-
question_answer1)
When an ideal diatomic gas is heated at constant pressure, the fraction of the heat energy supplied which increases the internal energy of the gas, is [IIT 1990; UPSEAT 1998; RPET 2000]
A)
\[\frac{2}{5}\] done
clear
B)
\[\frac{3}{5}\] done
clear
C)
\[\frac{3}{7}\] done
clear
D)
\[\frac{5}{7}\] done
clear
View Solution play_arrow
-
question_answer2)
\[1c{{m}^{3}}\] of water at its boiling point absorbs 540 calories of heat to become steam with a volume of \[1671c{{m}^{3}}\].If the atmospheric pressure = \[1.013x{{10}^{5}}N/{{m}^{2}}\] and the mechanical equivalent of heat = \[4.19J/calorie\], the energy spent in this process in overcoming intermolecular forces is [MP PET 1999, 2001; Orissa JEE 2002]
A)
540 cal done
clear
B)
40 cal done
clear
C)
500 cal done
clear
D)
Zero done
clear
View Solution play_arrow
-
question_answer3)
During the melting of a slab of ice at 273 K at atmospheric pressure [IIT 1998]
A)
Positive work is done by ice-water system on the atmosphere done
clear
B)
Positive work is done on the ice-water system by the atmosphere done
clear
C)
The internal energy of the ice-water system increases done
clear
D)
The internal energy of the ice-water system decreases done
clear
View Solution play_arrow
-
question_answer4)
Two identical containers A and B with frictionless pistons contain the same ideal gas at the same temperature and the same volume V. The mass of the gas in A is \[{{m}_{A}}\] and that in B is \[{{m}_{B}}\]. The gas in each cylinder is now allowed to expand isothermally to the same final volume 2V. The changes in the pressure in A and B are found to be \[\Delta P\] and 1.5 \[\Delta P\] respectively. Then [IIT 1998]
A)
\[4{{m}_{A}}=9{{m}_{B}}\] done
clear
B)
\[2{{m}_{A}}=3{{m}_{B}}\] done
clear
C)
\[3{{m}_{A}}=2{{m}_{B}}\] done
clear
D)
\[9{{m}_{A}}=3{{m}_{B}}\] done
clear
View Solution play_arrow
-
question_answer5)
A monoatomic ideal gas, initially at temperature \[{{T}_{1}},\] is enclosed in a cylinder fitted with a frictionless piston. The gas is allowed to expand adiabatically to a temperature. \[{{T}_{2}}\] by releasing the piston suddenly. If \[{{L}_{1}}\] and \[{{L}_{2}}\]are the lengths of the gas column before and after expansion respectively, then \[{{T}_{1}}/{{T}_{2}}\] is given by [IIT-JEE (Screening) 2000]
A)
\[{{\left( \frac{{{L}_{1}}}{{{L}_{2}}} \right)}^{2/3}}\] done
clear
B)
\[\frac{{{L}_{1}}}{{{L}_{2}}}\] done
clear
C)
\[\frac{{{L}_{2}}}{{{L}_{1}}}\] done
clear
D)
\[{{\left( \frac{{{L}_{2}}}{{{L}_{1}}} \right)}^{2/3}}\] done
clear
View Solution play_arrow
-
question_answer6)
A closed hollow insulated cylinder is filled with gas at \[{{0}^{o}}C\] and also contains an insulated piston of negligible weight and negligible thickness at the middle point. The gas on one side of the piston is heated to \[{{100}^{o}}C.\] If the piston moves \[5\,cm,\] the length of the hollow cylinder is [EAMCET 2001]
A)
13.65 cm done
clear
B)
27.3 cm done
clear
C)
38.6 cm done
clear
D)
64.6 cm done
clear
View Solution play_arrow
-
question_answer7)
A mono atomic gas is supplied the heat Q very slowly keeping the pressure constant. The work done by the gas will be [BHU 2003; CPMT 2004]
A)
\[\frac{2}{3}Q\] done
clear
B)
