Answer:
(b, c)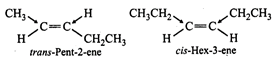 Since the \[+I\] effect of \[C{{H}_{2}}C{{H}_{3}}\]
group is higher than that of\[C{{H}_{3}}\] group, therefore, the dipole moment \[C-C{{H}_{3}}\]
and\[C-C{{H}_{2}}C{{H}_{3}}\] bonds are unequal. Although these two dipoles
oppose each other, yet they do not exactly cancel out each other and hence
trans-2-pentene has small but finite dipole moment.
In cis-hex-3-ene, although the
dipole moments of the two \[C-C{{H}_{2}}C{{H}_{3}}\] bond are equal, but are
inclined to each other at an angle of \[60{}^\circ \] and hence have a finite dipole moment.
Since the \[+I\] effect of \[C{{H}_{2}}C{{H}_{3}}\]
group is higher than that of\[C{{H}_{3}}\] group, therefore, the dipole moment \[C-C{{H}_{3}}\]
and\[C-C{{H}_{2}}C{{H}_{3}}\] bonds are unequal. Although these two dipoles
oppose each other, yet they do not exactly cancel out each other and hence
trans-2-pentene has small but finite dipole moment.
In cis-hex-3-ene, although the
dipole moments of the two \[C-C{{H}_{2}}C{{H}_{3}}\] bond are equal, but are
inclined to each other at an angle of \[60{}^\circ \] and hence have a finite dipole moment.
You need to login to perform this action.
You will be redirected in
3 sec
