-
question_answer1)
| Direction (1 - 5): Phenols and alcohols have both the same functional group (-OH) but phenols are stronger acids than alcohols. This is mainly due to the reason that the phenoxide ion left after the release of \[{{H}^{+}}\] ion in phenol is resonance stabilised while the alkoxide ion in alcohols does not show similar characteristics. The electron withdrawing groups tend to increase the acidic strengths of phenols while electron releasing groups tend to decrease it. The effect of both types of groups is more pronounced when present at the para position than when these are present at the ortho position in the ring. However, their effect at the meta position is relatively very small. |
The increasing order of reactivity of \[1{}^\circ \], \[2{}^\circ \] and \[3{}^\circ \] alcohols towards sodium metal is
A)
\[1{}^\circ \text{ }<\text{ }2{}^\circ \text{ }<\text{ }3{}^\circ \] done
clear
B)
\[1{}^\circ \text{ }<\text{ }3{}^\circ \text{ }<\text{ }2{}^\circ \] done
clear
C)
\[3{}^\circ \text{ }<\text{ }2{}^\circ \text{ }<\text{ }1{}^\circ \] done
clear
D)
\[2{}^\circ \text{ }<\text{ }1{}^\circ \text{ }<\text{ }3{}^\circ \]. done
clear
View Solution play_arrow
-
question_answer2)
Which of the following is the strongest acid?
A)
\[FC{{H}_{2}}C{{H}_{2}}C{{H}_{2}}C{{H}_{2}}OH\] done
clear
B)
\[FC{{H}_{2}}C{{H}_{2}}C{{H}_{2}}OH\] done
clear
C)
\[C{{H}_{3}}C{{H}_{2}}CHFC{{H}_{2}}OH\] done
clear
D)
\[C{{H}_{3}}C{{H}_{2}}C{{F}_{2}}C{{H}_{2}}OH\] done
clear
View Solution play_arrow
-
question_answer3)
The acidic strength increases in the order
A)
p-Nitrophenol < m-Nitrophenol < o-Nitrophenol done
clear
B)
p-Nitrophenol < o-Nitrophenol < m- Nitrophenol done
clear
C)
m-Nitrophenol < o-Nitrophenol < p-Nitrophenol done
clear
D)
o-Nitrophenol < m-Nitrophenol < p-Nitrophenol. done
clear
View Solution play_arrow
-
question_answer4)
The strongest acid among the following is
A)
o-methoxyphenol done
clear
B)
p-methoxyphenol done
clear
C)
m-methoxyphenol done
clear
D)
phenol. done
clear
View Solution play_arrow
-
question_answer5)
Which reaction shows the acidic nature of alocohols?
A)
Reaction with Na done
clear
B)
Reaction with conc.\[{{H}_{2}}S{{O}_{4}}\] done
clear
C)
Reaction with \[PC{{l}_{5}}\] done
clear
D)
Reaction with \[SOC{{l}_{2}}\] done
clear
View Solution play_arrow
-
question_answer6)
| Direction (6 - 10): The search for mild, versatile, selective reagents for the operationally simple oxidation of alcohols to carbonyl compounds has long been the objectives of many research laboratories. The development of newer methods and methodologies is gaining much attention currently due to the significance of this reaction. The first advantage of pyridinium chlorochromate is that it is prepared easily and safely : the addition of chromium trioxide to 6 N hydrochloric acid furnishes the unstable chlorochromic acid : subsequent addition of pyridine at \[0{}^\circ C\] immediately gives pyridinium chlorochromate as a yellow-orange solid which is not appreciably hygroscopic. |
 |
| The second advantage is that it shows a high capability to convert primary alcohols exclusively to aldehydes with great efficiency. |
| The utilization of the hydrogen peroxide as an oxidant for organic substrates has gained much attention in recent year, because of its environmental implications, water being the only chemical by-product; on the other hand, the water is actually used as process solvent, so the reactions are of general interest due to their potential in combinational chemistry, simple processes, easy workup, low cost and reduction in noxious waste materials. |
| (Reference : G. Piancateli, A Scettri, M. D'auria. Pyridinium Chlorochromate : A versatile oxidant in organic synthesis. Synthesis : International Journal of Methods in Synthetic Organic chemistry. 245-246. 1982 No. 4 : April.) |
| (Reference : Anil C, Ranveer, Suresh V, Ranade, Cyrus K. Mistry. Selective oxidation of alcohols to aldehydes by using hydrogen peroxide as an oxidant : A review. International Journal of Advance Research in Science and Engineering. Vol. No. 4, Special Issue (01), September 2015.) |
Which of the following can be used to oxidize \[1{}^\circ \] alcohols to aldehydes?
A)
\[Cr{{O}_{3}}\] done
clear
B)
PCC done
clear
C)
\[{{H}_{2}}{{O}_{2}}\] done
clear
D)
All the above done
clear
View Solution play_arrow
-
question_answer7)
Breathalyzer tubes used to detect alcohol intoxicated truck drivers contain the dichromate salt. This use is based on
A)
reduction of dichromate to give a characteristic colour done
clear
B)
reduction of dichromate to give a characteristic smell done
clear
C)
the conversion of alcohol to \[C{{O}_{2}}\] and aldehyde done
clear
D)
oxidation of dichromate to \[C{{r}^{3+}}\] salt. done
clear
View Solution play_arrow
-
question_answer8)
| The products of the oxidation |
| \[C{{H}_{3}}CH=CHCH\left( OH \right)C{{H}_{3}}\xrightarrow[acetone]{{{H}_{2}}Cr{{O}_{4}}\,in\,aq.}\] is/are |
A)
\[C{{H}_{3}}-CH=CH-CHO\] done
clear
B)
\[C{{H}_{3}}-CH=CH-COC{{H}_{3}}\] done
clear
C)
D)
View Solution play_arrow
-
question_answer9)
PCC acts as a :
A)
strong oxidizing agent done
clear
B)
mild oxidizing agent done
clear
C)
strong reducing agent done
clear
D)
mild reducing agent. done
clear
View Solution play_arrow
-
question_answer10)
One of following compounds is not affected when treated with Pyridinium Chlorochromate?
