-
Standard electrode potential
values, \[{{E}^{{}^\circ }}\]for \[A{{l}^{3+}}/Al\]is -1.66 V and that of\[T{{l}^{3+}}/Tl\]
is +1.26 V. Predict about the formation of \[{{M}^{3+}}\] ion in solution and
compare the electropositive character of the two metals.
View Answer play_arrow
-
White fumes appear around the
bottle of anhydrous aluminium chloride. Give reason.
View Answer play_arrow
-
Boron is unable to form \[BF_{6}^{3-}\]
ion. Explain.
View Answer play_arrow
-
Why is boric acid considered as a
weak acid?
View Answer play_arrow
-
Select the member(s) of group 14 that :
(i) forms the most acidic dioxide
(ii) is commonly found in +2 oxidation state
(iii) is used as a semiconductor.
View Answer play_arrow
-
\[{{[Si{{F}_{6}}]}^{2-}}\]is known
whereas \[{{[SiC{{l}_{6}}]}^{2-}}\]is not. Give possible reasons.
View Answer play_arrow
-
Diamond is covalent, yet it has a
high melting point. Why?
View Answer play_arrow
-
What are silicones?
View Answer play_arrow
-
Discuss the pattern of variation
in the oxidation states of: (i) B to \[Tl\](ii) C to\[Pb\].
View Answer play_arrow
-
How can you explain higher
stability of \[BC{{l}_{3}}\] as compared to \[TlC{{l}_{3}}\]?
View Answer play_arrow
-
Why does boron trifluoride behave as a Lewis acid?
View Answer play_arrow
-
Consider the compounds \[BC{{l}_{3}}\]
and\[CC{{l}_{4}}\]. How will they behave with water? Justify.
View Answer play_arrow
-
Is boric acid a protic acid?
Explain.
View Answer play_arrow
-
Explain what happens when boric
acid is heated.
View Answer play_arrow
-
Describe the shapes of \[B{{F}_{3}}\]and
\[{{[B{{F}_{4}}]}^{-}}\]. Assign the hybridization of boron in these species.
View Answer play_arrow
-
Write reactions to justify
amphoteric nature of aluminium.
View Answer play_arrow
-
What are electron deficient
compounds? Are \[BC{{l}_{3}}\]and \[SiC{{l}_{4}}\] electron deficient species?
Explain.
View Answer play_arrow
-
Write the resonance structures of \[CO_{3}^{2-}\]and\[HCO_{3}^{-}\].
View Answer play_arrow
-
What is the state of hybridization of carbon in:
(a) \[CO_{3}^{2-}\], (b) diamond
and (c) graphite?
View Answer play_arrow
-
Explain the difference in
properties of diamond and graphite on the basis of their structures.
View Answer play_arrow
-
Rationalise
the given statements and give chemical reactions.
(i) Lead
(II) chloride reacts with \[C{{l}_{2}}\] to give\[PbC{{l}_{4}}\].
(ii)
Lead (IV) chloride is highly unstable towards heat.
(iii) Lead is known not to form an
iodide, \[Pb{{l}_{4}}\].
View Answer play_arrow
-
Suggest reasons why the BF bond
lengths in \[B{{F}_{3}}\](130 pm) and \[BF_{4}^{-}\] (143 pm) do differ?
View Answer play_arrow
-
If
B-Cl bond has a dipole moment, explain why\[BC{{l}_{3}}\] molecule has a zero
dipole moment?
View Answer play_arrow
-
Aluminium trifluoride is insoluble in anhydrous HF but
dissolves on addition of\[NaF\]. Aluminium trifluoride precipitates out of the
resulting solution when gaseous \[B{{F}_{3}}\] is bubbled through. Give
reasons.
View Answer play_arrow
-
Suggest a reason as to why CO is
poisonous?
View Answer play_arrow
-
How is excessive content of \[C{{O}_{2}}\]
responsible for global warming?
View Answer play_arrow
-
Explain the structures of diborane
and boric acid.
View Answer play_arrow
-
Explain what happens when :
(a) Borax is heated strongly?
