A) \[C{{H}_{3}}CH=CHCI\]
B) \[{{\left( {{C}_{6}}{{H}_{5}} \right)}_{3}}CCl\]
C) \[ClC{{H}_{2}}CH=C{{H}_{2}}\]
D)
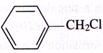
Correct Answer: B
Solution :
This is linked with the relative stabilities of the carbocation intermediates formed in the slow or rate determining step. The increasing order of stability is| [a] \[C{{H}_{3}}-CH=\overset{\oplus }{\mathop{CH}}\,\](Vinylic) (Least stable) |
[b]  |
| [c] \[ClC{{H}_{2}}CH=C{{H}_{2}}\] |
[d] 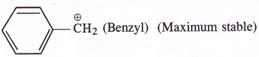 |
You need to login to perform this action.
You will be redirected in
3 sec