\[\frac{3}{5}Q\] done
clear
C)
\[\frac{2}{5}Q\] done
clear
D)
\[\frac{1}{5}Q\] done
clear
View Solution play_arrow
-
question_answer8)
A gas mixture consists of 2 moles of oxygen and 4 moles argon at temperature T. Neglecting all vibrational modes, the total internal energy of the system is [IIT 1999; UPSEAT 2003]
A)
4 RT done
clear
B)
15 RT done
clear
C)
9 RT done
clear
D)
11 RT done
clear
View Solution play_arrow
-
question_answer9)
An ideal gas expands isothermally from a volume \[{{V}_{1}}\] to \[{{V}_{2}}\] and then compressed to original volume \[{{V}_{1}}\]adiabatically. Initial pressure is \[{{P}_{1}}\] and final pressure is \[{{P}_{3}}\]. The total work done is W. Then [IIT-JEE (Screening) 2004]
A)
\[{{P}_{3}}>{{P}_{1}},\,\,W>0\] done
clear
B)
\[{{P}_{3}}<{{P}_{1}},\,\,W<0\] done
clear
C)
\[{{P}_{3}}>{{P}_{1}},\,\,W<0\] done
clear
D)
\[{{P}_{3}}={{P}_{1}},\,\,W=0\] done
clear
View Solution play_arrow
-
question_answer10)
Work done by a system under isothermal change from a volume \[{{V}_{1}}\] to \[{{V}_{2}}\] for a gas which obeys Vander Waal's equation \[(V-\beta n)\,\left( P+\frac{\alpha {{n}^{2}}}{V} \right)=nRT\]
A)
\[nRT{{\log }_{e}}\left( \frac{{{V}_{2}}-n\beta }{{{V}_{1}}-n\beta } \right)+\alpha \,{{n}^{2}}\,\left( \frac{{{V}_{1}}-{{V}_{2}}}{{{V}_{1}}{{V}_{2}}} \right)\] done
clear
B)
\[nRT{{\log }_{10}}\left( \frac{{{V}_{2}}-\alpha \beta }{{{V}_{1}}-\alpha \beta } \right)+\alpha \,{{n}^{2}}\,\left( \frac{{{V}_{1}}-{{V}_{2}}}{{{V}_{1}}{{V}_{2}}} \right)\] done
clear
C)
\[nRT{{\log }_{e}}\left( \frac{{{V}_{2}}-n\alpha }{{{V}_{1}}-n\alpha } \right)+\beta \,{{n}^{2}}\,\left( \frac{{{V}_{1}}-{{V}_{2}}}{{{V}_{1}}{{V}_{2}}} \right)\] done
clear
D)
\[nRT{{\log }_{e}}\left( \frac{{{V}_{1}}-n\beta }{{{V}_{2}}-n\beta } \right)+\alpha \,{{n}^{2}}\,\left( \frac{{{V}_{1}}{{V}_{2}}}{{{V}_{1}}-{{V}_{2}}} \right)\] done
clear
View Solution play_arrow
-
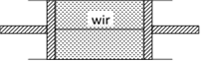
question_answer11)
A cylindrical tube of uniform cross-sectional area A is fitted with two air tight frictionless pistons. The pistons are connected to each other by a metallic wire. Initially the pressure of the gas is P0 and temperature is T0, atmospheric pressure is also P0. Now the temperature of the gas is increased to 2T0, the tension in the wire will be
A)
\[2{{P}_{0}}A\] done
clear
B)
\[{{P}_{0}}A\] done
clear
C)
\[\frac{{{P}_{0}}A}{2}\] done
clear
D)
\[4{{P}_{0}}A\] done
clear
View Solution play_arrow
-
question_answer12)
The molar heat capacity in a process of a diatomic gas if it does a work of \[\frac{Q}{4}\]when a heat of Q is supplied to it is
A)
\[\frac{2}{5}R\] done
clear
B)
\[\frac{5}{2}R\] done
clear
C)
\[\frac{10}{3}R\] done
clear
D)
\[\frac{6}{7}R\] done
clear
View Solution play_arrow
-
question_answer13)
An insulator container contains 4 moles of an ideal diatomic gas at temperature T. Heat Q is supplied to this gas, due to which 2 moles of the gas are dissociated into atoms but temperature of the gas remains constant. Then
A)
\[Q=2RT\] done
clear
B)
\[Q=RT\] done
clear
C)
\[Q=3RT\] done
clear
D)
\[Q=4RT\] done
clear
View Solution play_arrow
-
question_answer14)
The volume of air increases by 5% in its adiabatic expansion. The percentage decrease in its pressure will be
