Answer:
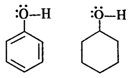
In phenol, the oxygen atom of O?H
group becomes electron deficient due to the conjugation of electron pairs on it
with the pi electron pairs of ring. Therefore, \[{{H}^{+}}\]can be released. However,
in cyclohexanol, the electron pairs on the oxygen atom are not involved in any conjugation
with the ring since it has no pi electron pairs. Therefore, the release of \[{{H}^{+}}\]ion
is quite difficult.
You need to login to perform this action.
You will be redirected in
3 sec
