Answer:
The reactions of zinc granules with various
reagents are as follows
(i) Dilute![]()
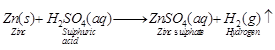 (ii) Dilute HCl
(ii) Dilute HCl
![]() (iii) Dilute HNO3 is an
oxidising acid therefore, its reaction with a metal(e.g., Zn) is different than
other dilute acids.
(iii) Dilute HNO3 is an
oxidising acid therefore, its reaction with a metal(e.g., Zn) is different than
other dilute acids.
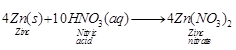
![]() (iv) NaCl solution Reaction does not
take place
(iv) NaCl solution Reaction does not
take place
![]() (v) NaOH solution
(v) NaOH solution
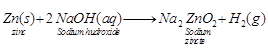 This hydrogen gas is evolved with
This hydrogen gas is evolved with![]() ,
HCl and NaOH, nitrous oxide gas is evolved with dilute HNO3 and no
reaction takes place with NaCI solution.
,
HCl and NaOH, nitrous oxide gas is evolved with dilute HNO3 and no
reaction takes place with NaCI solution.
You need to login to perform this action.
You will be redirected in
3 sec