A)
n - Butyl alcohol done
clear
B)
Isobutyl alcohol done
clear
C)
sec - Butyl alcohol done
clear
D)
tert - Butyl alcohol done
clear
View Solution play_arrow
-
question_answer11)
| Direction (11 - 15): When we are asked to make ether starting from alkyl halides by substitution reactions of alkoxide anion nucleophiles with alkyl halide electrophiles (The Williamson ether synthesis) The two ways to prepare the target ether can be there : |
| Route A involving a secondary \[\left( 2{}^\circ \right)\] or tertiary \[\left( 3{}^\circ \right)\] alkyl halide is not preferred because we expect the major product to result from elimination by \[{{E}_{2}}\] mechanism. |
| Route B on the other hand, employs a primary alkyl halide that can not give elimination (It has no hydrogen on the \[\beta \]-carbon) and that is excellent. |
Which of the following reagents when heated will give a good yield of an ether?
A)
Isopropyi bromide and sodium isopropoxide done
clear
B)
Isopropyi bromide and sodium ethoxide done
clear
C)
Bromobenzene and sodium phenoxide done
clear
D)
Sodium tert butoxide and ethyl bromide. done
clear
View Solution play_arrow
-
question_answer12)
Which of the following ethers can be prepared by Williamson's synthesis?
A)
Benzyl methyl ether done
clear
B)
Methyl vinyl ether done
clear
C)
Divinyl ether done
clear
D)
Diphenyl ether done
clear
View Solution play_arrow
-
question_answer13)
Methyl tertiary butyl ether (MTBE) is an important gasoline additive for improving octane number. Select the best method of its synthesis out of the following
A)
\[{{\left( C{{H}_{3}} \right)}_{3}}CONa+C{{H}_{3}}I\to \] done
clear
B)
\[{{\left( C{{H}_{3}} \right)}_{3}}C=C{{H}_{2}}+C{{H}_{3}}OH\xrightarrow{{{H}_{2}}S{{O}_{4}}}\] done
clear
C)
\[{{\left( C{{H}_{3}} \right)}_{3}}CBr+C{{H}_{3}}ONa\to \] done
clear
D)
all give 100% yield done
clear
View Solution play_arrow
-
question_answer14)
Benzyl ethyl ether reacts with HI to form
A)
p-iodotoluene and ethyl alcohol done
clear
B)
Benzyl alcohol and ethyl iodide done
clear
C)
Benzyl iodide and ethyl alcohol done
clear
D)
Iodobenzene and ethyl alcohol. done
clear
View Solution play_arrow
-
question_answer15)
Alkyi phenyl ether can be prepared by heating
A)
\[{{C}_{6}}{{H}_{5}}Br+C{{H}_{2}}=CH-C{{H}_{2}}ONa\] done
clear
B)
\[C{{H}_{2}}=CHC{{H}_{2}}Br+{{C}_{6}}{{H}_{5}}ONa\] done
clear
C)
\[{{C}_{6}}{{H}_{5}}CH=CHBr+C{{H}_{3}}ONa\] done
clear
D)
\[C{{H}_{2}}=CHBr+{{C}_{6}}{{H}_{5}}C{{H}_{2}}ONa\] done
clear
View Solution play_arrow
-
question_answer16)
| Direction (16 - 20): The cumene-phenol process is the most important commercial method for the preparation of phenol and is carried out by the oxidation of cumene to its hydroperoxide, which is then decomposed by acid. The other synthetic methods available are from chlorobenzene and sodium benzene sulphonate. Phenol is a colourless crystalline solid which turns pink on exposure to air and light. Many of the reactions of phenol are those which are example of electrophilic aromatic substitution and since the hydroxyl group is o-Ip- orienting, these are always the products formed. |
| (I. L Finar, Organic Chemistry Vol I: The fundamental principles. Sixth Edition) |
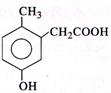
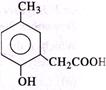
p-cresol reacts with chloroform in alkaline medium to give the compound 'A' which adds hydrogen cyanide to form the compound 'B'. The latter on acidic hydrolysis gives chiral carboxylic acid. The structure of the carboxylic acid is
A)
B)
C)
D)
View Solution play_arrow
-
question_answer17)
Chlorobenzene can be converted to phenol by heating it with caustic soda under pressure. This is known as
A)
Dow's process done
clear
B)
Kolbe's process done
clear
C)
Fittig reaction done
clear
D)
Reimer Tiemann reaction. done
clear
View Solution play_arrow
-
question_answer18)
When phenol is treated with chloromethane in presence of \[AIC{{l}_{3}}\] we get
A)
o-cresol done
clear
B)
w-cresol done
clear
C)
p-cresol done
clear
D)
mixture of o and p- cresol. done
clear
View Solution play_arrow
-
question_answer19)
In cumene process, phenol is manufactured starting from :
A)
Chlorobenzene and alkali done
clear
B)
Benzene and propylene done
clear
C)
Chlorobenzene and ethanol done
clear
D)
Benzene and acetyl chloride. done
clear
View Solution play_arrow
-
question_answer20)
Phenol is colourless solid but turns pink on exposure to air and light. This is due to the formation of
A)
p-quinol done
clear
B)
p-Benzoquinone done
clear
C)
p-Nitrophenol done
clear
D)
benzene done
clear
View Solution play_arrow
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