A)
5% done
clear
B)
6% done
clear
C)
7% done
clear
D)
8% done
clear
View Solution play_arrow
-
question_answer15)
The temperature of a hypothetical gas increases to \[\sqrt{2}\] times when compressed adiabatically to half the volume. Its equation can be written as
A)
\[P{{V}^{3/2}}\]= constant done
clear
B)
\[P{{V}^{5/2}}\]= constant done
clear
C)
\[P{{V}^{7/3}}\]= constant done
clear
D)
\[P{{V}^{4/3}}\]= constant done
clear
View Solution play_arrow
-
question_answer16)
Two Carnot engines A and B are operated in succession. The first one, A receives heat from a source at \[{{T}_{1}}=800k\] and rejects to sink at \[{{T}_{2}}k.\]. The second engine B receives heat rejected by the first engine and rejects to another sink at \[{{T}_{3}}=300k.\] If the work outputs of two engines are equal, then the value of \[{{T}_{2}}\] is
A)
100K done
clear
B)
300K done
clear
C)
550K done
clear
D)
700K done
clear
View Solution play_arrow
-
question_answer17)
When an ideal monoatomic gas is heated at constant pressure, fraction of heat energy supplied which increases the internal energy of gas, is [AIIMS 1995]
A)
\[\frac{2}{5}\] done
clear
B)
\[\frac{3}{5}\] done
clear
C)
\[\frac{3}{7}\] done
clear
D)
\[\frac{3}{4}\] done
clear
View Solution play_arrow
-
question_answer18)
When an ideal gas \[(\gamma =5/3\]) is heated under constant pressure, then what percentage of given heat energy will be utilised in doing external work [RPET 1999]
A)
40 % done
clear
B)
30 % done
clear
C)
60 % done
clear
D)
20 % done
clear
View Solution play_arrow
-
question_answer19)
Which one of the following gases possesses the largest internal energy [SCRA 1998]
A)
2 moles of helium occupying \[1{{m}^{3}}\]at 300 K done
clear
B)
56 kg of nitrogen at \[107N{{m}^{-2}}\] and 300 K done
clear
C)
8 grams of oxygen at 8 atm and 300 K done
clear
D)
\[6\times {{10}^{26}}\] molecules of argon occupying \[40{{m}^{3}}\] at 900 K done
clear
View Solution play_arrow
-
question_answer20)
Two samples A and B of a gas initially at the same pressure and temperature are compressed from volume V to V/2 (A isothermally and adiabatically). The final pressure of A is [MP PET 1996, 99; MP PMT 1997, 99]
A)
Greater than the final pressure of B done
clear
B)
Equal to the final pressure of B done
clear
C)
Less than the final pressure of B done
clear
D)
Twice the final pressure of B done
clear
View Solution play_arrow
-
question_answer21)
Initial pressure and volume of a gas are P and V respectively. First it is expanded isothermally to volume 4V and then compressed adiabatically to volume V. The final pressure of gas will be [CBSE PMT 1999]
A)
1P done
clear
B)
2P done
clear
C)
4P done
clear
D)
8P done
clear
View Solution play_arrow
-
question_answer22)
A thermally insulated rigid container contains an ideal gas heated by a filament of resistance 100 W through a current of 1A for 5 min then change in internal energy is [IIT-JEE (Screening) 2005]
A)
0 kJ done
clear
B)
10 kJ done
clear
C)
20 kJ done
clear
D)
30 kJ done
clear
View Solution play_arrow
-
question_answer23)
A reversible engine converts one-sixth of the heat input into work. When the temperature of the sink is reduced by 62°C, the efficiency of the engine is doubled. The temperatures of the source and sink are [CBSE PMT 2000]
