A) \[{{I}_{2}}\] is an oxidant.
B) \[{{I}_{2}}\] is reduced.
C) \[HN{{O}_{3}}\] is oxidized.
D) \[HN{{O}_{3}}\] is reduced.
Correct Answer: D
Solution :
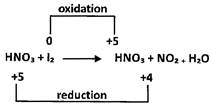 \[HN{{O}_{3}}-\]oxidizing agent or oxidant \[{{I}_{2}}-\] Reducing agent
\[HN{{O}_{3}}-\]oxidizing agent or oxidant \[{{I}_{2}}-\] Reducing agent
You need to login to perform this action.
You will be redirected in
3 sec
