-
question_answer1)
| Direction (1 - 5): Read the given passages and answer the questions that follow. |
| There are certain substances that are neither good conductors (metals) nor insulators (glass). A substance which has crystalline structure and contains very few electrons at room temperature is called semiconductor. At room temperature, it behaves like an insulator. Its resistivity lies between that of conductor and insulator. If suitable impurities are added to the semiconductor, controlled conductivity can be provided. Some examples of semiconductors are silicon, germanium etc. |
| Current conduction in a semiconductor occurs through the movement of free electrons and holes collectively known as charge carriers. Adding impurity atoms to a semiconducting material, known as 'doping', greatly increases the number of charge carriers within it. When a doped semiconductor contains mostly free holes it is called "p-type" and when it contains mostly free electrons it is known as "n-type". |
| (Reference: Md. F. Rahman (2014). A Review on Semiconductors Including Applications and Temperature Effects in Semiconductors. American Scientific Research Journal for Engineering, Technology, and Sciences, Vol 7, No. 1, pp 50-70) |
Silicon doped with electron-rich impurity forms:
A)
p-type semiconductor done
clear
B)
n-type semiconductor done
clear
C)
intrinsic semiconductor done
clear
D)
insulator done
clear
View Solution play_arrow
-
question_answer2)
Which of the following represents correct order of conductivity in solids ?
A)
\[{{K}_{metals}}>>{{K}_{insulators}}<{{K}_{semiconductors}}\] done
clear
B)
\[{{K}_{metals}}<<{{K}_{insulators}}<{{K}_{semiconductors}}\] done
clear
C)
\[{{K}_{metals}}={{K}_{semiconductors}}<{{K}_{insulators}}=zero\] done
clear
D)
\[{{K}_{metals}}<{{K}_{insulators}}<{{K}_{semiconductors}}\ne zero\] done
clear
View Solution play_arrow
-
question_answer3)
Which of the following is true about the charge acquired by p-type semiconductors?
A)
positive done
clear
B)
neutral done
clear
C)
negative done
clear
D)
depends on concentration of p-type impurity done
clear
View Solution play_arrow
-
question_answer4)
|
Q.4 and Q.5 are assertion-reason type. Choose the correct answer out of the following choices:
|
|
Assertion: Semi-conductors are solids with conductivities in the intermediate range from \[{{10}^{-6}}-{{10}^{4}}\,oh{{m}^{-1}}\,m\]
|
|
Reason: Intermediate conductivity in semiconductor is due to partially filled valence band.
|
A)
Assertion and reason both are correct statements and reason is correct explanation for assertion. done
clear
B)
Assertion and reason both are correct statements but reason is not correct explanation for assertion. done
clear
C)
Assertion is correct statement but reason is wrong statement. done
clear
D)
Assertion is wrong statement but reason is correct statement. done
clear
View Solution play_arrow
-
question_answer5)
| Assertion: Silicon doped with boron form n-type semiconductor. |
| Reason: Boron in silicon behaves as an electron deficient impurity. |
A)
Assertion and reason both are correct statements and reason is correct explanation for assertion. done
clear
B)
Assertion and reason both are correct statements but reason is not correct explanation for assertion. done
clear
C)
Assertion is correct statement but reason is wrong statement. done
clear
D)
Assertion is wrong statement but reason is correct statement. done
clear
View Solution play_arrow
-
question_answer6)
| Direction (6 - 9): Point defects are the irregularities from ideal arrangement around a point or an atom in a crystalline substance. In ionic solids. Frenkel defect is shown when the smaller ion is dislocated from its normal site while Schottky defect is basically a vacancy defect. (Source N.C.E.R.T. Book) |
 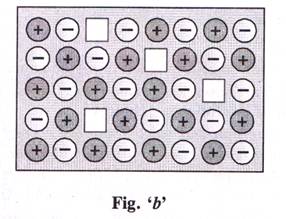 |
The defect shown by ionic compounds as in fig. 'a' and 'b' are called respectively as
A)
Schottky, Frenkel done
clear
B)
Frenkel, Schottky done
clear
C)
Electronic defects, F-centre done
clear
D)
None of these. done
clear
View Solution play_arrow
-
question_answer7)
What is the effect of these defects on the density of the solid?
A)
Increases, increases done
clear
B)
Decreases, decreases done
clear
C)
No change, decreases done
clear
D)
Decreases, no change. done
clear
View Solution play_arrow
-
question_answer8)
Which ionic compound can show defect shown in Fig. 'b'.
A)
ZnS done
clear
B)
AgCl done
clear
C)
NaCI done
clear
D)
None of these. done
clear
View Solution play_arrow
-
question_answer9)
Which of the following defects is also known as dislocation defect?
A)
Frenkel defect done
clear
B)
Schottky defect done
clear
C)
Non-stoichiometric defect done
clear
D)
Simple interstitial defect. done
clear
View Solution play_arrow
-
question_answer10)
| Direction (10 - 14): No crystal is found to be perfect at room temperature. The defects present in the crystals can be stoichiometric or non-stoichiometric. Due to non-stoichiometric defects, the formula of the ionic compound is different from the ideal formula. For example, the ideal formula of ferrous oxide should be FeO but actually in one sample, it was found to be \[F{{e}_{0.93}}O\]. This is because the crystal may have some ferric ions in place of frrous ions. These defects change the properties of the crystals. In some cases, defects are introduced to have crystals of desired properties as required in the field of electronics. Doping of elements of group 14 with those of group 13 or 15 is most common. In ionic compounds, usually the impurities are introduced in which the cation has higher valency than the cation of the parent cyrstal e.g., \[SrC{{l}_{2}}\] into NaCI. |
Which of the following dopings will produce p-type semiconductor?
