Answer:
Butadiene is a conjugated diene and its free radical polymerisation can occur in two ways:
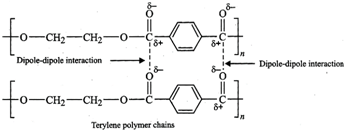
(i) When the polymerisation takes place at \[{{C}_{1}}\] and \[{{C}_{4}}\] of butadiene, an unbranched polymer results. It can exist either as trans-polybutadiene or as cis isomer or as the mixture of both.
 (ii) 1, 3 butadiene can also undergo polymerisation at \[{{C}_{1}}\] and \[{{C}_{2}}\] to give polyvinyl polyethene as the product.
(ii) 1, 3 butadiene can also undergo polymerisation at \[{{C}_{1}}\] and \[{{C}_{2}}\] to give polyvinyl polyethene as the product.
You need to login to perform this action.
You will be redirected in
3 sec