A)
80°C, 37°C done
clear
B)
95°C, 28°C done
clear
C)
90°C, 37°C done
clear
D)
99°C, 37°C done
clear
View Solution play_arrow
-
question_answer24)
An engineer claims to have made an engine delivering 10 kW power with fuel consumption of 1 g/sec. The calorific value of the fuel is 2 kcal/g. Is the claim of the engineer [J & K CET 2000]
A)
Valid done
clear
B)
Invalid done
clear
C)
Depends on engine design done
clear
D)
Depends of the load done
clear
View Solution play_arrow
-
question_answer25)
Find the change in the entropy in the following process 100 gm of ice at 0°C melts when dropped in a bucket of water at 50°C (Assume temperature of water does not change) [BHU (Med.) 2000]
A)
? 4.5 cal/K done
clear
B)
+ 4.5 cal/K done
clear
C)
+5.4 cal/K done
clear
D)
? 5.4 cal/K done
clear
View Solution play_arrow
-
question_answer26)
An ideal gas expands in such a manner that its pressure and volume can be related by equation \[P{{V}^{2}}=\] constant. During this process, the gas is [UPSEAT 2002]
A)
Heated done
clear
B)
Cooled done
clear
C)
Neither heated nor cooled done
clear
D)
First heated and then cooled done
clear
View Solution play_arrow
-
question_answer27)
A Carnot engine whose low temperature reservoir is at 7°C has an efficiency of 50%. It is desired to increase the efficiency to 70%. By how many degrees should the temperature of the high temperature reservoir be increased [UPSEAT 2005]
A)
840 K done
clear
B)
280 K done
clear
C)
560 K done
clear
D)
380 K done
clear
View Solution play_arrow
-
question_answer28)
P-V diagram of a diatomic gas is a straight line passing through origin. The molar heat capacity of the gas in the process will be
A)
4 R done
clear
B)
2.5 R done
clear
C)
3 R done
clear
D)
\[\frac{4R}{3}\] done
clear
View Solution play_arrow
-
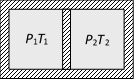
question_answer29)
Following figure shows on adiabatic cylindrical container of volume \[{{V}_{0}}\] divided by an adiabatic smooth piston (area of cross-section = A) in two equal parts. An ideal gas \[({{C}_{P}}/{{C}_{V}}=\gamma )\] is at pressure P1 and temperature T1 in left part and gas at pressure P2 and temperature T2 in right part. The piston is slowly displaced and released at a position where it can stay in equilibrium. The final pressure of the two parts will be (Suppose x = displacement of the piston)
A)
\[{{P}_{2}}\] done
clear
B)
\[{{P}_{1}}\] done
clear
C)
\[\frac{{{P}_{1}}{{\left( \frac{{{V}_{0}}}{2} \right)}^{\gamma }}}{{{\left( \frac{{{V}_{0}}}{2}+Ax \right)}^{\gamma }}}\] done
clear
D)
\[\frac{{{P}_{2}}{{\left( \frac{{{V}_{0}}}{2} \right)}^{\gamma }}}{{{\left( \frac{{{V}_{0}}}{2}+Ax \right)}^{\gamma }}}\] done
clear
View Solution play_arrow
-
question_answer30)
Two cylinders A and B fitted with pistons contain equal amounts of an ideal diatomic gas at 300 K. The piston of A is free to move while that of B is held fixed. The same amount of heat is given to the gas in each cylinder. If the rise in temperature of the gas in A is 30 K, then the rise in temperature of the gas in B is [IIT 1998]
A)
30 K done
clear
B)
18 K done
clear
C)
50 K done
clear
D)
42 K done
clear
View Solution play_arrow
