Answer:
Two products are formed in this reaction
\[\underset{\text{2-Bromo-3-methylbutane}}{\mathop{C{{H}_{3}}-\underset{\begin{smallmatrix} | \\ Br \end{smallmatrix}}{\mathop{C}}\,H-\underset{\begin{smallmatrix} | \\ C{{H}_{3}} \end{smallmatrix}}{\mathop{C}}\,H-C{{H}_{3}}}}\,\xrightarrow{{{C}_{2}}{{H}_{5}}^{-}O\overset{+}{\mathop{N}}\,a}\] 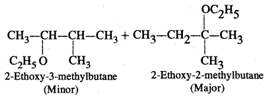 Mechanism:
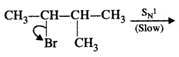
Mechanism:
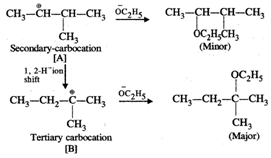 Please note that the tetiary carbocation formed as a result of 1, 2-hydride ion shift is more stable than the secondary carbocation.
Please note that the tetiary carbocation formed as a result of 1, 2-hydride ion shift is more stable than the secondary carbocation.
You need to login to perform this action.
You will be redirected in
3 sec