A)
Silicon doped with arsenic done
clear
B)
Germanium doped with phosphorus done
clear
C)
Germanium doped with aluminium done
clear
D)
Silicon doped with phosphorus. done
clear
View Solution play_arrow
-
question_answer11)
Zinc oxide (ZnO) is white when cold and yelllow when hot. It is due to the development of :
A)
Frenkel defect done
clear
B)
Schottky defect done
clear
C)
Metal excess defect done
clear
D)
Metal deficiency defect. done
clear
View Solution play_arrow
-
question_answer12)
NaCI was doped with \[{{10}^{-3}}\] mol % \[SrC{{l}_{2}}\]. The concentration of the cation vacancies is
A)
\[6.02\times {{10}^{18}}\text{ }mo{{l}^{-1}}\] done
clear
B)
\[6.02\times {{10}^{15}}\text{ }mo{{l}^{-1}}\] done
clear
C)
\[6.02\times {{10}^{21}}\text{ }mo{{l}^{-1}}\] done
clear
D)
\[6.02\times {{10}^{12}}\text{ }mo{{l}^{-1}}\] done
clear
View Solution play_arrow
-
question_answer13)
A metal oxide has empirical formula \[{{M}_{0.96}}{{O}_{1.00}}\]. What will be the precentage of \[{{M}^{2+}}\] ions in the crystal?
A)
\[90\centerdot 67\] done
clear
B)
\[91\centerdot 67\] done
clear
C)
\[8\centerdot 33\] done
clear
D)
\[9\centerdot 33\] done
clear
View Solution play_arrow
-
question_answer14)
Which type of 'defect' has the presence of cations in the interstitial sites?
A)
Schottky defect done
clear
B)
Frenkel defect done
clear
C)
Vacancy defect done
clear
D)
Metal deficiency defect done
clear
View Solution play_arrow
-
question_answer15)
| Direction (15 - 19): A student of class XII studied the close packing of spheres, with vacant sites (or voids) known as holes. These are of two types i. e. tetrahedral and octahedral voids. A tetrahedral void is formed when four spheres placed at the corners of a tetrahedron touch each other. An octahedral void is created, when three spheres arranged in the corners of an equilateral triangle are placed over another similar set of three spheres pointing in the opposite direction. The actual size of these voids is linked to that of the spheres i.e. greater the size of the spheres, more will be the size of the voids. |
| In the following questions, a statement of assertion followed by a statement of reason is given. Choose the correct answer out of the following choices : |
| Assertion: Hexagonal close packing is more tight than cubic close packing. |
| Reason: Co-ordination number in both the cases is 12. |
A)
Assertion and reason both are correct statements and reason is correct explanation for assertion. done
clear
B)
Assertion and reason both are correct statements but reason is not correct explanation for assertion. done
clear
C)
Assertion is correct statement but reason is wrong statement. done
clear
D)
Assertion is wrong statement but reason is correct statement. done
clear
View Solution play_arrow
-
question_answer16)
| Assertion: In a close packing of spheres, a tetrahedral hole is surrounded by four spheres whereas an octahedral hole is surrounded by six spheres. |
| Reason: A tetrahedral void has a tetrahedral shape while an octahedral void has an octahedral shape. |
A)
Assertion and reason both are correct statements and reason is correct explanation for assertion. done
clear
B)
Assertion and reason both are correct statements but reason is not correct explanation for assertion. done
clear
C)
Assertion is correct statement but reason is wrong statement. done
clear
D)
Assertion is wrong statement but reason is correct statement. done
clear
View Solution play_arrow
-
question_answer17)
| Assertion: In NaCI crystal, each \[N{{a}^{+}}\] ion is touching six \[C{{l}^{-}}\] ions but these \[C{{l}^{-}}\] ions donot touch each other. |
| Reason: The ratius ration \[{{r}_{Na}}+/{{r}_{C{{l}^{-}}}}\] is greater than 0.414 required for exact fitting. |
A)
Assertion and reason both are correct statements and reason is correct explanation for assertion. done
clear
B)
Assertion and reason both are correct statements but reason is not correct explanation for assertion. done
clear
C)
Assertion is correct statement but reason is wrong statement. done
clear
D)
Assertion is wrong statement but reason is correct statement. done
clear
View Solution play_arrow
-
question_answer18)
| Assertion: In NaCI structure, \[N{{a}^{+}}\] ions occupy octahedral holes and \[C{{l}^{-}}\] ions occupy c.c.p. |
| Reason: The distance of the nearest neighbours in NaCI structure is all, where a/2, is the edge length of the cube. |
A)
Assertion and reason both are correct statements and reason is correct explanation for assertion. done
clear
B)
Assertion and reason both are correct statements but reason is not correct explanation for assertion. done
clear
C)
Assertion is correct statement but reason is wrong statement. done
clear
D)
Assertion is wrong statement but reason is correct statement. done
clear
View Solution play_arrow
-
question_answer19)
| Assertion: The number of tetrahedral voids is double the number of octahedral voids, |
| Reason: The size of the tetrahedral voids is half than that of the octahedral voids. |
A)
Assertion and reason both are correct statements and reason is correct explanation for assertion. done
clear
B)
Assertion and reason both are correct statements but reason is not correct explanation for assertion. done
clear
C)
Assertion is correct statement but reason is wrong statement. done
clear
D)
Assertion is wrong statement but reason is correct statement. done
clear
View Solution play_arrow
