A) t-butyl chloride
B) s-butyl chloride
C) Isobutyl chloride
D) n-butyl chloride
E) None of these
Correct Answer: A
Solution :
\[2C{{H}_{3}}-\underset{Cl\ \ \ \ \ \ \,}{\overset{C{{H}_{3}}\ \ \ \ }{\mathop{\underset{|}{\overset{|}{\mathop{C}}}\,-C{{H}_{3}}}}}\,\underset{2Na}{\mathop{\xrightarrow{\text{ether}}}}\,C{{H}_{3}}-\underset{C{{H}_{3}}}{\overset{C{{H}_{3}}}{\mathop{\underset{|}{\overset{|}{\mathop{C}}}\,\,--}}}\,\underset{C{{H}_{3}}\ \ \ \ \ }{\overset{C{{H}_{3}}\ \ \ \ \,}{\mathop{\underset{|}{\overset{|}{\mathop{C}}}\,-C{{H}_{3}}}}}\,+2NaCl\]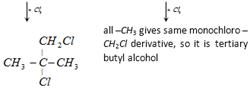
You need to login to perform this action.
You will be redirected in
3 sec
