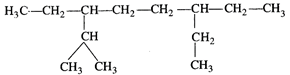
Answer:
The correct order of reactivity in electrophilic substitution is:
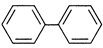 The electrophile attack will be the easiest in case the ring is maximum activated out of the groups attached in different molecules.
• \['C{{H}_{3}}'\] is highly activating due to hyper conjugation.
• \['Cl'\] is activating due to resonance (R) effect but at the same time it is deactivating due to\[-I\] effect.
• \['N{{O}_{2}}'\]is deactivating in nature.
In the light of the above observations, the order of reactivity is justified.
The electrophile attack will be the easiest in case the ring is maximum activated out of the groups attached in different molecules.
• \['C{{H}_{3}}'\] is highly activating due to hyper conjugation.
• \['Cl'\] is activating due to resonance (R) effect but at the same time it is deactivating due to\[-I\] effect.
• \['N{{O}_{2}}'\]is deactivating in nature.
In the light of the above observations, the order of reactivity is justified.
You need to login to perform this action.
You will be redirected in
3 sec
