A) Carbonium ion
B) Alkoxy ion
C) Alkyl hydrogen sulphate
D) None of these
Correct Answer: A
Solution :
\[-\underset{\underset{H}{\mathop{|}}\,}{\overset{|}{\mathop{C}}}\,\,\,\,\,\,-\underset{\underset{\,\,\,\,OH}{\mathop{|}}\,}{\overset{|}{\mathop{C}}}\,-\]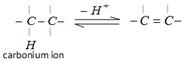 In all cases intermediate is carbonium ion, and there may be 1, 2-hydride or 1, 2-methyl shift to form more stable carbonium ion.
In all cases intermediate is carbonium ion, and there may be 1, 2-hydride or 1, 2-methyl shift to form more stable carbonium ion.
You need to login to perform this action.
You will be redirected in
3 sec
