-
question_answer1)
| Directions : (1 - 5) |
| Case Based MCQs |
| Case I : Read the passage given below and answer the following questions. |
| The sequence of bases along the DNA and RNA chain establishes its primary structure which controls the specific properties of the nucleic acid. An RNA molecule is usually a single chain of ribose-containing nucleotide. On the basis of X-ray analysis of DNA, J.D., Watson and F.H.C. crick (shared noble prize in 1962) proposed a three dimensional secondary structure for DNA. DNA molecule is a long and highly complex, spirally twisted, double helix, ladder like structure. The two polynucleotide chains or strands are linked up by hydrogen bonding between the nitrogeneous base molecules of their nucleotide monomers. Adenine (purine) always links with thymine (pyrimidine) with the help of two hydrogen bonds and guanine (purine) with cytosine (pyrimidine) with the help of three hydrogen bonds. Hence, the two strands extend in opposite directions, i.e., are antiparallel and complimentary. |
| In the following questions (Q. No. 1-5), a statement of assertion followed by a statement of reason is given. Choose the correct answer out of the following choices on the basis of the above passage. |
| Assertion : DNA molecules and RNA molecules are found in the nucleus of a cell. |
| Reason : There are two types of nitrogenous bases, purines and pyrimidines. Adenine [A] and guanine [G] are substituted purines; cytosine [C], thymine [T] and uracil [U] are substituted pyrimidines. |
A)
Assertion and reason both are correct statements and reason is correct explanation for assertion. done
clear
B)
Assertion and reason both are correct statements but reason is not correct explanation for assertion. done
clear
C)
Assertion is correct statement but reason is wrong statement. done
clear
D)
Assertion is wrong statement but reason is correct statement. done
clear
View Solution play_arrow
-
question_answer2)
| Assertion : In both DNA and RNA, heterocyclic base and phosphate ester linkages are at C-1' and C-5' respectively of the sugar molecule. |
| Reason : Nucleotides and nucleosides mainly differ from each other in presence of phosphate units. |
A)
Assertion and reason both are correct statements and reason is correct explanation for assertion. done
clear
B)
Assertion and reason both are correct statements but reason is not correct explanation for assertion. done
clear
C)
Assertion is correct statement but reason is wrong statement. done
clear
D)
Assertion is wrong statement but reason is correct statement. done
clear
View Solution play_arrow
-
question_answer3)
| Assertion : The backbone of RNA molecule is a linear chain consisting of an alternating units of a heterocylic base, P-ribose and a phosphate. |
| Reason : The segment of DNA which acts as the instruction manual for the synthesis of protein is ribose. |
A)
Assertion and reason both are correct statements and reason is correct explanation for assertion. done
clear
B)
Assertion and reason both are correct statements but reason is not correct explanation for assertion. done
clear
C)
Assertion is correct statement but reason is wrong statement. done
clear
D)
Assertion is wrong statement but reason is correct statement. done
clear
View Solution play_arrow
-
question_answer4)
| Assertion : The double helical structure of DNA was proposed by Emil Fischer. |
| Reason : A nucleoside is an N-glycoside of heterocyclic base. |
A)
Assertion and reason both are correct statements and reason is correct explanation for assertion. done
clear
B)
Assertion and reason both are correct statements but reason is not correct explanation for assertion. done
clear
C)
Assertion is correct statement but reason is wrong statement. done
clear
D)
Assertion is wrong statement but reason is correct statement. done
clear
View Solution play_arrow
-
question_answer5)
| Assertion : In DNA, the complementary bases are, adenine and guanine; thymine and cytosine. |
| Reason : The phenomenon of mutation is chemical change in DNA molecule. |
A)
Assertion and reason both are correct statements and reason is correct explanation for assertion. done
clear
B)
Assertion and reason both are correct statements but reason is not correct explanation for assertion. done
clear
C)
Assertion is correct statement but reason is wrong statement. done
clear
D)
Assertion is wrong statement but reason is correct statement. done
clear
View Solution play_arrow
-
question_answer6)
| Directions : (6 - 10) |
| Case II : Read the passage given below and answer the following questions from 6 to 10. |
| Carbohydrates can exist in either of two conformations, as determined by the orientation of the hydroxyl group about the asymmetric carbon farthest from the carbonyl. |
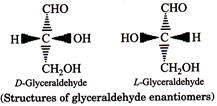 |
| By convention, a monosaccharide is said to have-D-configuration if the hydroxyl group attached to the asymmetric carbon atom adjacent to the \[-C{{H}_{2}}OH\]group is on the right hand side irrespective of the positions of the other hydroxyl groups. On the other hand, the molecule is assigned L-configuration if the \[OH\]group attached to the carbon adjacent to the \[-C{{H}_{2}}OH\]group is on the left hand side. |
D-Glyceraldehyde and L-Glyceraldehyde are
A)
epimers done
clear
B)
enantiomers done
clear
C)
anomers done
clear
D)
conformational diasteriomers. done
clear
View Solution play_arrow
-
question_answer7)
Which of the following monosaccharides, is the majority found in the human body?