(b) Boric acid is added to water?
(c) Aluminium is treated with\[NaOH\]?
(d)\[B{{F}_{3}}\] is reacted with ammonia ?
View Answer play_arrow
-
Explain
the following reactions :
(a) Silicon is heated with methyl chloride at high temperature
in presence of copper.
(b) CO is heated with\[ZnO\].
(c) Silicon dioxide is treated with hydrogen fluoride.
(d) Hydrated alumina is treated
with aqueous \[NaOH\]solution.
View Answer play_arrow
-
Give reasons:
(i) Conc. \[HN{{O}_{3}}\]can be transported in aluminium containers.
(ii) Graphite is used as a lubricant.
(iii) Diamond is used as an abrasive.
(iv) A mixture of \[NaOH\]and aluminium pieces is used to
open the drain.
(v) Aluminium alloys are used to make aircraft body.
(vi) Aluminium utensils should not be kept in water overnight.
(vii) Aluminium wire is used to make transmission cables.
View Answer play_arrow
-
Explain why there is a phenomenal
decrease in ionisation enthalpy from carbon to silicon?
View Answer play_arrow
-
How would you explain the lower
atomic radius of \[Ga\]as compared to Al?
View Answer play_arrow
-
What are allotropes? Sketch the
structure of two allotropes of carbon namely diamond and graphite. What is the
impact of structure on physical properties of two allotropes?
View Answer play_arrow
-
Classify the following oxides as neutral, acidic, basic or amphoteric:
\[CO,{{B}_{2}}{{O}_{3}},Si{{O}_{2}},C{{O}_{2}},A{{l}_{2}}{{O}_{3}},Pb{{O}_{2}},T{{l}_{2}}{{O}_{3}}\]
View Answer play_arrow
-
In some of the reactions thallium
resembles aluminium, whereas in others it resembles with group \[I\] metals.
Support this statement by giving some evidences.
View Answer play_arrow
-
When metal X is treated with
sodium hydroxide; a white precipitate (A) is obtained, which is soluble in excess
of \[NaOH\]to give a soluble complex (B). Compound (A) is soluble in \[HCl\](dilute)
to form compound (C). The compound (A) when heated strongly gives (D), which is
used to extract metal. Identify (X), (A), (B), (C) and (D). Write suitable equations
to support their identities.
View Answer play_arrow
-
What do you understand by:
(a) inert pair effect (b)
allotropy (c) catenation?
View Answer play_arrow
-
A certain salt (X) gives the following results:
(i) Its solution is alkaline to litmus.
(ii) It swells upto glassy material \[(Y)\] on strong heating.
(iii) When concentrated \[{{H}_{2}}S{{O}_{4}}\]
is added to hot solution of (X), a white crystal of an acid (Z) separates out.
Write equations for all the above reactions and identify (X), (Y) and (Z).
View Answer play_arrow
-
Write balanced equations for:
(i) \[B{{F}_{3}}+LiH\to \]
(ii) \[{{B}_{2}}{{H}_{6}}+{{H}_{2}}O\to \]
(iii) \[NaH+{{B}_{2}}{{H}_{6}}\to \]
(iv)\[{{H}_{3}}B{{O}_{3}}\xrightarrow{Heat}\]
(v) \[Al+NaOH+{{H}_{2}}O\to \]
(vi)\[{{B}_{2}}{{H}_{6}}+N{{H}_{3}}\to \]
View Answer play_arrow
-
Give one method for industrial
preparation and one for laboratory preparation of CO and \[C{{O}_{2}}\] each.
View Answer play_arrow
-
An
aqueous solution of borax is:
(a) neutral (b) amphoteric (c)
basic (d) acidic
View Answer play_arrow
-
Boric acid is polymeric due to :
(i) its acidic nature
(ii) the presence of hydrogen bonds
(iii) the monobasic nature
(iv) its geometry
View Answer play_arrow
-
The
type of hybridisation of boron in diborane is :
(a) \[sp\] (b) \[s{{p}^{2}}\]
(c) \[s{{p}^{3}}\] (d)
\[ds{{p}^{2}}\]
View Answer play_arrow
-
Thermodynamically the most stable form of carbon is:
(a) diamond (b) graphite (c) fullerenes (d) coal
View Answer play_arrow
-
Elements of group 14:
(a) exhibit oxidation state of + 4 only
(b) exhibit oxidation state of + 2 and + 4
(c) form\[{{M}^{2+}}\] and\[{{M}^{4+}}\] ions
(d) form\[{{M}^{2-}}\] and \[{{M}^{4+}}\] ions.
