Answer:
The atomic number (Z) for carbon is six and its electronic configuration is 2, 4. Carbon has four
valence electrons. Each oxygen atom (Z = 8) has six valence electrons (2, 6). In order to complete
its octet, the carbon atom shares its four valence electrons with the four electrons of the two oxygen
atoms as follows :
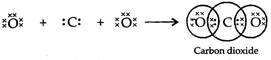 Or
Or ![]() Thus, in carbon dioxide molecule/ the carbon atom is linked to two oxygen atoms on both sides by two shared pairs of electrons resulting in double bonds on either sides. Both carbon and oxygen atoms complete their octet as a result of electron sharing.
Thus, in carbon dioxide molecule/ the carbon atom is linked to two oxygen atoms on both sides by two shared pairs of electrons resulting in double bonds on either sides. Both carbon and oxygen atoms complete their octet as a result of electron sharing.
You need to login to perform this action.
You will be redirected in
3 sec
