Answer:
Fluorine (F) being a highly electronegative
element has a strong -\[I\]effect. Therefore, when \[{{H}^{+}}\] from acid (electrophile)
attacks the substrate, a primary carbocation is preferably formed. This reacts
with the anion to give the desired product.
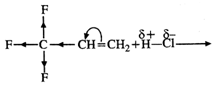
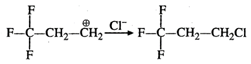
You need to login to perform this action.
You will be redirected in
3 sec