View Answer play_arrow
-
If
the starting material for the manufacture of silicones is\[RSiC{{l}_{3}}\],
write the structure of the product formed.
View Answer play_arrow
-
question_answer47)
The element which exists in liquid state
for a wide range of temperature and can be used for measuring high temperature
is :
(a) B (b) Al (c)\[Ga\] (d)\[In\]
View Answer play_arrow
-
question_answer48)
Which of the following is a Lewis acid?
(a) \[AlC{{l}_{3}}\] (b)
\[MgC{{l}_{2}}\] (c)\[CaC{{l}_{2}}\] (d)
\[BaC{{l}_{2}}\]
View Answer play_arrow
-
question_answer49)
The geometry of a complex
species can be understood from the knowledge of type of hybridisation of
orbitals of central atom. The hybridisation of central atom in \[{{[B{{(OH)}_{4}}]}^{-}}\]
and the geometry of the complex are respectively :
(a)\[s{{p}^{3}}\],
tetrahedral (b) \[s{{p}^{3}}\],
square planar
(c) \[s{{p}^{3}}{{d}^{2}}\],
octahedral (d) \[ds{{p}^{2}}\],
square planar
View Answer play_arrow
-
question_answer50)
Which of the following oxides is
acidic in nature?
(a) \[{{B}_{2}}{{O}_{3}}\] (b)
\[A{{l}_{2}}{{O}_{3}}\] (c)\[G{{a}_{2}}{{O}_{3}}\] (d)
\[{{\ln }_{2}}{{O}_{3}}\]
View Answer play_arrow
-
question_answer51)
The exhibition of the highest
coordination number depends on the availability of vacant orbitals in the
central atom. Which of the following elements is not likely to act as central
atom in\[MF_{6}^{3-}\]?
(a) B (b)
Al (c)\[Ga\] (d)
In
View Answer play_arrow
-
question_answer52)
Boric acid is an acid because
the molecule :
(a) contains replaceable
hydrogen ion (b) gives up a proton
(c) accepts\[O{{H}^{-}}\] from
water releasing proton (d) combines with proton from water molecule
View Answer play_arrow
-
question_answer53)
Catenation, i.e., linking of
similar atoms depends on size and electronic configuration of atoms. The
tendency of catenation in group 14 elements follows the order :
(a) \[C\text{ }>\text{
}Si>\text{ }Ge\text{ }>\text{ }Sn\] (b)
\[C\text{ }>\text{ }Si\text{ }>\text{ }Ge\text{ }\approx \text{ }Sn\]
(c) \[Si\text{ }>\text{
}C\text{ }>\text{ }Sn\text{ }>\text{ }Ge\] (d)
\[Ge\text{ }>\text{ }Sn\text{ }>\text{ }Si\text{ }>\text{ }C\]
View Answer play_arrow
-
question_answer54)
Silicon has strong tendency to
form polymers like silicones.
