
done
clear
D)
endosperm
done
clear
View Answer play_arrow
question_answer 289) Deposition of which of the following material takes place on casparian strip:
A)
Lignin
done
clear
B)
Cutin
done
clear
C)
Suberin
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 290) Fungus used in genetical experiments is:
A)
Neurospora
done
clear
B)
Rhizopus
done
clear
C)
Mucor
done
clear
D)
Anbaena
done
clear
View Answer play_arrow
question_answer 291) Symbiotic relationship between blue-green algae and fungus is called:
A)
mycorrhiza
done
clear
B)
lichen
done
clear
C)
bryophyta
done
clear
D)
gymnosperm
done
clear
View Answer play_arrow
question_answer 292) The reason of heteros is formation in plants is:
A)
crossing between unrelated plants
done
clear
B)
vegetative reproduction
done
clear
C)
colchicine
done
clear
D)
treatment with mutagen
done
clear
View Answer play_arrow
question_answer 293) Edible oil is obtained from which legumenous plant:
A)
Glycine max
done
clear
B)
Ricinus communis
done
clear
C)
Vigna sinensis
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 294) Photosynthesis takes place maximum in light of:
A)
white colour
done
clear
B)
red colour
done
clear
C)
voilet colour
done
clear
D)
green colour
done
clear
View Answer play_arrow
question_answer 295) Asafoctida (Heeng) obtains from:
A)
root secretion
done
clear
B)
leaf
done
clear
C)
seed
done
clear
D)
fruit
done
clear
View Answer play_arrow
question_answer 296) Mesophyll of dorsiventral leaf is consist of only spongy mesophyll
A)
palisade tissue
done
clear
B)
spongy parenchyma and palisade
done
clear
C)
tissue
done
clear
D)
sometimes spongy and sometimes palisade tissue
done
clear
View Answer play_arrow
question_answer 297) The shape of stomata in Cynodon dactylon (doob) is:
A)
oval
done
clear
B)
heart shaped
done
clear
C)
dumb-bell shaped
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 298) Infolding of plasma membrane in gram (+ve) bacterial cell is called:
A)
mesosome
done
clear
B)
rcticulum
done
clear
C)
lamellae
done
clear
D)
cristae
done
clear
View Answer play_arrow
question_answer 299) Adventitious roots originates from which part of the stem :
A)
epidermis
done
clear
B)
cortex
done
clear
C)
pericycle
done
clear
D)
endodermis
done
clear
View Answer play_arrow
question_answer 300) After virus infection host cell secretes a substance for their resistance is known as:
A)
aflatoxins
done
clear
B)
antibody
done
clear
C)
interferons
done
clear
D)
antigen
done
clear
View Answer play_arrow
question_answer 301) Who proposed the principle of limiting factors?
A)
Hill
done
clear
B)
Blackmann
done
clear
C)
Amon
done
clear
D)
Liebig
done
clear
View Answer play_arrow
question_answer 302) Characteristics of xerophytes are:
A)
long roots
done
clear
B)
thick epidermis
done
clear
C)
sunken stomata
done
clear
D)
all of the above
done
clear
View Answer play_arrow
question_answer 303) Characteristic of hydrophyte is:
A)
well developed roots
done
clear
B)
well developed xylem
done
clear
C)
less developed roots
done
clear
D)
well developed stem with sclerenchymatous tissue
done
clear
View Answer play_arrow
question_answer 304) Which of the following follows Chargaff rule?
A)
\[\text{A+G=T+C}\]
done
clear
B)
\[\text{A=C}\]
done
clear
C)
\[\text{G=T}\]
done
clear
D)
\[\frac{\text{A+T}}{\text{G+C}}\text{=1}\]
done
clear
View Answer play_arrow
question_answer 305) Function of cytokinin is:
A)
induction of cell division and delay in senescence
done
clear
B)
induction of dormancy
done
clear
C)
inhibiting the abscission
done
clear
D)
inhibiting the cell division
done
clear
View Answer play_arrow
question_answer 306) The endosperm of gymnosperm is:
A)
haploid
done
clear
B)
diploid
done
clear
C)
triploid
done
clear
D)
tetraploid
done
clear
View Answer play_arrow
question_answer 307) Which type of embryo development occurs in Capsella ?
