| Directions: (21-25) |
| Millikan's Oil-Drop Experiment |
| In 1909, Robert Millikan was the first to find the charge of an electron in his now-famous oil-drop experiment. In that experiment, tiny oil drops were sprayed into a uniform electric field between a horizontal pair of oppositely charged plates. The drops were observed with a magnifying eyepiece, and the electric field was adjusted so that the upward force on some negatively charged oil drops was just sufficient to balance the downward force of gravity. That is, when suspended, upward force qE just equaled Mg. Millikan accurately measured the charges on many oil drops and found the values to be whole number multiples of \[1.6\times {{10}^{-19}}\text{ }C\] the charge of the electron. For this, he won the Nobel prize. |
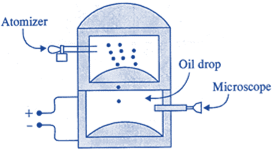 |
A) \[6\centerdot 40\times {{10}^{-19}}C\]
B) \[3\centerdot 2\times {{10}^{-19}}C\]
C) \[1\centerdot 6\times {{10}^{-19}}C\]
D) \[4\centerdot 8\times {{10}^{-19}}C.\]
Correct Answer: A
Solution :
As, \[qE=mg\Rightarrow q=\frac{1.08\times {{10}^{-14}}\times 9\,.\,8}{1\,.\,68\times {{10}^{5}}}\] \[=6\,.\,4\times {{10}^{-19}}C\]You need to login to perform this action.
You will be redirected in
3 sec