The chain length of silicone
polymer can be controlled by adding:
(a)\[MeSiC{{l}_{3}}\] (b)
\[M{{e}_{2}}SiC{{l}_{2}}\] (c) \[M{{e}_{3}}SiCl\] (d)\[M{{e}_{4}}Si\]
View Answer play_arrow
-
question_answer55)
Ionisation enthalpy \[({{\Delta
}_{i}}{{H}_{i}}kJmo{{l}^{-1}})\] for the elements of group 13 follows the order
:
(a) \[B\text{ }>\text{
}Al\text{ }>\text{ }Ga\text{ }>\text{ }In\text{ }>\text{ }Tl\] (b)
\[B\text{ }<\text{ }Al\text{ }<\text{ }Ga\text{ }<\text{ }In\text{
}<\text{ }Tl\]
(c) \[B<Al>Ga<ln>Tl\] (d)\[B\text{
}>\text{ }Al\text{ }<\text{ }Ga\text{ }>\text{ }In\text{ }<\text{
}Tl\]
View Answer play_arrow
-
question_answer56)
In the structure of diborane:
(a) all hydrogen atoms lie in
one plane and boron atoms lie in a plane perpendicular to this plane
(b) 2 boron atoms and 4 terminal
hydrogen atoms lie in the same plane and 2 bridging hydrogen atoms lie in the perpendicular
plane
(c) 4 bridging hydrogen atoms
and boron lie in one plane and two terminal hydrogen atoms lie in a plane perpendicular
to this plane
(d) all the atoms are in the
same plane
View Answer play_arrow
-
question_answer57)
A compound X of boron reacts
with ammonia on heating to give another compound Y which is called inorganic benzene.
The compound X can be prepared by treating \[B{{F}_{3}}\]with lithium aluminium
hydride. The compound X and Y are represented by the formulae :
(a) \[{{B}_{2}}{{H}_{6}},{{B}_{3}}{{N}_{3}}{{H}_{6}}\] (b)
\[{{B}_{2}}{{O}_{3}},{{B}_{3}}{{N}_{3}}{{H}_{6}}\] (c)\[B{{F}_{3}},{{B}_{3}}{{N}_{3}}{{H}_{6}}\] (d)
\[{{B}_{3}}{{N}_{3}}{{H}_{6}},{{B}_{2}}{{H}_{6}}\]
View Answer play_arrow
-
question_answer58)
Quartz is extensively used as
piezoelectric material, it contains :
(a)\[Pb\] (b)\[Si\] (c)\[Ti\] (d)\[Sn\]
View Answer play_arrow
-
question_answer59)
The most commonly used reducing
agent is :
(a)\[AlC{{l}_{3}}\] (b)\[PbC{{l}_{2}}\] (c)\[SnC{{l}_{4}}_{_{~}}~\] (d)\[SnC{{l}_{2}}\]
View Answer play_arrow
-
question_answer60)
Dry ice is :
(a) solid\[N{{H}_{3}}\] (b)
solid \[S{{O}_{2}}\] (c) solid\[C{{O}_{2}}\] (d)
solid \[{{N}_{2}}\]
View Answer play_arrow
-
question_answer61)
Cement, the important building
material is a mixture of oxides of several elements. Besides calcium, iron and sulphur,
oxides of elements of which of the group(s) is (are) present in the mixture?
(a) group 2 (b)
group 2, 13, 14 (c) group 2 and 13 (d) group 2 and 14
View Answer play_arrow
-
question_answer62)
The reason for small radius of \[Ga\]compared
to Al is..........
(a) poor screening effect of d-
and f-orbitals (b) increase in nuclear charge
(c) presence of higher orbitals (d)
high atomic number
View Answer play_arrow
-
question_answer63)
The linear shape of \[C{{O}_{2}}\]
is due to.......
(a) \[s{{p}^{3}}\]-hybridisation
of carbon
(b) \[sp\]hybridisation of
carbon
(c) \[p\pi -p\pi \]bonding
between carbon and oxygen
(d) \[s{{p}^{2}}\]
-hybridisation of carbon
View Answer play_arrow
-
question_answer64)
The \[M{{e}_{3}}SiCl\] is used
during polymerisation of organo- silicones because:
(a) the chain length of
organosilicone polymers can be controlled by adding \[M{{e}_{3}}SiCl\]
(b) \[M{{e}_{3}}SiCl\]block the
end terminal of silicone polymer
(c) \[M{{e}_{3}}SiCl\]improves
the quality and yield of the polymer
(d) \[M{{e}_{3}}SiCl\]acts as a
catalyst during polymerization
View Answer play_arrow
-
question_answer65)
Which of the following
statements are correct?
(a) Fullerenes have dangling
bonds.
(b) Fullerenes are cage-like
molecules.
