A) Nitrobenzene
B) Phenol
C) Anisole
D) Chlorobenzene
Correct Answer: A
Solution :
Nitrobenzene is least reactive towards bromine because of presence of \[-N{{O}_{2}}\] group decreases electron density at o and p positions and hence attack of electrophile on the benzene nucleus is difficult because + ve charge on o and p position repel the incoming electrophile.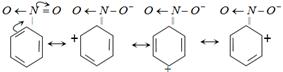
You need to login to perform this action.
You will be redirected in
3 sec
