Answer:
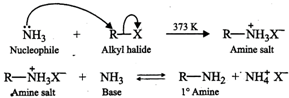
During ammonolysis of alkyl halides,
the acid liberated during the reaction combines with the amine formed to form
the amine salt. To liberate free amine from the amine salt, a base is needed.
The base used is excess of alcoholic \[N{{H}_{3}}\] which serves both as a
nucleophile as well as a base. For example
You need to login to perform this action.
You will be redirected in
3 sec