(c) Graphite is
thermodynamically the most stable allotrope of carbon.
(d) Graphite is slippery and
hard and therefore used as a dry lubricant in machines.
View Answer play_arrow
-
question_answer66)
Which of the following
statements are correct? Answer on the basis of given figure.
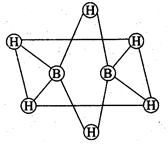 (a) The two bridged hydrogen
atoms and the two boron atoms lie in one plane.
(b) Out of six B-H bonds two
bonds can be described in terms of 3 centre 2-electron bonds.
(c) Out of six B-H bonds four B-H
bonds can be described in terms of 3 centre 2-electron bonds.
(d) The four terminal B-H bonds
are two centre two electron regular bonds.
(a) The two bridged hydrogen
atoms and the two boron atoms lie in one plane.
(b) Out of six B-H bonds two
bonds can be described in terms of 3 centre 2-electron bonds.
(c) Out of six B-H bonds four B-H
bonds can be described in terms of 3 centre 2-electron bonds.
(d) The four terminal B-H bonds
are two centre two electron regular bonds.
View Answer play_arrow
-
question_answer67)
Identify the correct resonance
structures of carbon dioxide from the ones given below :
(a) \[O-C=O\] (b)
\[O=C=O\] (c) \[^{-}O\equiv C-{{O}^{+}}\] (d)
\[^{-}O-C\equiv {{O}^{+}}\]
View Answer play_arrow
-
question_answer68)
Draw the structures of \[BC{{l}_{3}},N{{H}_{3}}\]
and\[AlC{{l}_{3}}\] (dimer).
View Answer play_arrow
-
question_answer69)
Explain the nature of boric acid
as a Lewis acid in water.
View Answer play_arrow
-
question_answer70)
Draw the structure of boric acid
showing hydrogen bonding.
Which species is present in
water? What is hybridisation of boron in this species?
View Answer play_arrow
-
question_answer71)
Explain why the following
compounds act as Lewis acids?
(a) \[BC{{l}_{3}}\] (b)
\[AlC{{l}_{3}}\]
View Answer play_arrow
-
question_answer72)
Give reasons for the following :
(a) \[CC{{l}_{4}}\] is immiscible
in water whereas \[SiC{{l}_{4}}\] is easily hydrolysed.
(b) Carbon has a strong tendency
for catenation compared to silicon.
View Answer play_arrow
-
question_answer73)
Explain the following :
(a) \[C{{O}_{2}}\] is a gas
whereas \[Si{{O}_{2}}\] is a solid.
(b) Silicon forms \[SiF_{6}^{2-}\]
ion whereas corresponding fluoro compound of carbon is not known.
View Answer play_arrow
-
question_answer74)
The +1 oxidation state in group
13 and +2 oxidation state in group 14 becomes more and more stable with
increasing atomic number. Explain.
View Answer play_arrow
-
question_answer75)
Carbon and silicon both belong
to group 14, but inspite of the stoichiometric similarity, the dioxides, (i.e.,
carbon dioxide and silicon dioxide) differ in their structures. Comment.
View Answer play_arrow
-
question_answer76)
If a trivalent atom replaces a
few silicon atoms in three dimensional network of silicon dioxide, what would
be the type of charge on overall structure?
View Answer play_arrow
-
question_answer77)
When \[BC{{l}_{3}}\] is treated
with water, it hydrolyses and forms\[{{[B{{(OH)}_{4}}]}^{-}}\] only whereas \[AlC{{l}_{3}}\]
in acidified aqueous solution forms \[{{[Al{{({{H}_{2}}O)}_{6}}]}^{3+}}\] ion.
Explain what is the hybridisation of boron and aluminium in these species?
View Answer play_arrow
-
question_answer78)
Aluminium dissolves in mineral
acids and aqueous alkalies and thus shows amphoteric character. A piece of
aluminium foil is treated with dilute hydrochloric acid or dilute sodium hydroxide
in a test tube and on bringing a burning matchstick near the mouth of the test
tube, a pop sound indicate the evolution of hydrogen gas. The same activity
when performed with concentrate nitric acid, reaction does not proceed. Explain
the reason.
