Answer:
Hydrogen from dilute
acids can only be replaced if electrons are supplied to H+
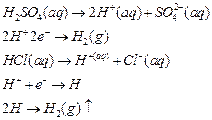 Metals have a tendency to lose electrons and hence they
supply electrons, i.e., they are electron donors. That is why metals displace
hydrogen from dilute acids. On the other hand, non-metal is an electron
acceptor. It cannot supply electrons to H^ and hence, it does not displace
hydrogen from dilute acids.
Metals have a tendency to lose electrons and hence they
supply electrons, i.e., they are electron donors. That is why metals displace
hydrogen from dilute acids. On the other hand, non-metal is an electron
acceptor. It cannot supply electrons to H^ and hence, it does not displace
hydrogen from dilute acids.
You need to login to perform this action.
You will be redirected in
3 sec
