A) There are two bridging hydrogen atoms in diborane
B) Each boron atom forms four bonds in diborane
C) The hydrogen atoms are not in the same plane in diborane
D) All B - H bonds in diborane are similar
Correct Answer: D
Solution :
\[{{B}_{2}}{{H}_{6}}\] has two types of \[B-H\] bonds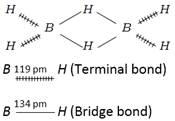
You need to login to perform this action.
You will be redirected in
3 sec