A)
D-type done
clear
B)
L-type done
clear
C)
Both of these done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer8)
The two functional groups present in a typical carbohydrate are
A)
-OH and\[-COOH\] done
clear
B)
-CHO and\[-COOH\] done
clear
C)
\[>C=O\]and \[-OH\] done
clear
D)
\[-OH\]and \[CHO\] done
clear
View Solution play_arrow
-
question_answer9)
Monosaccharides contain
A)
always six carbon atoms done
clear
B)
always five carbon atoms done
clear
C)
always four carbon atoms done
clear
D)
may contain 3 to 7 carbon atoms. done
clear
View Solution play_arrow
-
question_answer10)
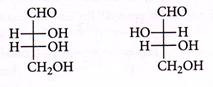
| The correct corresponding order of names of given four aldoses with configuration. |
 |
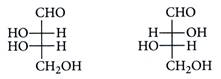 |
| respectively, is |
A)
L-erythrose, L-threose, L-erythrose, D-threose done
clear
B)
D-threose, D-erythrose, L-threose, L-erythrose done
clear
C)
L-erythrose, L-threose, D-erythrose, D-threose done
clear
D)
D-erythrose, D-threose, L-erythrose, L-threose. done
clear
View Solution play_arrow
-
question_answer11)
| Directions : (11 - 15) |
| Case III : Read the passage given below an answer the following questions from 11 to 15. |
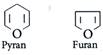
| Pentose and hexose undergo intramolecular hemiacetal or hemiketal formation due to combination of the \[-OH\] group with the carbonyl group. The actual structure is either of five or six membered ring containing an oxygen atom. |
| In the free state all pentoses and hexoses exist in pyranose form (resembling pyran). However, in the combined state some of them exist as five membered cyclic structures, called furanose (resembling furan). |
 |
| The cyclic structure of glucose is represented by Haworth structure : |
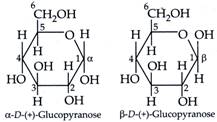 |
| \[\alpha \] and \[\beta \]-D-glucose have different configuration at anomeric (C-1) carbon atom, hence are called anomers and the C-1 carbon atom is called anomeric carbon (glycosidic carbon). |
| The six membered cyclic structure of glucose is called pyranose structure. |
\[\alpha \]-D-(+)-glucose and \[\beta \]-D-(+)-glucose are
A)
enantiomers done
clear
B)
conformers done
clear
C)
epimers done
clear
D)
anomers. done
clear
View Solution play_arrow
-
question_answer12)
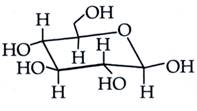
| The following carbohydrate is |
 |
A)
a ketohexose done
clear
B)
an aldohexose done
clear
C)
an \[\alpha \]-furanose done
clear
D)
an \[\alpha \]-pyranose. done
clear
View Solution play_arrow
-
question_answer13)
| In the following structure, |
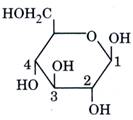 |
| anomeric carbon is |
A)
C-1 done
clear
B)
C-2 done
clear
C)
C-3 done
clear
D)
C-4 done
clear
View Solution play_arrow
-
question_answer14)
The term anomers of glucose refers to
A)
isomers of glucose that differ in configurations at carbons one and four (C-1 and C-4) done
clear
B)
a mixture of [D]-glucose and [L]-glucose done
clear
C)
enantiomers of glucose done
clear
D)
isomers of glucose that differ in configuration at carbon one (C-1). done
clear
View Solution play_arrow
-
question_answer15)
What percentage of \[\beta \]-D-(+) glucopyranose is found at equilibrium in the aqueous solution?
