Answer:
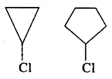
In \[{{S}_{{{N}^{1}}}}\] reaction, a carbocation intermediate is formed in the slow or rate determining step by the cleavage of \[C-Cl\] bond. Now, the cyclopropyl carbocation intermediate is less stable than cyclopentyl carbocation due to greater strain. Therefore, cyclopropyl chloride is less reactive than cyclopentyl chloride towards \[{{S}_{{{N}^{1}}}}\] reaction.
You need to login to perform this action.
You will be redirected in
3 sec
