Answer:
In ethyl carbocation, there is only hyper-conjugation of the three \[\alpha \]-hydrogen atoms and as a result, the following contributing structures are feasible.
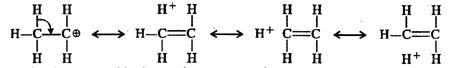 But benzyl carbocation is more stable due to the presence of resonance and the following resonating structures are possible:
But benzyl carbocation is more stable due to the presence of resonance and the following resonating structures are possible: 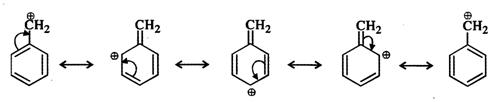
You need to login to perform this action.
You will be redirected in
3 sec