A)
50% done
clear
B)
\[\approx 100%\] done
clear
C)
36% done
clear
D)
64% done
clear
View Solution play_arrow
-
question_answer16)
| Directions : (16 - 20) |
| Case IV : Read the passage given below and answer the following questions. |
| Proteins are high molecular mass complex biomolecules of amino acids. The important proteins required for our body are enzymes, hormones, antibodies, transport proteins, structural proteins, contractile proteins etc. Except for glycine, all \[\alpha \]-amino acids have chiral carbon atom and most of them have L-configuration. The amino acids exists as dipolar ion called zwitter ion, in which a proton goes from the carboxyl group to the amino group. A large number of \[\alpha \]-amino acids are joined by peptide bonds forming polypeptides. The peptides having very large molecular mass (more than 10,000) are called proteins. The structure of proteins is described as primary structure giving sequence of linking of amino acids; secondary structure giving manner in which polypeptide chains are arranged and folded; tertiary structure giving folding, coiling or bonding polypeptide chains producing three dimensional structures and quaternary structure giving arrangement of sub- units in an aggregate protein molecule. |
| In the following questions (Q. No. 16-20), a statement of assertion followed by a statement of reason is given. Choose the correct answer out of the following choices on the basis of the above passage. |
| Assertion : Except glycine, all naturally occurring a-amino acids are optically active. |
| Reason : All naturally occurring \[\alpha \]-amino acids, except glycine, has at least one asymmetric carbon. |
A)
Assertion and reason both are correct statements and reason is correct explanation for assertion. done
clear
B)
Assertion and reason both are correct statements but reason is not correct explanation for assertion. done
clear
C)
Assertion is correct statement but reason is wrong statement. done
clear
D)
Assertion is wrong statement but reason is correct statement. done
clear
View Solution play_arrow
-
question_answer17)
| Assertion : All amino acids are optically active. |
| Reason : Amino acids contain asymmetric carbon atoms. |
A)
Assertion and reason both are correct statements and reason is correct explanation for assertion. done
clear
B)
Assertion and reason both are correct statements but reason is not correct explanation for assertion. done
clear
C)
Assertion is correct statement but reason is wrong statement. done
clear
D)
Assertion is wrong statement but reason is correct statement. done
clear
View Solution play_arrow
-
question_answer18)
| Assertion : In \[\alpha \]-helix structure, intramolecular H-bonding takes place whereas in \[\beta \]-pleated structure, intermolecular H-bonding takes place. |
| Reason : An egg contains a soluble globular protein called albumin which is present in the white part. |
A)
Assertion and reason both are correct statements and reason is correct explanation for assertion. done
clear
B)
Assertion and reason both are correct statements but reason is not correct explanation for assertion. done
clear
C)
Assertion is correct statement but reason is wrong statement. done
clear
D)
Assertion is wrong statement but reason is correct statement. done
clear
View Solution play_arrow
-
question_answer19)
| Assertion : Secondary structure of protein refers to regular folding patterns of continues portions of the polypeptide chain. |
| Reason : Out of 20 amino acids, only 12 amino acids can be synthesised by human body. |
A)
Assertion and reason both are correct statements and reason is correct explanation for assertion. done
clear
B)
Assertion and reason both are correct statements but reason is not correct explanation for assertion. done
