A) \[Conc.\,HCl,\,C{{O}_{2}}\]
B) \[Conc.\,{{H}_{2}}S{{O}_{4}},HCl\,gas\]
C) \[Conc.\,HN{{O}_{3}},{{H}_{2}}\]
D) \[Conc.\,NaOH,\,C{{l}_{2}}\]
Correct Answer: B
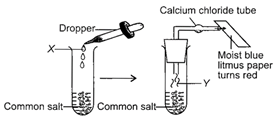
Solution :
\[\underset{\begin{smallmatrix} Common \\ salt \end{smallmatrix}}{\mathop{2NaC{{l}_{(aq.)}}}}\,+\underset{\begin{smallmatrix} Sulphuric\,acid \\ \,\,(X) \end{smallmatrix}}{\mathop{{{H}_{2}}S{{O}_{4}}(conc.)}}\,\xrightarrow{{}}\underset{\begin{smallmatrix} Sodium \\ sulphate \end{smallmatrix}}{\mathop{N{{a}_{2}}S{{O}_{4(aq)}}}}\,+\underset{\begin{smallmatrix} Hydrogen \\ chloride\,gas \\ \,\,\,\,\,\,(y) \end{smallmatrix}}{\mathop{2HC{{l}_{(g)}}}}\,\] Dry HCI gas turns moist blue litmus paper red as it is acidic in nature.You need to login to perform this action.
You will be redirected in
3 sec