View Answer play_arrow
-
question_answer79)
Explain the following :
(i) Gallium has higher
ionisation enthalpy than aluminium.
(ii) Boron does not exist as \[{{B}^{3+}}\]
ion.
(iii) Aluminium forms \[{{[Al{{F}_{6}}]}^{3-}}\]
ion but boron does not form\[{{[B{{F}_{6}}]}^{3-}}\] ion.
(iv) \[Pb{{X}_{2}}\]is more
stable than \[Pb{{X}_{4}}\].
(v) \[P{{b}^{4+}}\]acts as an
oxidising agent but \[S{{n}^{2+}}\] acts as a reducing agent.
(vi) Electron gain enthalpy of
chlorine is more negative as compared to fluorine.
(vii) acts as an oxidising
agent.
(viii) Carbon shows catenation
property but lead does not.
(ix) \[B{{F}_{3}}\]does not hydrolyse.
(x) Why does the element silicon
not form a graphite like structure whereas carbon does?
View Answer play_arrow
-
question_answer80)
Identify the compounds 'A', 'X?
and 'Z? in the following reactions.
(i)\[A+2HCl+5{{H}_{2}}O\to
2NaCl+X\]
(ii) \[X\xrightarrow[370K]{\Delta
}HB{{O}_{2}}\xrightarrow[>370K]{\Delta }z\]
View Answer play_arrow
-
question_answer81)
Complete the following chemical
equations :
\[Z+3LiAl{{H}_{4}}\to
X+3LiF+3Al{{F}_{3}}\]
\[X+6{{H}_{2}}O\to
Y+6{{H}_{2}}\]
\[3X+3{{O}_{2}}\to
{{B}_{2}}{{O}_{3}}+3{{H}_{2}}O\]
View Answer play_arrow
-
question_answer82)
In the following questions
more than one correlation is possible between options of column I and column
II. Make as many correlations as you can.
Match the species given in
column I with the properties mentioned in column II.
|
Column I
|
Column II
|
|
(i) \[BF_{4}^{-}\]
|
(a) Oxidation state of central atom is
+4
|
|
(ii) \[AlC{{l}_{3}}\]
|
(b) Strong oxidising agent
|
|
(iii) \[SnO\]
|
(c) Lewis acid
|
|
(iv) \[Pb{{O}_{2}}\]
|
(d) Can be further oxidised
|
|
|
(e) Tetrahedral shape
|
View Answer play_arrow
-
question_answer83)
Match the species given in
column I with properties given in column II.
|
Column I
|
Column II
|
|
(i)
|
Diborane
|
(a)
|
Used as a flux for soldering metals
|
|
(ii)
|
Gallium
|
(b)
|
Crystalline form of silica
|
|
(iii)
|
Borax
|
(c)
|
Banana bonds
|
|
(iv)
|
Alumino silicate
|
(d)
|
Low melting, high boiling, useful for measuring
high temperatures
|
|
(V)
|
Quartz
|
(e)
|
Used as catalyst in petrochemical industries
|
View Answer play_arrow
-
question_answer84)
Match the species given in
column I with the hybridization given in column II.
|
Column I
|
Column II
|
|
(i) Boron is\[{{[B{{(OH)}_{4}}]}^{-}}\]
|
(a)\[s{{p}^{2}}\]
|
|
(ii) Aluminium in \[{{[Al{{({{H}_{2}}O)}_{6}}]}^{^{3+}}}\]
|
(b) \[s{{p}^{3}}\]
|
|
(iii) Boron in \[{{B}_{2}}{{H}_{6}}\]
|
(c)\[s{{p}^{3}}{{d}^{2}}\]
|
|
(iv) Silicon in \[SiO_{4}^{4-}\]
|
|
|
(vi) Germanium in \[{{[GeC{{l}_{6}}]}^{2-}}\]
|
|
View Answer play_arrow
-
question_answer85)
In the following questions a
statement of Assertion (A) followed by a statement of Reason (K) is given.
