A) trimethyl acetaldehyde
B) acetaldehyde
C) benzaldehyde
D) formaldehyde
Correct Answer: B
Solution :
\[~~\underset{\text{trimethyl}\,\text{acetaldehyde}}{\mathop{C{{H}_{3}}-\underset{C{{H}_{3}}}{\overset{C{{H}_{3}}}{\mathop{\underset{|}{\overset{|}{\mathop{C}}}\,}}}\,-\overset{H}{\mathop{\overset{|}{\mathop{C}}\,}}\,=O}}\,\] \[\underset{\text{acetaldehyde}}{\mathop{{{H}_{\alpha }}-\underset{{{H}_{\alpha }}}{\overset{{{H}_{\alpha }}}{\mathop{\underset{|}{\overset{|}{\mathop{C}}}\,}}}\,-\overset{H}{\mathop{\overset{|}{\mathop{C}}\,}}\,=O,}}\,\]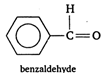 \[\underset{\text{formaldehyde}}{\mathop{H-\overset{H}{\mathop{\overset{|}{\mathop{C}}\,}}\,=0}}\,\] Cannizaro reaction is given by only those aldehydes which do not have \[\alpha -\]hydrogen atom. As such acetaldehyde will not give Cannizaro reaction.
\[\underset{\text{formaldehyde}}{\mathop{H-\overset{H}{\mathop{\overset{|}{\mathop{C}}\,}}\,=0}}\,\] Cannizaro reaction is given by only those aldehydes which do not have \[\alpha -\]hydrogen atom. As such acetaldehyde will not give Cannizaro reaction.
You need to login to perform this action.
You will be redirected in
3 sec