A)
Crucifer type
done
clear
B)
Solanoid type
done
clear
C)
Aestroid type
done
clear
D)
Mixed and irregular type
done
clear
View Answer play_arrow
question_answer 308) Which of the following takes place in \[{{G}_{1}}\] of in terphase?
A)
Active synthesis of RNA
done
clear
B)
Active synthesis of protein
done
clear
C)
Active synthesis of DNA
done
clear
D)
Synthesis of RNA and protein
done
clear
View Answer play_arrow
question_answer 309) Which type of movement is present in Mimosa pudica?
A)
Nycnastic
done
clear
B)
Seismonastic
done
clear
C)
Chemonastic
done
clear
D)
Phototrophic
done
clear
View Answer play_arrow
question_answer 310) Cotton is obtained from which part of the plant:
A)
fruit
done
clear
B)
seed coat
done
clear
C)
pericarp
done
clear
D)
embryo
done
clear
View Answer play_arrow
question_answer 311) In eukaryotic cell, m-RNA synthesis takes place in:
A)
nucleus
done
clear
B)
nucleolus
done
clear
C)
cytoplasm
done
clear
D)
rough endoplasmic reticulum
done
clear
View Answer play_arrow
question_answer 312) In which of the following manner distribution of stomata occurs in dorsiventral leaf?
A)
More in palisade tissue and less in spongy parenchyma
done
clear
B)
Equal in both palisade and spongy parenchyma
done
clear
C)
More in spongy parenchyma and less in palisade tissue
done
clear
D)
Absent in all of these
done
clear
View Answer play_arrow
question_answer 313) Capselta shows characteristic of which of the following family?
A)
Cruciferae
done
clear
B)
Solanaceae
done
clear
C)
Liliaceae
done
clear
D)
Compositae
done
clear
View Answer play_arrow
question_answer 314) Chemical composition of protoplasm contains:
A)
O, C, H and N
done
clear
B)
O, N and Fe
done
clear
C)
O, protein and minerals
done
clear
D)
H, C and Fe
done
clear
View Answer play_arrow
question_answer 315) In which of the following the pollen tube discharge their contents during fertilization?
A)
In synergid
done
clear
B)
In egg
done
clear
C)
In antipodals
done
clear
D)
In embryo sac
done
clear
View Answer play_arrow
question_answer 316) During conjugation the two bacterial cells attach with:
A)
pili
done
clear
B)
flagella
done
clear
C)
cilia
done
clear
D)
hairs
done
clear
View Answer play_arrow
question_answer 317) Group of genes present in an ecosystem is called as:
A)
genome
done
clear
B)
gene pool
done
clear
C)
genotype
done
clear
D)
phenotype
done
clear
View Answer play_arrow
question_answer 318) The important thing for the formation of new species is:
A)
sexual reproduction
done
clear
B)
variations
done
clear
C)
inbreeding
done
clear
D)
reproductive isolcition
done
clear
View Answer play_arrow
question_answer 319) Pigment innolved in the perception of the photo periodic stimulus is :
A)
cytochrome
done
clear
B)
phytochrome
done
clear
C)
chlorophyll
done
clear
D)
carotenoids
done
clear
View Answer play_arrow
question_answer 320) The special structure present in \[{{C}_{4}}\] plants is:
A)
thin cuticle
done
clear
B)
multi layered epidermis
done
clear
C)
Kranz type body
done
clear
D)
one layered epidermis
done
clear
View Answer play_arrow
question_answer 321) White rust of Brassicu is caused by:
A)
Albugo candida
done
clear
B)
Ustilago
done
clear
C)
Phytophthera
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 322) Function of rough endoplasmic reticulum is:
A)
fat synthesis
done
clear
B)
protein synthesis
done
clear
C)
starch synthesis
done
clear
D)
autolysis
done
clear
View Answer play_arrow
question_answer 323) Which of the following is a viral causing disease?