Choose the correct option out of the choices given below each question.
Assertion (A): If
aluminium atoms replace a few silicon atoms in three dimensional network of
silicon dioxide, die overall structure acquire a negative charge.
Reason (R): Aluminium is
trivalent while silicon is tetravalent.
(a) Both A and R
are correct and R is the correct explanation of A.
(b)Both A and R
are correct but R is not the correct explanation of A.
(c) Both A and R
are not correct.
(d) A is not correct but R
is correct.
View Answer play_arrow
-
question_answer86)
Assertion (A): Silicones
are water repelling in nature.
Reason (R): Silicones are
organosilicon polymers, which have \[(-{{R}_{2}}SiO-)\] as repeating unit.
(a) Both A and R
are correct and R is the correct explanation of A.
(b) Both A and R
are correct but R is not the correct explanation of A.
(c) Both A and R
are not correct.
(d) A is not correct but R
is correct.
View Answer play_arrow
-
question_answer87)
Describe the general trends in
the following properties of the groups 13 and 14.
(i) Atomic size
(ii) Ionisation enthalpy
(iii) Metallic character
(iv) Oxidation states
(v) Nature of halides
View Answer play_arrow
-
question_answer88)
Account for the following
observations :
(i) \[AlC{{l}_{3}}\]is a Lewis
acid.
(ii) Though fluorine: is mere electronegative
than chlorine yet \[B{{F}_{3}}\]is a weaker Lewis acid than\[BC{{l}_{3}}\].
(iii) \[Pb{{O}_{2}}\]is a stronger
oxidising agent than \[Sn{{O}_{2}}\].
(iv) The +1 oxidation state of
thallium is more stable than its +3 state.
View Answer play_arrow
-
question_answer89)
When aqueous solution of borax
is acidified with hydrochloric acid, a white crystalline solid which is soapy to
touch. Is this solid acidic or basic in nature? Explain.
View Answer play_arrow
-
question_answer90)
The pairs of compounds are given
below. Identify the compound in each of the pairs which has group 13 element is
more stable oxidation state. The reason for your choice.
State the nature of bonding also,
(i) \[TlCl,TlCl\]
(ii) \[AlC{{l}_{3}},AlCl\]
(iii) \[InC{{l}_{3}},InCl\]
View Answer play_arrow
-
question_answer91)
\[BC{{l}_{3}}\]exists as monomer whereas
\[AlC{{l}_{3}}\] is dimerised through halogen bridging. Give reason. Explain
the structure of the dimer of \[AlC{{l}_{3}}\] also.
View Answer play_arrow
-
question_answer92)
Boron fluoride exists as \[B{{F}_{3}}\]
but boron hydride does not exist as\[B{{H}_{3}}\]. Give reason. In which form
does it exist?
Explain its structure.
View Answer play_arrow
-
question_answer93)
(i) What are silicones? State
the uses of silicones.
(ii) What are boranes? Give
chemical equation for the preparation of diborane.
View Answer play_arrow
-
question_answer94)
A compound (A) of boron reacts
with \[NM{{e}_{3}}\] to give an adduct (B) which on hydrolysis gives a compound
(C) and hydrogen gas. Compound (C) is an acid. Identify the compounds A, B and
C. Give the reactions involved.
View Answer play_arrow
-
question_answer95)
A non-metallic element of group
13 used in making bullet-proof vests is extremely hard solid of black colour.
It can exist in many allotropic forms and has unusually high melting point. Its
trifluoride acts as Lewis acid towards ammonia. The element exhibits maximum
covalency of four. Identify the element and write the reasons of its trifluoride
with ammonia. Explain why does the trifluoride act as a Lewis acid?
View Answer play_arrow
-
question_answer96)
A tetravalent element forms
monoxide and dioxide with oxygen. When air is passed over heated element
(1273K), producer gas is obtained. Monoxide of the element is a powerful
reducing agent and reduces ferric oxide to iron.
Identify the element and write formulae
of its monoxide and dioxide. Write chemical equations for the formation of producer
gas and reduction of ferric oxide with the monoxide.
View Answer play_arrow
