Answer:
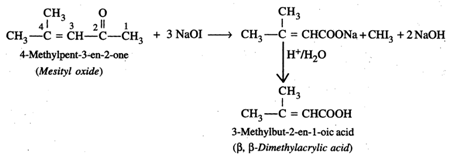
Alkaline \[KMn{{O}_{4}},\] acidified \[{{K}_{2}}C{{r}_{2}}{{O}_{7}}\] or \[HN{{O}_{3}}\] cannot be used since all of these will cleave the molecule at the site of the double bond giving a mixture of ketones/acids. The most suitable reagent for this oxidation is \[NaOI\,({{I}_{2}}/NaOH)\] since methyl ketones on treatment with \[NaOI\] undergo iodoform reaction to give iodoform along with the Na salt of a carboxylic acid having one carbon atom less than the starting methyl ketone.
You need to login to perform this action.
You will be redirected in
3 sec
