| The correct decreasing order of \[p{{K}_{a}}\] value is |
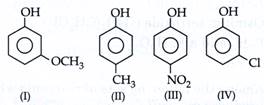 |
A) II>IV>I>III
B) IV>II>III>I
C) III>II>IV>I
D) IV>I>II>III
Correct Answer: A
Solution :
Weaker acids have higher \[p{{K}_{a}}\]. \[-OC{{H}_{3}}\]at mete-position exerts only-l effect, hence increases the acidity. -l effect order: \[-N{{O}_{2}}>-OC{{H}_{3}}>-Cl\] - \[C{{H}_{3}}\]has + l effect. So, order is [a].You need to login to perform this action.
You will be redirected in
3 sec