clear
C)
Assertion is correct statement but reason is wrong statement. done
clear
D)
Assertion is wrong statement but reason is correct statement. done
clear
View Solution play_arrow
-
question_answer20)
| Assertion : The helical structure of protein is stabilised by intramolecular hydrogen bond between -NH and carbonyl oxygen. |
| Reason : Sanger's reagent is used for the identification of N-terminal amino acid of peptide chain. |
A)
Assertion and reason both are correct statements and reason is correct explanation for assertion. done
clear
B)
Assertion and reason both are correct statements but reason is not correct explanation for assertion. done
clear
C)
Assertion is correct statement but reason is wrong statement. done
clear
D)
Assertion is wrong statement but reason is correct statement. done
clear
View Solution play_arrow
-
question_answer21)
| Directions : (21 - 25) |
| Case V : Read the passage given below and answer the following questions from 21 to 25. |
| Carbohydrates are polyhydroxy aldehydes and ketones and those compounds which on hydrolysis give such compounds are also carbohydrates. The carbohydrates which are not hydrolysed are called monosaccharides. Monosaccharides with aldehydic group are called aldose and those which free ketonic groups are called ketose. Carbohydrates are optically active. Number of optical isomers \[=\text{ }{{2}^{n}}\] |
| Where n = number of asymmetric carbons. |
| Carbohydrates are mainly synthesised by plants during photosynthesis. The monosaccharides give the characteristic reactions of alcohols and carbonyl group (aldehydes and ketones). It has been found that these monosaccharides exist in the form of cyclic structures. In cyclization, the OH groups (generally \[{{C}_{5}}\] or \[{{C}_{4}}\] in aldohexoses and \[{{C}_{5}}\] or \[{{C}_{6}}\] in ketohexoses) combine with the aldehyde or keto group. As a result, cycli structures of five or six membered rings containing one oxygen atom are formed, e.g., glucose forms a ring structure. Glucose contains one aldehyde group, one \[1{}^\circ \] alcoholic group and four \[2{}^\circ \] alcoholic groups in its open chain structure. |
First member of ketos sugar is
A)
ketotriose done
clear
B)
ketotetrose done
clear
C)
ketopentose done
clear
D)
ketohexose. done
clear
View Solution play_arrow
-
question_answer22)
In \[C{{H}_{2}}OHCHOHCHOHCHOHCHOHCHO\], the number of optical isomers will be
A)
16 done
clear
B)
8 done
clear
C)
32 done
clear
D)
4 done
clear
View Solution play_arrow
-
question_answer23)
| Some statements are given below : |
| 1. Glucose is aldohexose. |
| 2. Naturally occurring glucose is dextrorotatory. |
| 3. Glucose contains three chiral centres. |
| 4. Glucose contains one \[1{}^\circ \]alcoholic group and four \[2{}^\circ \]alcoholic groups. |
| Among the above, correct statements are |
A)
1 and 2 only done
clear
B)
3 and 4 only done
clear
C)
1, 2 and 4 only done
clear
D)
1, 2, 3 and 4 done
clear
View Solution play_arrow
-
question_answer24)
Two hexoses form the same osazone, find the correct statement about these hexoses.
A)
Both of them must be aldoses. done
clear
B)
They are epimers at \[C-3\]. done
clear
C)
The carbon atoms 1 and 2 in both have the same configuration. done
clear
D)
The carbon atoms 3, 4 and 5 in both have the same configuration. done
clear
View Solution play_arrow
-
question_answer25)
Which of the following reactions of glucose can be explained only by its cyclic structure?
A)
Glucose forms cyanohydrin with HCN. done
clear
B)
Glucose reacts with hydroxylamine to form an oxime. done
clear
C)
Pentaacetate of glucose does not react with hydroxylamine. done
clear
D)
Glucose is oxidised by nitric acid to gluconic acid. done
clear
View Solution play_arrow


