Answer:
(a) 2-Bromooctane is a chiral
molecule and exists as two optical isomers (enantiomers), i.e., (+) and (-).
2-bromooctane. When (-)-2-bromooctanes is treated with \[NaOH,\] it undergoes \[{{S}_{N}}2\]
reaction in which \[O{{H}^{-}}\]ion attacks from the backside. Inversion of
configuration occurs and (+)-octan-2-ol is formed (refer to Fig. 10.10, page
10/36).
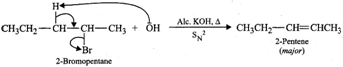
(b) 2-Bromopentane on heating with alc. KOH undergoes
dehydrohalogenation to give a mixture of two alkenes, i.e., 2-pentene as the
major product (Sayzeff product) and 1-pentene as the minor product.
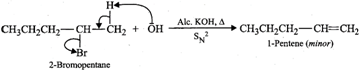
You need to login to perform this action.
You will be redirected in
3 sec