A)
Leaf curl of Papaya
done
clear
B)
Red rot of Sugarcane
done
clear
C)
Black rust of Wheat
done
clear
D)
Late blight of Potato
done
clear
View Answer play_arrow
question_answer 324) If organic acids arc used as substrate what will be the value of R.Q?
A)
1
done
clear
B)
> 1
done
clear
C)
< 1
done
clear
D)
0
done
clear
View Answer play_arrow
question_answer 325) Co-factor required for the formation of acetyle CoA is:
A)
TPP
done
clear
B)
lipoic acid
done
clear
C)
\[M{{g}^{+2}}\] and CoA
done
clear
D)
all of the above
done
clear
View Answer play_arrow
question_answer 326) All number of genotypes for Aa Bb Cc will be:
A)
8
done
clear
B)
4
done
clear
C)
12
done
clear
D)
16
done
clear
View Answer play_arrow
question_answer 327) Tetradynamous androecium is found in:
A)
mustard
done
clear
B)
onion
done
clear
C)
tomato
done
clear
D)
sunflower
done
clear
View Answer play_arrow
question_answer 328) Cell wall of green algae is made up of:
A)
cellulose
done
clear
B)
chitin
done
clear
C)
glycogen
done
clear
D)
muramic acid
done
clear
View Answer play_arrow
question_answer 329) Hnploid number of chromosomes in a cell are 10, what will be the number of pairs of homologous chromosomes:
A)
5
done
clear
B)
10
done
clear
C)
15
done
clear
D)
20
done
clear
View Answer play_arrow
question_answer 330) In mitochondria, enzymes responsible for Krebs cycle are present in :
A)
matrix
done
clear
B)
inner membrane
done
clear
C)
outer membrane
done
clear
D)
both (a) and (b)
done
clear
View Answer play_arrow
question_answer 331) Gymnospenn may be distinguish from angiospcrm by which of the following characteristic ?
A)
Absence of true seed
done
clear
B)
Absence of xerophytic nature
done
clear
C)
Absence of secondary growth
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 332) Phallogen is also referred as:
A)
pericycle
done
clear
B)
cork cambium
done
clear
C)
mcristem
done
clear
D)
ratidom
done
clear
View Answer play_arrow
question_answer 333) In ferns and mosses, movement of anthcrozoids towards female component is called:
A)
pholotaxis
done
clear
B)
chemotaxis
done
clear
C)
hydrotropism
done
clear
D)
thigmotropism
done
clear
View Answer play_arrow
question_answer 334) What will be the genotypic ratio of \[{{F}_{2}}\] generation in dihybrid cross:
A)
12 : 3 :1
done
clear
B)
9 : 3 : 3 : 1
done
clear
C)
1 : 2 : 2 : 4 : 1 : 2 : 1 : 2 : 1
done
clear
D)
15 : 1
done
clear
View Answer play_arrow
question_answer 335) Plasmid is a:
A)
fungus
done
clear
B)
plastid
done
clear
C)
part of plasma membrane
done
clear
D)
extra chromosomal DNA in bacterial cell
done
clear
View Answer play_arrow
question_answer 336) The reason for apical do minance is:
A)
ethylene
done
clear
B)
auxin
done
clear
C)
cytokinin
done
clear
D)
gibberellins
done
clear
View Answer play_arrow
question_answer 337) Witches broom in Potato plant is caused by:
A)
bacteria
done
clear
B)
virus
done
clear
C)
fungus
done
clear
D)
mycoplasma
done
clear
View Answer play_arrow
question_answer 338) Which of the following is monocarpic?
A)
Coconut
done
clear
B)
Mango
done
clear
C)
Apple
done
clear
D)
Bamboo
done
clear
View Answer play_arrow
question_answer 339) A plant bearing different type of leaves is called:
A)
heterophylly
done
clear
B)
hetcrotrophy
done
clear
C)
hetcrozygosity
done
clear
D)
heteronasty
done
clear
View Answer play_arrow
question_answer 340) Prothallus of Pteridium bears:
A)
anthcridia on upper surface anal archcgonia on lower surface
done
clear
B)
anthcridia on lower surface and archegonia on upper surface
done
clear
C)
both archegonia and antheridia on upper surface
done
clear
D)
both antheridia and archegonia on lower surface
done
clear
View Answer play_arrow
question_answer 341) Balloon like out growth formed in xylem vessels is called:
A)
tylosis
done
clear
B)
histogen
done
clear
C)
callus
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 342) RNA molecule joins new nucleotides by which of the following bonds?
