Answer:
Due to the presence of lone pairs of electrons on oxygen, it has +R-effect while C = O group has -R-effect.
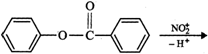
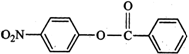
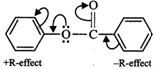 As a result, electron-density is more in the ring attached to O-atom than in the ring attached to C = O group. In other words, \[N{{O}_{2}}^{+}\] will attack the ring attached to oxygen. Further, due to +R-effect of the O-atom, electron-density increases at o- and p-positions. But, due to steric hindrance at o-position, electrophilic substitution mainly occurs at the p-position. Thus,
As a result, electron-density is more in the ring attached to O-atom than in the ring attached to C = O group. In other words, \[N{{O}_{2}}^{+}\] will attack the ring attached to oxygen. Further, due to +R-effect of the O-atom, electron-density increases at o- and p-positions. But, due to steric hindrance at o-position, electrophilic substitution mainly occurs at the p-position. Thus,
You need to login to perform this action.
You will be redirected in
3 sec
