| Direction (16 - 20): The cumene-phenol process is the most important commercial method for the preparation of phenol and is carried out by the oxidation of cumene to its hydroperoxide, which is then decomposed by acid. The other synthetic methods available are from chlorobenzene and sodium benzene sulphonate. Phenol is a colourless crystalline solid which turns pink on exposure to air and light. Many of the reactions of phenol are those which are example of electrophilic aromatic substitution and since the hydroxyl group is o-Ip- orienting, these are always the products formed. |
| (I. L Finar, Organic Chemistry Vol I: The fundamental principles. Sixth Edition) |
A)

B)

C)
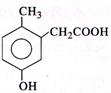
D)
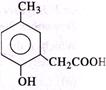
Correct Answer: B
Solution :

You need to login to perform this action.
You will be redirected in
3 sec