A)
Phospho diester bond
done
clear
B)
Polypeptide bond
done
clear
C)
Covalent bond
done
clear
D)
Hydrogen bond
done
clear
View Answer play_arrow
question_answer 343) The rate of transpiration is measured by:
A)
micrometer
done
clear
B)
photometer
done
clear
C)
auxinometer
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 344) Which of the following have correct group of Malvaceae members?
A)
Gossypium, Hibiscus, Abutilon
done
clear
B)
Gossypium, Brassica, Lycopersicum
done
clear
C)
Brassica, Gossypium, Hibiscus
done
clear
D)
Gossypium, Hibiscus, Lycopersicum
done
clear
View Answer play_arrow
question_answer 345) The flow of the energy in an ecosystem is:
A)
unidirectional
done
clear
B)
multidirectional
done
clear
C)
bidirectional
done
clear
D)
irregular
done
clear
View Answer play_arrow
question_answer 346) Which of the following is a part of a epidermal layer?
A)
Guard cell
done
clear
B)
Root hairs
done
clear
C)
Trichomes
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 347) Respiratory roots and viviparous reproduction are the characteristic of:
A)
hydrophytes
done
clear
B)
xerophytes
done
clear
C)
mesophytes
done
clear
D)
mangroves
done
clear
View Answer play_arrow
question_answer 348) What is nucleosome?
A)
DNA wrapped around histone protein
done
clear
B)
RNA wrapped around histone protein
done
clear
C)
Polymer
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 349) The father of binomial nomenclature was:
A)
Linnaeus
done
clear
B)
Engler and Prantl
done
clear
C)
Bentham and Hooker
done
clear
D)
Aristotle
done
clear
View Answer play_arrow
question_answer 350) Living fossil is:
A)
Ephedra
done
clear
B)
Ginkgo biloba
done
clear
C)
Equisetum
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 351) An ecosystem is constructing by: (1) producers (2) primary consumer (3) secondary consumer (4) decomposers Which of the above has highest energy?
A)
1
done
clear
B)
2
done
clear
C)
3
done
clear
D)
4
done
clear
View Answer play_arrow
question_answer 352) Commercial opium is obtained from:
A)
mature leaves
done
clear
B)
mature seeds
done
clear
C)
unripened capsule
done
clear
D)
germinated plants
done
clear
View Answer play_arrow
question_answer 353) When viral DNA is integrated with that of bacterial DNA the phenomenon is known as:
A)
prophase
done
clear
B)
lytic cycle
done
clear
C)
lysogenic cycle
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 354) Transposable element-I was discovered from:
A)
Maize
done
clear
B)
Wheat
done
clear
C)
Gram
done
clear
D)
Pea
done
clear
View Answer play_arrow
question_answer 355) Which of the following is fruit sugar?
