Answer:
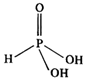
In the structural formula,
phosphorus acid \[({{H}_{3}}P{{O}_{3}})\] has only two OH groups and therefore,
there are only two ionisable hydrogen atoms. Since the third hydrogen atom is
attached directly to the phosphorus atom, it is not ionised. Thus, the acid is
diprotic in nature.
You need to login to perform this action.
You will be redirected in
3 sec
