Answer:
The reactivity in \[{{S}_{N}}1\] reactions depends upon the stability of the intermediate carbocation which an alkyl halide gives on ionization.
(i)
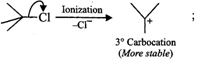
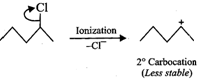 Since \[3{}^\circ \] carbocations are more stable than\[2{}^\circ \] carbocations, therefore,
Since \[3{}^\circ \] carbocations are more stable than\[2{}^\circ \] carbocations, therefore, ![]() will react faster than
will react faster than ![]() (ii)
(ii)
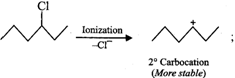
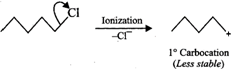 Since \[2{}^\circ \] carbocations are more stable than \[1{}^\circ \]carbocations, therefore,
Since \[2{}^\circ \] carbocations are more stable than \[1{}^\circ \]carbocations, therefore, ![]() reacts faster than
reacts faster than ![]() .
.
You need to login to perform this action.
You will be redirected in
3 sec