A)
Glucose
done
clear
B)
Fructose
done
clear
C)
Ribose
done
clear
D)
Sucrose
done
clear
View Answer play_arrow
question_answer 356) In angiosperms which form of the carbohydrate is translocated:
A)
fructose
done
clear
B)
glucose
done
clear
C)
sucrose
done
clear
D)
ribose
done
clear
View Answer play_arrow
question_answer 357) The net gain of ATP in anaerobic respiration is:
A)
4 ATP
done
clear
B)
8 ATP
done
clear
C)
2 ATP
done
clear
D)
3 ATP
done
clear
View Answer play_arrow
question_answer 358) A monocot plant which shows secondary growth in stem is:
A)
Sugarcane
done
clear
B)
Yucca
done
clear
C)
Maize
done
clear
D)
Coconut
done
clear
View Answer play_arrow
question_answer 359) Amphivasal vascular bundle possess:
A)
xylem around phloem
done
clear
B)
phloem around xylem
done
clear
C)
phloem on both sides of xylem
done
clear
D)
phloem towards centre and xylem towards periphery
done
clear
View Answer play_arrow
question_answer 360) Sporophyte of Riccia contains:
A)
elaters and spores
done
clear
B)
spores and nutritive cells
done
clear
C)
only spores
done
clear
D)
both (a) and (b)
done
clear
View Answer play_arrow
question_answer 361) Zygotic division in Capselh bursa takes place by:
A)
longitudinal division
done
clear
B)
equal transverse division
done
clear
C)
unequal transverse division
done
clear
D)
oblique division
done
clear
View Answer play_arrow
question_answer 362) Mendels law of independent assortment can be proved by:
A)
back cross
done
clear
B)
test cross
done
clear
C)
monohybrid cross
done
clear
D)
dihybrid cross
done
clear
View Answer play_arrow
question_answer 363) During development of embryo in archegonium of bryophyta its posterior part form protective embryo cover which is called:
A)
calyptra
done
clear
B)
paraphysis
done
clear
C)
apophysis
done
clear
D)
hypophysis
done
clear
View Answer play_arrow
question_answer 364) Synaptonemal complex forms in:
A)
leptotene
done
clear
B)
zygotene
done
clear
C)
pachytene
done
clear
D)
diplotene
done
clear
View Answer play_arrow
question_answer 365) Scopolamine alkaloids obtains from:
A)
Datura stramonium
done
clear
B)
Atropa belladonna
done
clear
C)
Curcuma longa
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 366) In which of the following three radial vascular bundles are present?
A)
Monocot root
done
clear
B)
Dicot root
done
clear
C)
Monocot stem
done
clear
D)
Dicot stem
done
clear
View Answer play_arrow
question_answer 367) Floral formula \[\oplus _{\begin{smallmatrix} O \\ + \end{smallmatrix}}^{\uparrow }\,{{K}_{(5)}}\,{{C}_{(5)}}\,{{A}_{\infty }}\,{{G}_{(5)}}\] belongs to family:
A)
Malvaceae
done
clear
B)
Crudferae
done
clear
C)
Compositae
done
clear
D)
Solanaceae
done
clear
View Answer play_arrow
question_answer 368) Bicarpillary, syncarpous bilocular axile placentation and oblique septum are characteristic of:
A)
Solanaceae
done
clear
B)
Crucifereae
done
clear
C)
Malvaceae
done
clear
D)
Compositae
done
clear
View Answer play_arrow
question_answer 369) Cork cambium and cambium are the example of:
A)
lateral meristem
done
clear
B)
primary meristem
done
clear
C)
intercalary meristem
done
clear
D)
apical meristem
done
clear
View Answer play_arrow
question_answer 370) Thigmotropism is shown by:
A)
leaf let
done
clear
B)
stem
done
clear
C)
shoot apex
done
clear
D)
tendril
done
clear
View Answer play_arrow
question_answer 371) In an example of lateral conjugation involving three Spirogyra filaments zoospores will form in :
A)
all three filaments
done
clear
B)
two lateral filaments
done
clear
C)
only middle filaments
done
clear
D)
either middle or lateral filaments
done
clear
View Answer play_arrow
question_answer 372) Bacteriophage is consist of:
A)
only proteins
done
clear
B)
nucleoproteins
done
clear
C)
protein and lipid
done
clear
D)
only DNA
done
clear
View Answer play_arrow
question_answer 373) In which part of Funaria stomatas are present?
A)
Stem
done
clear
B)
Leaf
done
clear
C)
Capsule
done
clear
D)
Foot
done
clear
View Answer play_arrow
question_answer 374) Seed habit is developed first time in:
A)
some ferns
done
clear
B)
some pinus
done
clear
C)
some monocots
done
clear
D)
some dicots
done
clear
View Answer play_arrow
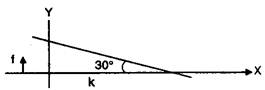
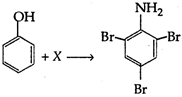 X is:
X is:
