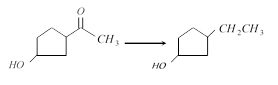 [IIT-JEE (Screening) 2000]
[IIT-JEE (Screening) 2000]
A) \[Zn(Hg),\,HCl\]
B) \[N{{H}_{2}}N{{H}_{2}}O{{H}^{-}}\]
C) \[{{H}_{2}}/Ni\]
D) \[NaB{{H}_{4}}\]
Correct Answer: B
Solution :
Both \[Zn(Hg),\,HCl\] and \[N{{H}_{2}}N{{H}_{2}},\,\,O{{H}^{-}}\] can reduce \[-COC{{H}_{3}}\] group to \[-C{{H}_{2}}-C{{H}_{3}}\] group, but HCl will also bring about dehydration of alcohol to form alkene. Therefore, appropriate reagent for the conversion is \[N{{H}_{2}}N{{H}_{2}},\,\,O{{H}^{-}}\].You need to login to perform this action.
You will be redirected in
3 sec
