Answer:
Aluminium oxide (Al2O3)
shows basic as well as acidic behaviour because it reacts with both acids and
bases. Thus, it is considered an amphoteric oxide. The two types of reactions
given by Al2O3 are as follows:
(i)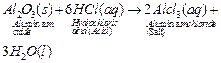 (ii)
(ii) 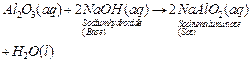 In
this reaction, Al2O3 behaves as a basic oxide because it
reacts with an acid to form salt and water.
In
this reaction, Al2O3 behaves as a basic oxide because it
reacts with an acid to form salt and water.
You need to login to perform this action.
You will be redirected in
3 sec
